Introduction: When Pharmacology Meets the Black Market
Modern pharmacology prides itself on precision. Drugs like tadalafil are designed, tested, and regulated with meticulous care. Yet outside the controlled environment of clinical medicine, a parallel and far less disciplined world exists—one where molecules are modified, repurposed, and quietly slipped into products marketed as “natural.”
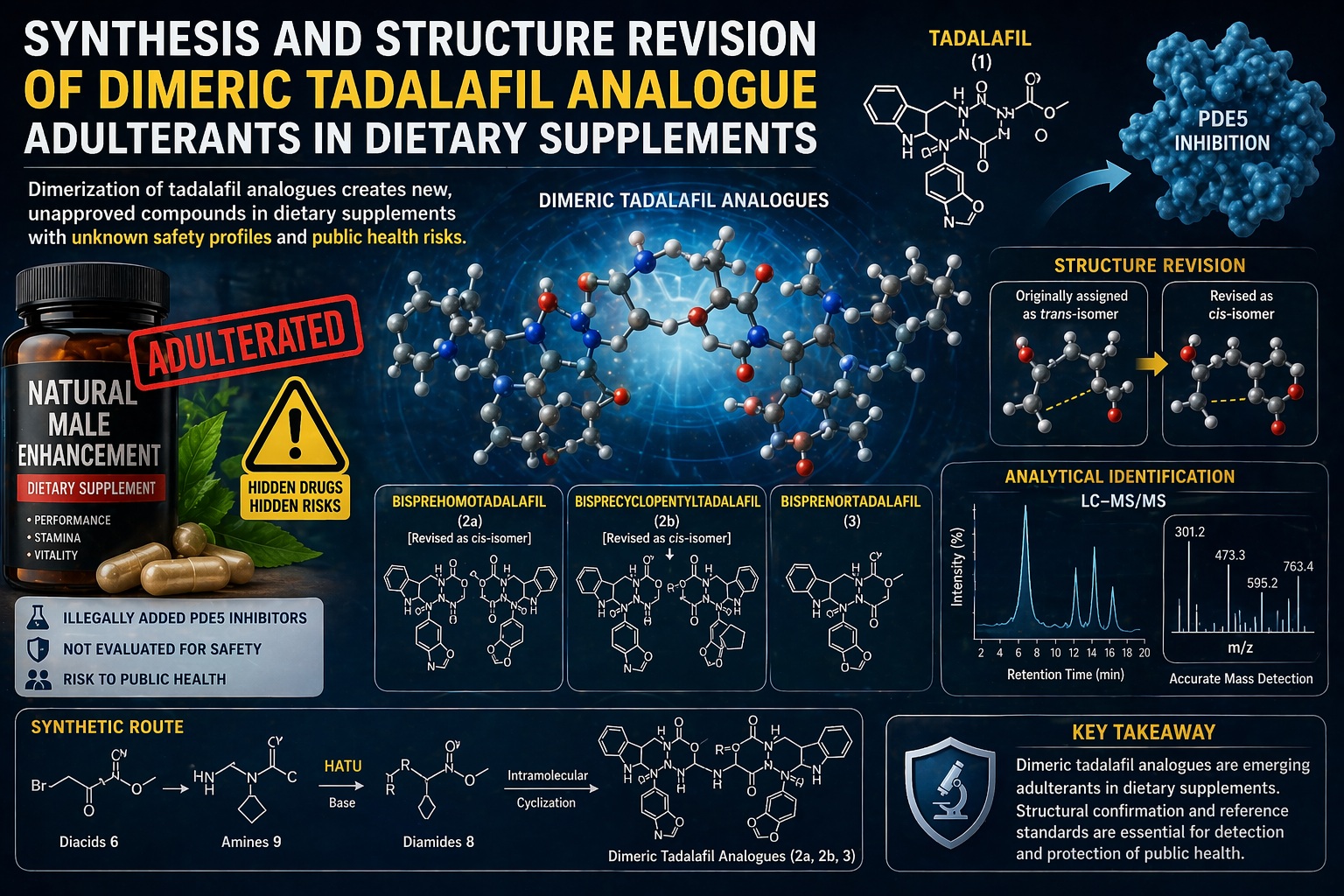
The study reveals a particularly concerning phenomenon: the emergence of dimeric tadalafil analogues deliberately synthesized or unintentionally generated and then detected in dietary supplements. These compounds are not simply minor variations of tadalafil. They are structurally complex derivatives, often designed to evade detection while preserving pharmacological activity.
