Erectile dysfunction (ED) is often treated as a local mechanical problem with a systemic medication. That sentence sounds contradictory, but it’s an accurate summary of modern practice. We prescribe phosphodiesterase type 5 inhibitors (PDE5 inhibitors) to improve penile blood flow, while quietly recognizing that ED is frequently a vascular and endothelial disorder long before it is a purely sexual complaint. In many men, ED is not the first problem—it is the first symptom the patient is willing to discuss.
This is where folic acid (FA) enters the conversation, not as a wellness trend but as a biologically plausible marker and potential adjunct. Folic acid participates in nitric oxide (NO) biology, endothelial function, and homocysteine metabolism—three topics that repeatedly show up whenever vascular ED is discussed seriously. The systematic review and meta-analysis you provided was built to answer a very practical question: are serum folic acid levels different in men with ED compared with healthy controls, does folic acid track with ED severity, and can supplementation improve erectile function scores?
The conclusions were clear enough to warrant attention, but cautious enough to avoid overselling. Across six studies comparing folate levels in ED patients vs controls (982 ED patients and 860 controls), folic acid was lower in ED (pooled standardized mean difference, SMD −0.94). Severity analysis suggested folic acid levels decline as ED becomes more severe. Supplementation studies showed improved IIEF-5 scores, and one randomized trial in men with type 2 diabetes suggested that adding folic acid to tadalafil improved IIEF changes more than tadalafil plus placebo—yet the authors themselves warned that statistical improvements may not always meet the bar for clinical significance.
This article explains what these results mean, how to apply them safely, and how to talk about folic acid in ED without turning evidence-based medicine into a vitamin commercial.
The Hidden Theme of ED: Endothelial Dysfunction Is Often the Real Diagnosis
Vascular ED is the most common ED subtype, and its risk factors read like a cardiology intake form: obesity, diabetes mellitus, hypertension, dyslipidemia, metabolic syndrome, smoking, and sedentary lifestyle. The bridge connecting these factors to ED is endothelial dysfunction—the loss of the endothelium’s ability to regulate vascular tone, platelet activity, inflammation, and smooth muscle behavior.
