Introduction: From Vascular Drug to Renal Modulator
Tadalafil has long been associated with vascular medicine—most notably erectile dysfunction and pulmonary arterial hypertension. Its mechanism, centered on enhancing nitric oxide (NO)–mediated signaling, has been thoroughly studied in the context of smooth muscle relaxation and blood flow regulation. However, as is often the case in pharmacology, a drug’s story rarely ends where it begins.
In recent years, attention has shifted toward the potential renoprotective effects of tadalafil, particularly in the context of diabetic kidney disease. This shift is not accidental. Diabetes is not merely a metabolic disorder; it is a systemic disease characterized by oxidative stress, endothelial dysfunction, and progressive organ damage. The kidney, with its dense microvascular network, is especially vulnerable.
Among the cellular players in diabetic nephropathy, podocytes occupy a central role. These highly specialized cells form a critical component of the glomerular filtration barrier. When podocytes are injured, proteinuria develops, and the progression toward chronic kidney disease accelerates.
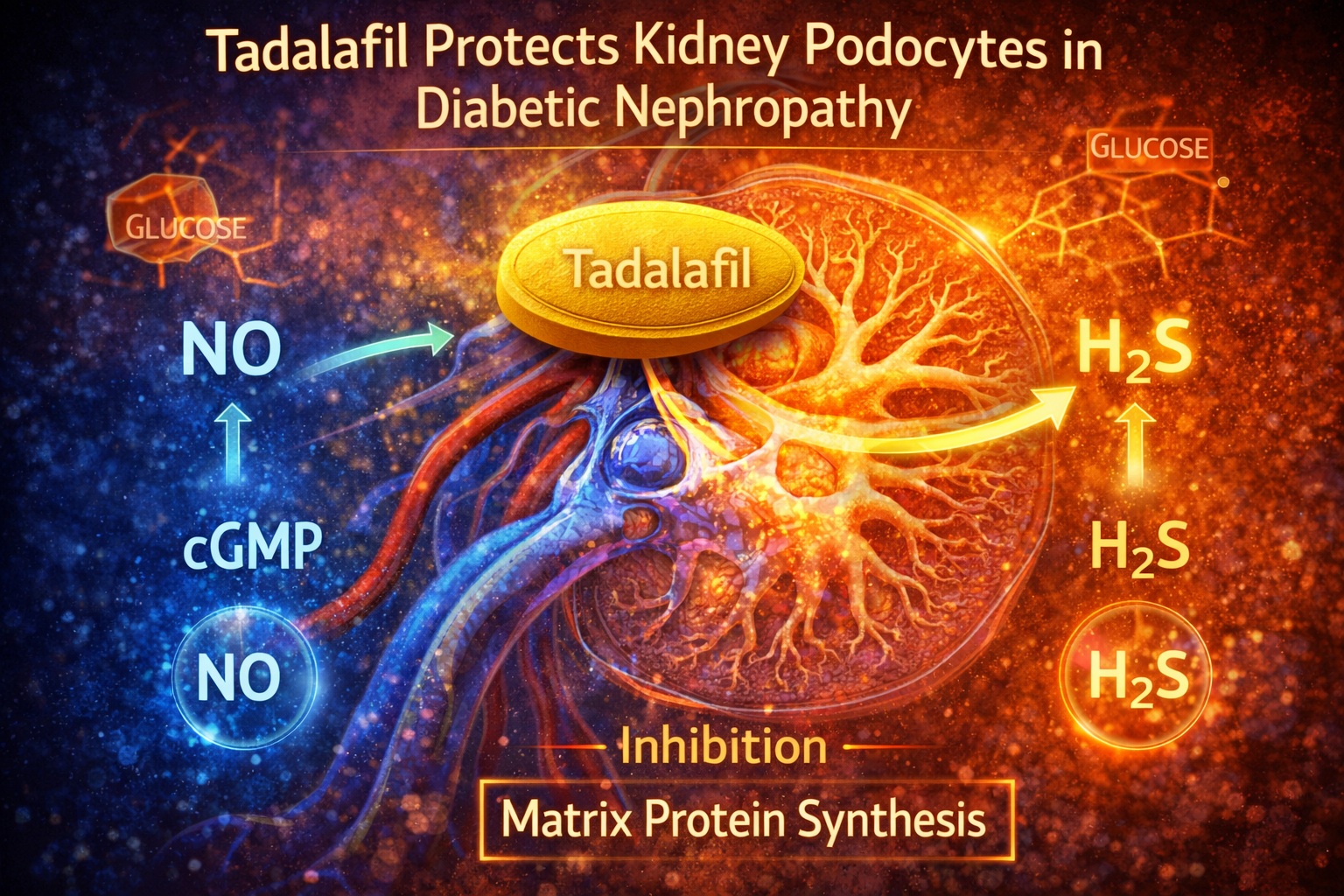
A growing body of research suggests that tadalafil may influence podocyte biology through mechanisms extending beyond classical NO signaling. Specifically, it appears to integrate the activity of nitric oxide and hydrogen sulfide (H₂S)—two gaseous signaling molecules with profound effects on cellular function.
