Introduction: When a Familiar Drug Enters Unfamiliar Territory
Tadalafil is widely recognized as a cornerstone therapy for erectile dysfunction, a drug that has transformed quality of life for millions of men worldwide. Its pharmacological identity as a phosphodiesterase type 5 (PDE5) inhibitor places it firmly within the domain of vascular modulation. Yet, as is often the case in medicine, drugs rarely remain confined to their original purpose. They wander—sometimes subtly, sometimes dramatically—into other physiological systems.
One such unexpected territory is the central nervous system. The study explores a question that has long lingered in clinical pharmacology: does tadalafil influence seizure susceptibility, and if so, how does it interact with antiepileptic therapies? This is not merely an academic curiosity. Erectile dysfunction frequently coexists with neurological disorders, including epilepsy, creating a clinical overlap where such interactions become highly relevant.
This article reinterprets and expands upon those findings, offering a comprehensive and clinically oriented analysis. We will examine how tadalafil may alter neuronal excitability, influence seizure thresholds, and interact with antiepileptic drugs. Along the way, we will consider the broader implications for patient safety, therapeutic strategy, and future research.
Pharmacological Foundations: Understanding Tadalafil Beyond Erectile Function
Tadalafil operates by inhibiting PDE5, thereby increasing intracellular levels of cyclic guanosine monophosphate (cGMP). This biochemical shift enhances smooth muscle relaxation and promotes vasodilation. In the context of erectile tissue, the effect is predictable and beneficial. In the brain, however, the consequences are less straightforward.
Unlike some of its pharmacological relatives, tadalafil has a long half-life—approximately 17.5 hours—which allows for sustained systemic and potentially central nervous system exposure. This prolonged activity may be advantageous in urological therapy but raises concerns when considering neurological effects. The longer a drug lingers, the more opportunities it has to interact with complex neuronal pathways.
Crucially, tadalafil crosses the blood–brain barrier and influences the nitric oxide (NO)/cGMP signaling pathway within neural tissue . This pathway is not merely decorative; it plays a role in synaptic plasticity, neurotransmitter release, and neuronal excitability. In other words, tadalafil is not just a peripheral vasodilator—it is a subtle neuromodulator.
Seizure Thresholds and Neuroexcitability: What the Evidence Reveals