Erection physiology depends on nitric oxide. NO is generated by endothelial nitric oxide synthase (eNOS) and triggers the cGMP pathway, enabling cavernosal smooth muscle relaxation and increased penile blood flow. When endothelial function is impaired, eNOS activity and NO availability decline, and the cGMP pathway loses its upstream fuel. PDE5 inhibitors help by preventing cGMP breakdown, but they can’t fully compensate for a system that isn’t producing enough NO to begin with.
This is why clinicians keep searching for “modifiable” biochemical factors that affect endothelial function. Testosterone, vitamin D, inflammatory indices, platelet parameters, and metabolic markers all appear in the literature. The folic acid story belongs in this category: not a magical cure, but a potentially important piece of endothelial health that might influence erectile physiology—especially in vasculogenic ED.
Why Folic Acid Is Biologically Plausible: NO, eNOS Uncoupling, BH4, and Homocysteine
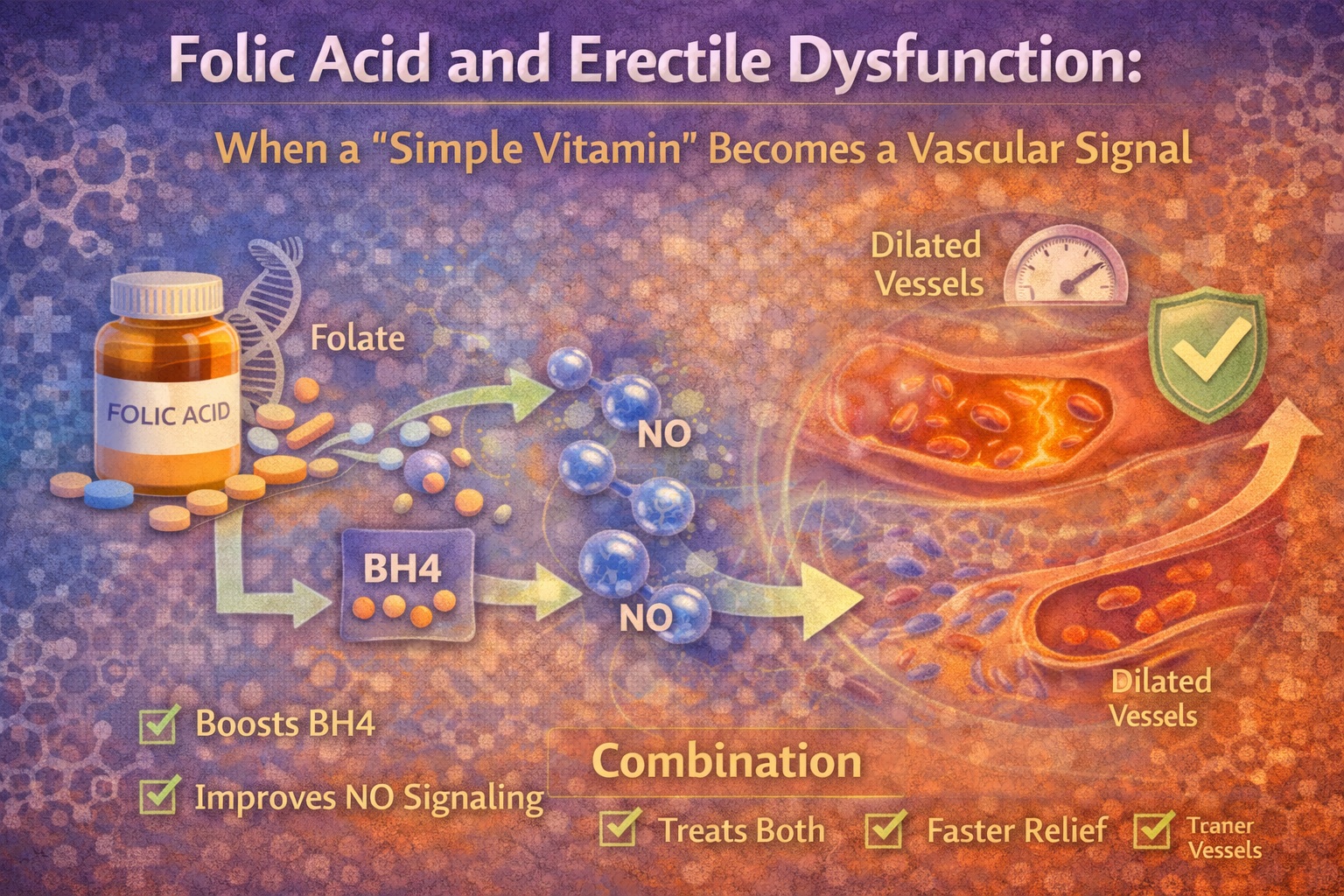
Folic acid is not a random nutrient chosen because it’s easy to measure. The meta-analysis explains why folate plausibly matters in ED through two interconnected pathways.
The first pathway is eNOS coupling. When eNOS is “coupled,” it produces NO efficiently. When eNOS becomes “uncoupled,” it can generate reactive oxygen species instead of NO, worsening endothelial dysfunction. The review notes that folic acid has been described as helping reverse eNOS uncoupling and improving NO metabolism, which makes it biologically aligned with endothelial-dependent erection physiology.
The second pathway involves tetrahydrobiopterin (BH4), an essential eNOS cofactor. BH4 supports NO synthesis and stabilizes eNOS function. The discussion connects folate deficiency to BH4 deficiency and describes how BH4 deficiency can promote eNOS uncoupling, thereby reducing NO and worsening endothelial dysfunction—an elegant mechanism that fits ED physiology tightly.
Homocysteine (Hcy) is the third player. Folate is involved in remethylation of homocysteine to methionine. Hyperhomocysteinemia has been discussed as a novel ED risk factor due to inhibitory effects on endothelium-dependent NO formation. Many studies interpret folate deficiency as a driver of elevated homocysteine, worsening endothelial function and contributing to ED. The authors also acknowledge complexity: at least one study reported no clear folate–homocysteine correlation in ED and controls, supporting the possibility that folate could be an independent risk factor rather than merely a homocysteine proxy.