This article explores how tadalafil interacts with these signaling pathways to inhibit high glucose–induced matrix protein synthesis in podocytes. In doing so, it reveals a fascinating intersection between vascular pharmacology and renal pathophysiology.
Diabetic Nephropathy: A Disease of Cellular Stress and Structural Damage
Diabetic nephropathy is one of the leading causes of end-stage renal disease worldwide. It develops gradually, often beginning with microalbuminuria and progressing to overt proteinuria and declining glomerular filtration rate.
At the cellular level, chronic hyperglycemia triggers a cascade of pathological events. These include oxidative stress, inflammation, and activation of fibrotic pathways. Over time, these processes lead to structural remodeling of the glomerulus.
One of the hallmark features of diabetic nephropathy is the accumulation of extracellular matrix proteins within the glomerulus. This accumulation thickens the basement membrane and disrupts normal filtration.
Podocytes play a crucial role in maintaining the integrity of the filtration barrier. When exposed to high glucose conditions, these cells undergo functional and structural changes, including increased synthesis of matrix proteins such as fibronectin and collagen.
This pathological process is not merely a passive response to hyperglycemia. It is actively driven by intracellular signaling pathways, many of which are influenced by oxidative stress and impaired gaseous signaling.
In this context, targeting the molecular mechanisms underlying matrix protein synthesis becomes a key therapeutic objective.
Podocytes: Guardians of the Glomerular Barrier
Podocytes are highly specialized epithelial cells that wrap around glomerular capillaries. Their unique structure, characterized by interdigitating foot processes, creates a filtration barrier that prevents protein leakage.
These cells are remarkably sensitive to metabolic and hemodynamic stress. Unlike many other cell types, podocytes have limited regenerative capacity. Once damaged, they are difficult to replace.
High glucose conditions, as seen in diabetes, exert direct toxic effects on podocytes. These effects include cytoskeletal disruption, altered gene expression, and increased production of extracellular matrix components.
As matrix proteins accumulate, the delicate architecture of the glomerulus is disrupted. This leads to proteinuria, a key clinical marker of diabetic kidney disease.
Protecting podocytes from high glucose–induced injury is therefore essential for preserving renal function. It is within this framework that tadalafil’s potential role becomes particularly interesting.
Nitric Oxide Signaling: A Double-Edged Sword
Nitric oxide is a fundamental regulator of vascular and cellular function. In the kidney, it modulates blood flow, glomerular filtration, and cellular signaling pathways.
Under physiological conditions, NO exerts protective effects. It promotes vasodilation, reduces oxidative stress, and inhibits inflammatory responses. However, in diabetes, NO signaling becomes dysregulated.
Hyperglycemia can impair NO production while simultaneously increasing reactive oxygen species that degrade available NO. This imbalance contributes to endothelial dysfunction and cellular injury.
Tadalafil enhances NO signaling by inhibiting PDE5, thereby increasing levels of cGMP. This effect is well established in vascular tissues, but its implications for renal cells are increasingly recognized.