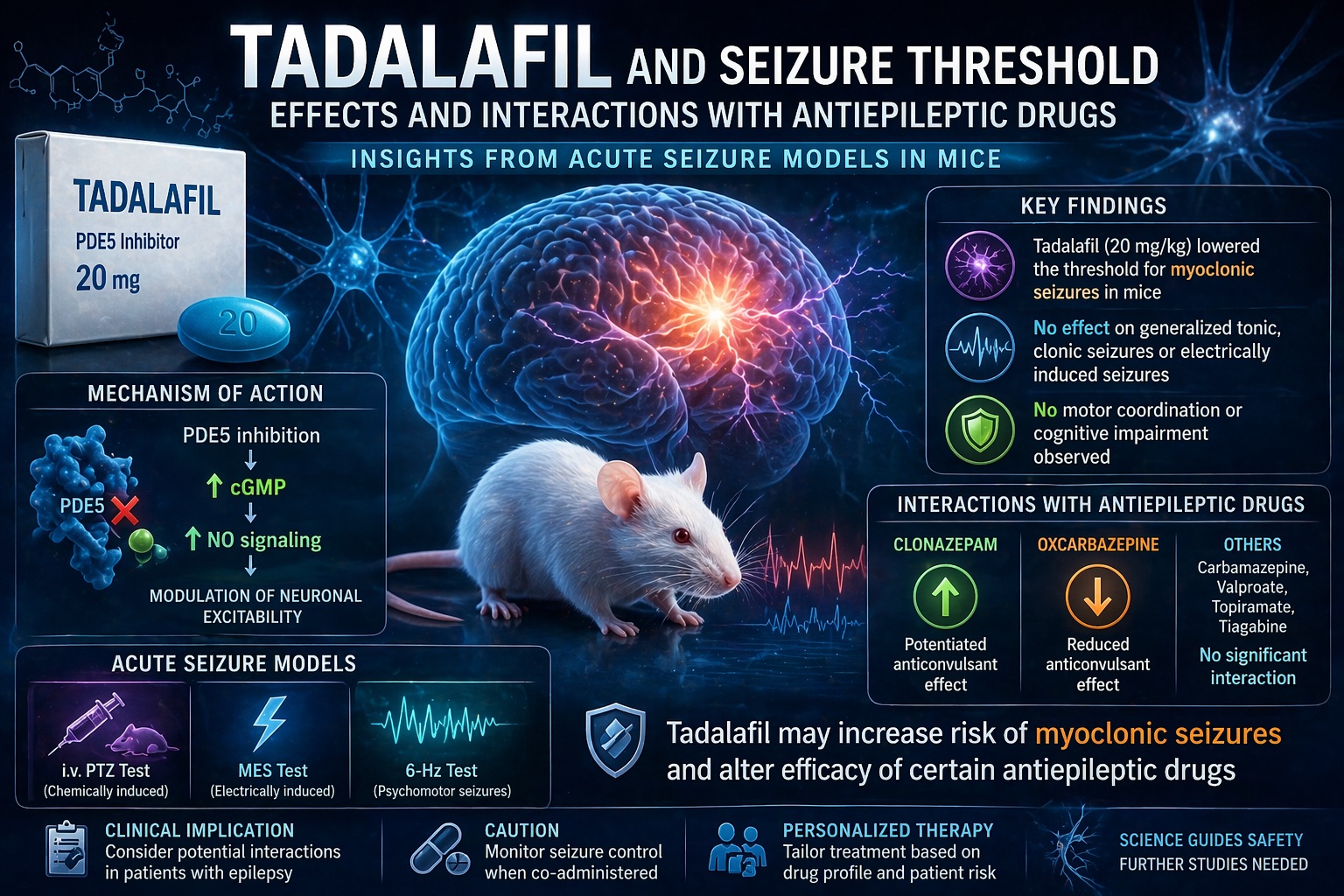
The central finding of the study is both intriguing and cautionary. At higher doses, tadalafil reduced the threshold for myoclonic seizures in mice. This means that less stimulation was required to trigger certain types of seizure activity. Importantly, this effect was selective—it did not significantly influence thresholds for generalized tonic or clonic seizures.
This selective vulnerability suggests that tadalafil’s impact is not uniform across all neural circuits. Myoclonic seizures are often associated with disruptions in inhibitory neurotransmission, particularly involving gamma-aminobutyric acid (GABA). The study proposes that increased cGMP levels may interfere with GABAergic signaling, thereby reducing inhibitory control and facilitating neuronal firing .
Interestingly, tadalafil did not alter seizure thresholds in electrically induced models, such as maximal electroshock or 6-Hz seizure tests. This discrepancy hints at a nuanced mechanism: tadalafil may influence chemically mediated seizure pathways more than those driven by electrical stimulation. In practical terms, the drug’s proconvulsant potential appears context-dependent rather than universal.
Mechanistic Insights: The NO–cGMP Axis and Neural Balance
To understand these findings, one must appreciate the delicate balance between excitation and inhibition in the brain. The NO–cGMP pathway plays a regulatory role in this balance, influencing both inhibitory GABAergic and excitatory glutamatergic systems.
Elevated cGMP levels can lead to phosphorylation of GABA_A receptor subunits, potentially reducing their inhibitory efficacy. At the same time, excessive cGMP may enhance glutamate release under certain conditions. The result is a shift toward excitation—a subtle but clinically significant tilt that may lower seizure thresholds.
This dual effect resembles a pharmacological tightrope. On one side lies therapeutic modulation; on the other, destabilization. Tadalafil, particularly at higher doses, appears capable of stepping closer to the latter.
It is worth noting that these mechanisms remain partially speculative. Direct evidence linking tadalafil to specific receptor-level changes is still evolving. However, the convergence of pharmacological theory and experimental observation provides a compelling narrative.
Interactions with Antiepileptic Drugs: A Complex Pharmacodynamic Landscape
Perhaps the most clinically relevant aspect of the study lies in its examination of drug interactions. Patients with epilepsy often require lifelong antiepileptic therapy, and any additional medication must be considered carefully.