If you want the clinical translation without biochemistry poetry: folate is plausibly linked to ED because it influences how well the endothelium produces NO, and erections are fundamentally an NO-dependent vascular event.
What the Meta-Analysis Found: Lower Folic Acid in ED, and a Severity Gradient That Looks Clinically Meaningful
The review included nine eligible studies (published up to October 31, 2020). Six studies compared folate levels in ED patients versus healthy controls; two evaluated IIEF-5 changes before and after folic acid supplementation; one evaluated IIEF changes comparing tadalafil plus folic acid versus tadalafil plus placebo. Study quality was assessed with the Newcastle–Ottawa Scale (NOS), and most studies scored in the moderate-to-high range.
The most direct result was the pooled difference in folate between ED patients and controls: SMD −0.94 (95% CI −1.59 to −0.30, p = 0.004). The heterogeneity was very high (I² ~98%), meaning the size of the effect varied substantially across studies. The authors performed sensitivity analysis and found the overall conclusion was stable when omitting studies one at a time, and Begg’s test did not show significant publication bias (p ≈ 0.06).
The severity analysis is where the paper becomes clinically interesting. Three studies stratified ED by IIEF-5 severity (mild 17–21, moderate 8–16, severe 1–7) and compared folate levels. Mild ED did not differ significantly from controls, but moderate and severe ED showed significantly lower folate than healthy subjects (SMD about −1.41 and −1.42, respectively). This suggests that folate deficiency is not necessarily a universal feature of all ED, but may become more prominent as ED severity increases—or may be more detectable when endothelial dysfunction is advanced enough to dominate the phenotype.
The authors also synthesized absolute folate level estimates by group, and the gradient is easy to remember because it behaves like a disease marker should: healthy controls around 11.847, mild ED around 9.496, moderate ED around 6.597, and severe ED around 5.623 (units presented as in the paper). These group estimates are displayed in the figures on pages 8–9 (Figures 5.1–5.4), and the pattern is the headline: lower folate tracks with worse ED.
Can Folic Acid Improve Erectile Function? The Signal Is Positive, but the Clinical Confidence Is Not Maximal
The supplementation evidence is smaller and therefore trickier. Two studies evaluated IIEF-5 before and after folic acid administration over three months (doses around 0.4–0.5 mg). The pooled effect showed a strong standardized improvement in IIEF-5 (SMD 1.89, 95% CI 1.60–2.17) with no heterogeneity. On paper, that looks impressive. In clinic, you still need to ask: how much did the score change in real terms, and was that change meaningful to patients?
The review itself injects an important caution: improvements in IIEF-5 or IIEF might be statistically significant yet still fall below a threshold for clinical significance. That is not a dismissal—it is a reminder that patient-reported outcomes can swing with expectation, adherence, placebo effects, and the sheer relief of being treated.
The most actionable trial for daily practice is the randomized study in men with type 2 diabetes: tadalafil (10 mg) combined with folic acid (5 mg) produced larger IIEF changes than tadalafil plus placebo over three months (mean IIEF change 5.14 vs 1.68). This is particularly relevant because diabetes-related ED is heavily driven by endothelial dysfunction and impaired NO availability, which is exactly where folate’s mechanistic rationale fits. Still, the evidence is based on one study, so it is hypothesis-strengthening rather than practice-defining.
A sensible clinical framing, consistent with the paper’s tone, is: folate supplementation may improve erectile function in selected men, especially those with low folate and vasculogenic features, but it should be considered an adjunct rather than a substitute for established ED evaluation and therapy.
How to Use These Findings in Real Practice: Screening, Interpretation, and Safe Supplementation
The paper makes a practical observation that many clinicians will recognize: when an ED patient sees an andrologist, labs often include testosterone, glucose, and lipid profile to identify reversible risk factors. The authors suggest that serum folic acid is worthy of consideration as another risk factor to check, because low folate may contribute to ED pathogenesis and may indicate endothelial vulnerability.
