Introduction: When an Old Drug Finds a New Heart
Few stories in pharmacology are as illustrative as that of phosphodiesterase type 5 (PDE5) inhibitors. Originally developed for cardiovascular disease, later repurposed for erectile dysfunction, these agents—particularly tadalafil—may now be returning to their cardiovascular origins with renewed purpose.
The concept is elegant: instead of inventing entirely new drugs, medicine can sometimes repurpose existing ones by understanding their mechanisms more deeply. The article explores precisely this idea, focusing on the potential of PDE5 inhibition as a therapeutic strategy in myocardial ischaemia/reperfusion injury and heart failure.
What emerges is a fascinating narrative. Drugs once associated primarily with sexual health are now being investigated as cardioprotective agents, capable of modulating cellular survival pathways, reducing oxidative stress, and improving cardiac function.
This is not merely a pharmacological curiosity. It is a potential shift in how we think about cardiovascular therapy.
The PDE5 Pathway: A Central Regulator of Cardiovascular Function
To understand the cardioprotective potential of PDE5 inhibitors, one must begin with the biology of cyclic guanosine monophosphate (cGMP).
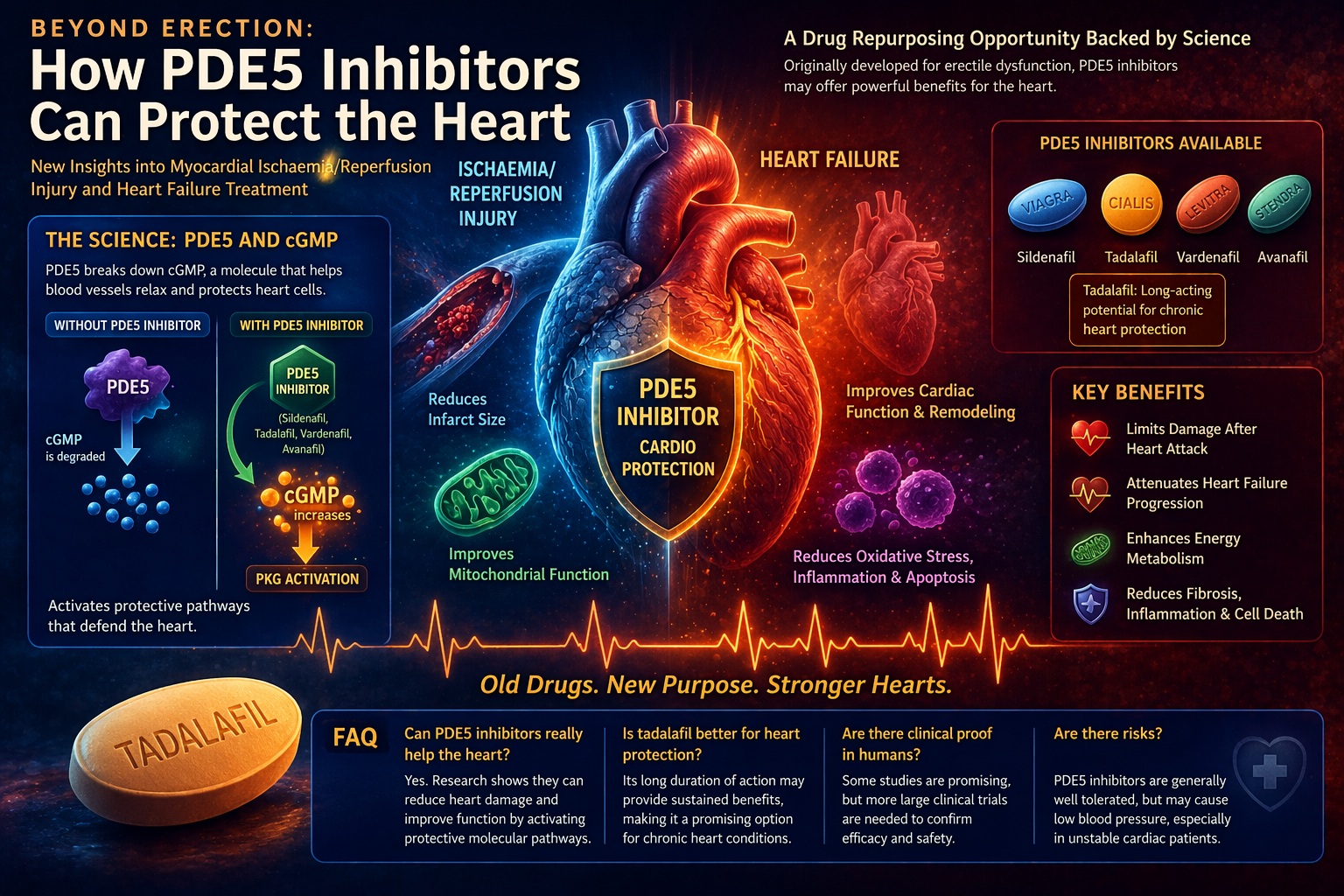
PDE5 is an enzyme responsible for the degradation of cGMP, a critical second messenger involved in vascular tone regulation. By breaking down cGMP, PDE5 effectively limits nitric oxide (NO)-mediated signaling.