In podocytes, improved NO signaling may reduce oxidative stress and inhibit pathways leading to matrix protein synthesis. However, NO alone does not fully explain the observed protective effects.
This is where hydrogen sulfide enters the picture.
Hydrogen Sulfide: The Unexpected Partner
Hydrogen sulfide (H₂S) is a gaseous signaling molecule that has gained attention for its role in cellular protection. Like nitric oxide, it modulates vascular tone, oxidative stress, and inflammatory responses.
In recent years, H₂S has been recognized as a key player in renal physiology. It influences glomerular function, protects against oxidative injury, and regulates cellular signaling pathways.
In diabetic conditions, H₂S production is often reduced. This deficiency contributes to increased oxidative stress and cellular dysfunction.
The interaction between NO and H₂S represents a complex and dynamic signaling network. These molecules can influence each other’s production and activity, creating a coordinated response to physiological stress.
Tadalafil appears to enhance this interaction. By improving NO signaling, it may indirectly promote H₂S activity, leading to a synergistic protective effect.
This integration of signaling pathways represents a sophisticated mechanism of cellular regulation.
Tadalafil’s Mechanism: Integrating NO and H₂S Pathways
Experimental studies have demonstrated that tadalafil can inhibit high glucose–induced matrix protein synthesis in podocytes. This effect appears to involve the coordinated activity of NO and H₂S signaling pathways.
When podocytes are exposed to high glucose conditions, matrix protein production increases. This process is driven by intracellular signaling pathways associated with oxidative stress and fibrosis.
Tadalafil treatment reduces the expression of these matrix proteins, suggesting a direct inhibitory effect on fibrotic pathways.
Mechanistically, this effect is associated with increased cGMP levels and enhanced NO signaling. At the same time, tadalafil appears to restore H₂S production, creating a dual protective effect.
Together, these pathways reduce oxidative stress, inhibit pro-fibrotic signaling, and preserve cellular function.
This integrated mechanism highlights the complexity of cellular responses to pharmacological intervention. It also underscores the importance of targeting multiple pathways simultaneously.
Clinical Implications: A New Role for an Established Drug
The potential renoprotective effects of tadalafil raise important questions for clinical practice. Could a drug traditionally used for erectile dysfunction play a role in preventing diabetic kidney disease?
While current evidence is primarily experimental, the implications are significant. Diabetic nephropathy remains a major clinical challenge, and new therapeutic strategies are urgently needed.
Tadalafil’s ability to modulate multiple signaling pathways positions it as a potential candidate for further investigation. Its established safety profile adds to its appeal.
However, translating these findings into clinical practice will require carefully designed trials. The complexity of diabetic kidney disease demands rigorous evaluation of both efficacy and safety.
For now, tadalafil remains a promising but exploratory option in the field of renal protection.
Conclusion: Expanding the Boundaries of Pharmacology
The study of tadalafil’s effects on podocytes illustrates a broader principle in medicine: drugs often have capabilities beyond their original purpose.
By integrating nitric oxide and hydrogen sulfide signaling, tadalafil demonstrates a sophisticated mechanism of cellular protection. This mechanism extends far beyond its traditional role in vascular medicine.
In the context of diabetic nephropathy, this represents a potential shift in therapeutic strategy—from managing symptoms to modifying disease processes at the cellular level.
As research continues, the boundaries between pharmacological categories may become increasingly blurred. Drugs will be defined not by their initial indication, but by their ability to influence complex biological systems.
And tadalafil, once known simply as a vascular agent, may yet prove to be a versatile tool in the fight against chronic disease.
FAQ
How does tadalafil affect kidney cells?
Tadalafil enhances nitric oxide signaling and appears to interact with hydrogen sulfide pathways, reducing oxidative stress and inhibiting harmful matrix protein production in podocytes.
What is the role of podocytes in kidney disease?
Podocytes are specialized cells that maintain the filtration barrier in the kidney. Damage to these cells leads to protein leakage and progression of kidney disease.
Can tadalafil be used to treat diabetic nephropathy?
Currently, tadalafil is not approved for this indication. However, experimental studies suggest it may have protective effects, and further clinical research is needed.