The study demonstrates that tadalafil does not uniformly interfere with antiepileptic drugs. Instead, its effects vary depending on the specific agent:
- It enhances the anticonvulsant effect of clonazepam, a benzodiazepine that potentiates GABAergic transmission.
- It reduces the efficacy of oxcarbazepine, a sodium channel blocker.
- It shows no significant interaction with carbamazepine, topiramate, valproate, or tiagabine in certain seizure models.
These findings suggest that tadalafil’s interactions are pharmacodynamic rather than pharmacokinetic. In other words, it alters how drugs work, not how they are distributed or metabolized .
From a clinical perspective, this is both reassuring and concerning. Reassuring, because drug levels remain stable; concerning, because therapeutic effects may still change unpredictably.
Clinical Interpretation: What Does This Mean for Patients?
Translating animal data into human clinical practice is always a delicate exercise. Nevertheless, several practical insights emerge.
First, tadalafil appears unlikely to provoke generalized seizures in most patients. Its effects are subtle and selective, primarily affecting myoclonic thresholds at high doses. However, in individuals already predisposed to seizures—particularly myoclonic epilepsy—the risk may be more significant.
Second, drug interactions must be considered carefully. The potentiation of clonazepam may be beneficial in some contexts, but the reduction of oxcarbazepine efficacy could compromise seizure control. Clinicians should remain vigilant when prescribing tadalafil to patients on antiepileptic therapy.
Third, the absence of motor or cognitive impairment in the study is encouraging. Tadalafil did not adversely affect coordination, muscle strength, or memory, suggesting that its central effects are specific rather than broadly neurotoxic.
Comparative Perspective: Tadalafil Versus Other PDE5 Inhibitors
Not all PDE5 inhibitors are created equal. Sildenafil, for example, has demonstrated both proconvulsant and anticonvulsant effects in various models. Tadalafil, by contrast, appears more restrained in its neurological impact.
This difference likely reflects variations in molecular structure, selectivity, and pharmacokinetics. Tadalafil’s high selectivity for PDE5 and its longer half-life create a distinct pharmacological profile. It is, in a sense, a quieter drug—less prone to dramatic swings but still capable of subtle influence.
From a therapeutic standpoint, this may make tadalafil preferable in patients with neurological comorbidities. However, caution remains warranted, particularly at higher doses or in combination with certain medications.
Future Directions: Where Research Must Go Next
The study raises as many questions as it answers. Human data remain limited, and the translation of animal findings into clinical recommendations requires further investigation.
Future research should focus on:
- Controlled clinical trials in patients with epilepsy
- Detailed analysis of neurotransmitter dynamics under PDE5 inhibition
- Long-term safety studies in neurologically vulnerable populations
There is also an opportunity to explore whether PDE5 inhibitors might have therapeutic roles in certain neurological conditions. After all, modulation of the NO–cGMP pathway is not inherently harmful—it is simply powerful.
Conclusion: A Drug with More Stories to Tell
Tadalafil is no longer just a treatment for erectile dysfunction. It is a pharmacological agent with systemic reach, capable of influencing vascular, urological, and neurological systems alike.
The evidence suggests that while tadalafil is generally safe, it carries nuanced effects on seizure susceptibility and antiepileptic drug activity. These effects are subtle, selective, and highly context-dependent.
In medicine, such nuance is both a challenge and an opportunity. It reminds us that no drug exists in isolation—and that even familiar therapies may hold unexpected complexities beneath their surface.
FAQ: Key Questions Answered
1. Can tadalafil trigger seizures in humans?
There is limited evidence in humans, but animal studies suggest it may lower the threshold for certain types of seizures, particularly myoclonic ones. The risk appears low but not negligible.
2. Is tadalafil safe for patients with epilepsy?
It can be used cautiously, but careful monitoring is recommended, especially in patients with poorly controlled epilepsy or those on specific antiepileptic drugs.
3. Does tadalafil interact with antiepileptic medications?
Yes, it can enhance the effect of some drugs like clonazepam and reduce the efficacy of others like oxcarbazepine, primarily through pharmacodynamic mechanisms.
4. Are these effects dose-dependent?
Yes. The proconvulsant effects were observed mainly at higher doses in animal models.
5. Should clinicians avoid prescribing tadalafil in neurological patients?
Not necessarily, but they should evaluate risks individually and consider potential drug interactions before prescribing.