This article expands upon those findings, offering a clinically grounded, chemically informed, and frankly sobering analysis of what these compounds mean for medicine, toxicology, and public health. Because when chemistry becomes clandestine, patients become unwitting participants in an uncontrolled experiment.
Tadalafil and Its Molecular Legacy: A Double-Edged Success Story
Tadalafil, a selective phosphodiesterase type 5 (PDE5) inhibitor, has earned its reputation through efficacy and relative safety. Its mechanism—enhancing nitric oxide–mediated vasodilation via cGMP accumulation—has made it a mainstay in the treatment of erectile dysfunction and pulmonary hypertension.
Yet success invites imitation. Over the past decade, tadalafil has inspired a growing number of structural analogues, many of which are not approved, not tested, and certainly not declared on product labels. These compounds are often engineered by modifying functional groups, altering stereochemistry, or truncating molecular fragments. The goal is simple: maintain biological activity while evading regulatory detection.
The study highlights a more sophisticated evolution—dimerization. Instead of modifying a single tadalafil molecule, chemists have linked two “pre-tadalafil” units into a single compound. The result is not merely a derivative but a fundamentally new chemical entity with unpredictable pharmacological behavior .
Dimeric Analogues: Structural Innovation with Uncertain Consequences
The compounds identified—such as bisprehomotadalafil, bisprecyclopentyltadalafil, and bisprenortadalafil—represent a new class of adulterants. Structurally, they consist of two tadalafil-like moieties connected through amide linkages, forming large, sterically complex molecules.
Initially, these compounds were mischaracterized as trans-isomers. However, through careful synthesis and spectroscopic analysis, researchers demonstrated that they are in fact cis-isomers, a seemingly subtle distinction with significant implications. Stereochemistry, after all, is not a cosmetic detail—it governs how molecules interact with biological targets.
The corrected structural assignment was achieved through advanced nuclear magnetic resonance (NMR) techniques and comparison with synthesized reference standards . This revision underscores an important point: even experts can misinterpret complex molecules, and such errors can propagate into regulatory blind spots.
From a clinical perspective, the existence of these dimers raises immediate concerns. Their size, polarity, and conformational flexibility differ markedly from tadalafil, suggesting altered absorption, distribution, metabolism, and excretion. In simpler terms, they behave differently—and unpredictably.
Chemical Synthesis: How These Compounds Are Made—and Why It Matters
The study provides a detailed synthetic pathway, revealing how these dimeric analogues can arise. The process involves amide coupling reactions between diacid intermediates and aminoester derivatives, often facilitated by coupling reagents such as HATU, which proved particularly effective .
Interestingly, the researchers propose that these dimers may not always be intentionally synthesized. Instead, they can form as by-products during the production of monomeric tadalafil analogues. Poor purification practices may then allow these compounds to persist in the final product.
This distinction—intentional versus accidental adulteration—may seem academic, but it is not. Intentional synthesis suggests deliberate fraud; by-product contamination suggests negligence. From the patient’s perspective, however, the outcome is identical: exposure to an untested pharmacological agent.
The synthetic challenges described in the study also highlight the role of steric hindrance and reagent selection. Traditional coupling methods failed, while HATU enabled efficient formation of the desired dimers. This technical detail, while chemical in nature, has broader implications—it demonstrates that these compounds are not trivial to produce, yet they are still appearing in consumer products.