Inhibition of PDE5 prevents this degradation, leading to increased intracellular cGMP levels. The result is activation of protein kinase G (PKG), which triggers a cascade of downstream effects, including reduced intracellular calcium and smooth muscle relaxation .
While this mechanism is well known in the context of penile vasodilation, it is equally relevant in the cardiovascular system. PDE5 is expressed in vascular smooth muscle, platelets, and—importantly—in diseased cardiac tissue.
In conditions such as heart failure and hypertrophy, PDE5 expression is upregulated, making it an attractive therapeutic target.
In short, the same pathway that facilitates erection may also protect the heart.
Mechanisms of Cardioprotection: Beyond Vasodilation
The cardioprotective effects of PDE5 inhibitors extend far beyond simple vasodilation. They involve a complex interplay of molecular pathways that influence cell survival, inflammation, and oxidative stress.
Activation of the NO–cGMP–PKG pathway leads to:
- Opening of mitochondrial ATP-sensitive potassium channels
- Reduction of intracellular calcium overload
- Inhibition of pro-apoptotic signaling
- Activation of protective kinases such as ERK
These mechanisms are illustrated in the diagram on page 5, which shows how PDE5 inhibition leads to reduced apoptosis, decreased inflammation, and improved mitochondrial function.
Additionally, PDE5 inhibitors have been shown to activate SIRT1 and PGC-1α signaling pathways, enhancing mitochondrial biogenesis and energy metabolism .
Tadalafil, with its long duration of action, may provide sustained activation of these protective pathways, making it particularly attractive for chronic conditions.
The implication is profound: PDE5 inhibitors may not only improve symptoms—they may modify disease processes.
Myocardial Ischaemia/Reperfusion Injury: A Paradox of Modern Medicine
Ischaemia/reperfusion (IR) injury is one of the most challenging paradoxes in cardiology. Restoring blood flow to ischaemic tissue is essential—yet it can also cause additional damage.
This injury results from multiple factors, including oxidative stress, calcium overload, and inflammatory responses. Clinically, it manifests as myocardial stunning, arrhythmias, and even irreversible cell death.
Experimental studies have shown that PDE5 inhibitors can significantly reduce infarct size and improve functional recovery when administered before or during reperfusion .
For example, sildenafil administered prior to ischaemia in animal models reduced myocardial injury and improved survival. Similar effects have been observed with postconditioning strategies, where the drug is given at the time of reperfusion.
These findings suggest that PDE5 inhibitors may act as pharmacological “preconditioning” agents, preparing the heart to withstand ischemic stress.
In a field where minutes determine outcomes, such protection could be transformative.
Experimental Evidence: Consistency Across Models
The cardioprotective effects of PDE5 inhibitors have been demonstrated across multiple experimental models and species.
Studies in rabbits, rats, mice, and dogs consistently show reduced infarct size, improved ventricular function, and decreased arrhythmias following PDE5 inhibition .
Notably, these effects are observed both in regional and global ischemia models, suggesting a broad applicability.
In transplantation models, preconditioning with PDE5 inhibitors improved graft function and reduced reperfusion injury. This has important implications for cardiac surgery and organ preservation.
Tadalafil, in particular, has shown protective effects in models of diabetic cardiomyopathy and chemotherapy-induced heart failure, highlighting its versatility.