Clinically, folate testing is most useful when interpreted as part of a vascular risk and nutrition profile, not as a stand-alone ED biomarker. If folate is low, the next question is why. Dietary insufficiency, alcohol use, malabsorption, certain medications, and broader nutritional patterns matter. In addition, measuring related variables—such as vitamin B12 and homocysteine—may help contextualize the finding, especially when folate deficiency is suspected to contribute through homocysteine pathways. The review notes the complex relationship between folate and homocysteine in ED populations, reinforcing the value of integrated interpretation rather than automatic assumptions.
As for supplementation, the included studies used relatively modest folic acid doses (0.4–0.5 mg in two trials, 5 mg in the tadalafil-combination diabetes trial). In general medical practice, folic acid is widely used and typically well tolerated, but it should still be prescribed thoughtfully—particularly because high-dose folate can mask vitamin B12 deficiency in some contexts. The paper does not provide a universal dose recommendation, and it shouldn’t; dosing should depend on deficiency status, dietary intake, comorbidities, and whether folate is being used as nutritional correction or as an adjunct therapeutic experiment.
Finally, folic acid should not become a distraction from core ED management. If a man has ED, the clinician should still evaluate cardiovascular risk, metabolic health, testosterone when indicated, medication contributors, psychological factors, and relationship context. Folate can be a useful signal and possible adjunct—nothing more, nothing less.
Limitations: The Evidence Is Promising, but It Is Not Yet “Prescribe for Everyone”
The authors are very clear about limitations, and they deserve to be repeated because they shape how responsibly we apply the findings.
First, the supplementation evidence is limited: only three studies contributed to IIEF outcome analyses (two pre/post folate studies and one tadalafil+folate trial). Second, heterogeneity in the folate-level meta-analysis was extreme. That heterogeneity likely reflects differences in sample size, age, ED duration, baseline health, and study design. The conclusion appears stable, but variability means you should not treat the effect size as precise.
Third, ED diagnosis in included studies was based on questionnaires (IIEF-5), not on physiologic testing such as penile Doppler ultrasound or RigiScan. Questionnaire-based diagnosis is clinically valid, but it can blur etiologic subtyping. Folate’s relevance might be strongest in vasculogenic ED, and a mixed ED population could dilute or distort associations.
Lastly, association is not causation. Low folate could be a marker of worse health status, poorer diet quality, metabolic syndrome, or inflammatory burden rather than a direct driver. The paper argues folate is likely an independent risk factor, but definitive causality would require well-designed prospective cohorts and larger randomized trials with clinically meaningful endpoints.
Conclusion: Folic Acid Looks Like a Vascular Clue—and Possibly a Useful Adjunct—Not a Stand-Alone ED Cure
This systematic review and meta-analysis supports a consistent association: men with erectile dysfunction tend to have lower serum folic acid than healthy controls, and folate levels appear to decrease with increasing ED severity. The biologic rationale aligns with endothelial function and NO signaling, offering a coherent mechanistic framework rather than a random statistical finding.
Supplementation data suggest that folic acid may improve erectile function scores, and one randomized trial suggests it can enhance tadalafil’s effect in men with type 2 diabetes. However, the evidence base for treatment efficacy remains small, and the authors correctly caution that statistically significant score changes may not always translate into clinically meaningful improvement.
The responsible clinical approach is therefore balanced: consider serum folate as part of the evaluation of men with ED—especially those with vasculogenic risk factors—correct deficiency when present, and consider folate as a low-risk adjunct in selected cases, while still treating ED as the systemic vascular health signal it often is.
FAQ
1) Should every man with erectile dysfunction take folic acid?
Not automatically. The meta-analysis shows an association between lower folate and ED and suggests possible benefit from supplementation, but treatment evidence is limited and not definitive. Testing folate and correcting deficiency is more rational than universal supplementation.
2) Does low folic acid cause erectile dysfunction, or is it just a marker?
The analysis supports folate as an independent risk factor and describes plausible mechanisms (NO metabolism, eNOS uncoupling, BH4), but most included studies are observational, so causality is not conclusively proven. Low folate may also reflect broader vascular or nutritional risk.
3) Can folic acid enhance the effect of tadalafil or other PDE5 inhibitors?
There is evidence from one randomized trial in men with type 2 diabetes that folic acid plus tadalafil improved IIEF changes more than tadalafil plus placebo over three months. This is promising but needs confirmation in larger trials and broader populations.