Pharmacological Implications: Beyond the Known Effects of Tadalafil
Tadalafil’s safety profile is well established, with known contraindications such as concurrent nitrate therapy due to the risk of severe hypotension. However, these safety parameters apply to a specific molecule—not to its unregulated analogues.
Dimeric tadalafil analogues introduce several layers of uncertainty:
- Their binding affinity for PDE5 may differ, potentially enhancing or reducing potency.
- Their selectivity for PDE5 versus other phosphodiesterases is unknown.
- Their metabolic pathways may generate active or toxic intermediates.
Moreover, their larger molecular size may affect tissue distribution, potentially leading to prolonged or unexpected effects. One might imagine a drug designed for precision behaving instead like a blunt instrument.
There is also the question of dose equivalence. If a supplement contains an undeclared dimeric analogue, what is the effective dose? How does it compare to tadalafil? The answer, at present, is: we do not know.
Public Health Perspective: The Silent Epidemic of Adulterated Supplements
The detection of these compounds in dietary supplements is not an isolated incident. It reflects a broader trend in which “natural” products are adulterated with pharmaceutical agents to enhance efficacy.
These products are often marketed for sexual enhancement, a category where consumer demand intersects with stigma and discretion. The result is a market ripe for exploitation.
The risks are compounded by several factors:
- Lack of labeling prevents informed decision-making.
- Absence of quality control allows variable dosing.
- Unknown pharmacology increases the risk of adverse events.
In effect, these supplements bypass every safeguard built into modern medicine. They are sold without prescriptions, consumed without supervision, and metabolized without understanding.
Analytical Challenges: Detecting the Undetectable
One of the most important contributions of the study is the creation of reference standards for these dimeric analogues. Without such standards, detection becomes nearly impossible.
Analytical techniques such as high-performance liquid chromatography (HPLC) and mass spectrometry rely on known signatures. When a compound is unknown, it can slip through even sophisticated screening systems.
By synthesizing these analogues and confirming their structures, researchers have provided the tools needed to identify them in commercial products . This is not merely an academic achievement—it is a critical step in protecting public health.
However, the challenge remains dynamic. As soon as one analogue is identified, others may emerge. It is, in many ways, a chemical arms race.
Clinical Guidance: What Should Physicians and Patients Do?
For clinicians, the implications are clear. Patients presenting with unexplained hypotension, drug interactions, or adverse reactions should be questioned about supplement use. The assumption that “natural” equals “safe” must be actively challenged.
For patients, the advice is equally straightforward, if less palatable. Avoid unregulated supplements, particularly those marketed for sexual enhancement. The promise of convenience or discretion is not worth the risk of unknown pharmacology.
There is also a role for education. Patients must understand that tadalafil, when prescribed, is safe because it is controlled—not because it is inherently benign. Remove the control, and the safety profile dissolves.
Conclusion: Chemistry Without Oversight Is Medicine Without Ethics
The story of dimeric tadalafil analogues is not just about chemistry. It is about the consequences of removing science from its ethical framework.
These compounds are sophisticated, cleverly designed, and scientifically interesting. They are also untested, unregulated, and potentially dangerous. Their presence in dietary supplements represents a failure—not of chemistry, but of oversight.
Tadalafil itself remains a valuable and effective medication. But its molecular descendants, when produced outside the bounds of regulation, become something else entirely.
In the end, the lesson is simple: a drug is only as safe as the system that governs it.
FAQ: Key Questions Answered
1. What are dimeric tadalafil analogues?
They are complex molecules formed by linking two tadalafil-like structures, often detected in adulterated dietary supplements.
2. Are these compounds safe?
Their safety is unknown. They have not undergone clinical testing, and their pharmacological effects may differ significantly from tadalafil.
3. Why are they added to supplements?
To enhance efficacy while avoiding detection by regulatory authorities.
4. How can they be detected?
Through advanced analytical methods using synthesized reference standards, as described in the study.
5. Should patients avoid supplements marketed for sexual enhancement?
Yes, especially if they are unregulated or lack transparent ingredient labeling.