The consistency of these findings strengthens the argument for clinical translation.
Heart Failure: A New Therapeutic Frontier
Heart failure (HF) remains one of the most challenging conditions in modern medicine. Despite advances in therapy, morbidity and mortality remain high.
PDE5 inhibitors offer a novel approach by targeting underlying molecular pathways rather than simply modifying hemodynamics.
In experimental models, chronic PDE5 inhibition has been shown to:
- Reduce cardiac hypertrophy
- Improve ventricular function
- Decrease fibrosis and apoptosis
- Enhance mitochondrial efficiency
Tadalafil, in particular, has demonstrated the ability to attenuate the progression of heart failure following myocardial infarction. It reduces necrosis, limits remodeling, and preserves cardiac function.
In diabetic models, it improves metabolic parameters and reduces inflammatory cytokines such as TNF-α and IL-1β.
These effects suggest that PDE5 inhibitors may act as disease-modifying agents, rather than merely symptomatic treatments.
Clinical Evidence: Promising but Incomplete
While preclinical data are compelling, clinical evidence remains limited.
Some studies have shown that sildenafil improves exercise capacity and quality of life in patients with heart failure and pulmonary hypertension . It also reduces pulmonary pressures and improves oxygen utilization.
However, not all trials have been positive. The RELAX trial, for example, found no significant benefit of sildenafil in patients with heart failure with preserved ejection fraction.
This discrepancy highlights the complexity of heart failure as a disease. Different subtypes may respond differently to PDE5 inhibition.
It also underscores the need for careful patient selection and further research.
Tadalafil: A Unique Candidate for Chronic Therapy
Among PDE5 inhibitors, tadalafil offers several advantages for cardiovascular applications.
Its long half-life allows for sustained activation of protective pathways. This may be particularly important in chronic conditions such as heart failure, where continuous modulation is required.
Additionally, tadalafil has demonstrated anti-inflammatory and antioxidant effects, reducing oxidative stress and improving mitochondrial function .
In models of doxorubicin-induced cardiomyopathy, tadalafil prevented cardiac damage without interfering with the drug’s anticancer effects—a critical consideration in oncology.
These properties position tadalafil as a promising candidate for long-term cardioprotective therapy.
Limitations and Challenges: The Gap Between Bench and Bedside
Despite promising data, several challenges remain.
First, most evidence comes from animal studies. Translating these findings to human patients is not straightforward.
Second, the heterogeneity of heart failure complicates clinical application. What works in one subtype may not work in another.
Third, potential side effects—such as hypotension—must be carefully managed, particularly in patients with advanced disease.
Finally, large-scale randomized clinical trials are needed to establish efficacy and safety in cardiovascular populations.
Until then, PDE5 inhibitors remain an intriguing—but not yet definitive—option.
Conclusion: A Return to Origins
The story of PDE5 inhibitors is coming full circle. From cardiovascular research to erectile dysfunction and back again, these drugs continue to reveal new possibilities.
The evidence suggests that targeting PDE5 may offer meaningful benefits in myocardial ischemia/reperfusion injury and heart failure. By modulating fundamental cellular pathways, these agents have the potential to protect the heart at its most vulnerable moments.
Tadalafil, with its unique pharmacological profile, stands at the forefront of this evolution.
Whether this promise will translate into routine clinical practice remains to be seen. But one thing is clear: sometimes, the future of medicine lies not in new discoveries, but in seeing old drugs with new eyes.
FAQ: Key Questions About PDE5 Inhibitors in Cardiology
1. Can PDE5 inhibitors really protect the heart?
Yes. Experimental studies show they reduce infarct size and improve cardiac function via the NO–cGMP–PKG pathway.
2. Is tadalafil used in heart failure treatment?
Not routinely yet, but research suggests it may improve cardiac function and reduce disease progression.
3. What is the main mechanism of cardioprotection?
Activation of cGMP signaling, leading to reduced apoptosis, improved mitochondrial function, and decreased inflammation.
4. Are there risks in cardiac patients?
Yes. Hypotension can occur, especially in unstable patients.
5. What is the future of this therapy?
Further clinical trials are needed, but PDE5 inhibitors represent a promising avenue for drug repurposing in cardiology.