Introduction: When Vasodilation Becomes a Lifeline
Pulmonary arterial hypertension (PAH) is not merely a disease of elevated pressure—it is a progressive, life-limiting disorder that silently transforms the pulmonary circulation and ultimately overwhelms the right ventricle. For decades, therapeutic progress has been incremental, often limited by complexity of administration and tolerability issues.
Into this landscape enters an unexpected candidate: tadalafil.
Originally developed for erectile dysfunction, tadalafil has steadily expanded its clinical identity. The article explores its emerging role in PAH, revealing a compelling intersection of pharmacology and pathophysiology. What appears at first to be a simple vasodilator reveals itself as a strategic modulator of pulmonary vascular tone and potentially disease progression.
This is not a story of coincidence. It is a story of mechanism—and how understanding biology can transform a drug’s destiny.
Pulmonary Arterial Hypertension: A Disease of Structure and Pressure
PAH is defined by elevated pulmonary artery pressure in the absence of left-sided heart disease. Clinically, it is characterized by progressive dyspnea, reduced exercise tolerance, and eventual right heart failure.
The disease is driven by structural remodeling of the pulmonary vasculature. This includes intimal thickening, medial hypertrophy, adventitial fibrosis, and in situ thrombosis. Over time, these changes narrow the vascular lumen and increase pulmonary vascular resistance .
The right ventricle, designed for low-pressure circulation, is forced to adapt. Initially, it compensates through hypertrophy. Eventually, however, it fails—leading to edema, ascites, and syncope.
The prognosis remains poor. Historical data suggest a median survival of less than three years without effective treatment.
In this context, even modest therapeutic improvements can have profound implications.
Nitric Oxide and cGMP: The Molecular Foundation of Therapy
At the core of PAH pathophysiology lies a dysregulation of endothelial signaling.
Nitric oxide (NO), a key vasodilator, is reduced in PAH. This deficiency leads to decreased production of cyclic guanosine monophosphate (cGMP), a second messenger responsible for smooth muscle relaxation.
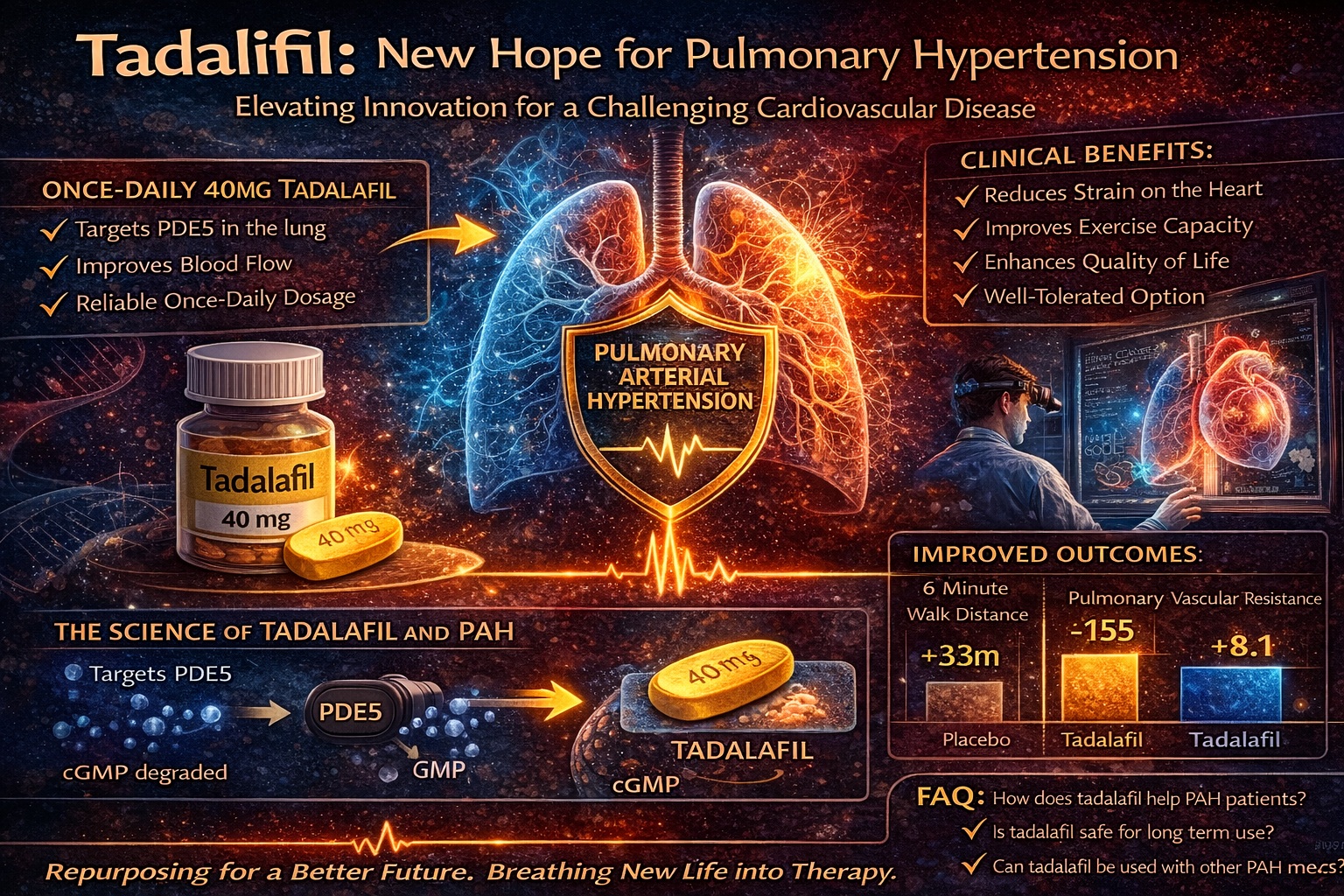
Phosphodiesterase type 5 (PDE5) plays a critical role in this system. It degrades cGMP, effectively limiting the vasodilatory effect of NO.
Tadalafil inhibits PDE5, preventing the breakdown of cGMP and enhancing its activity. The result is sustained vasodilation within the pulmonary circulation .
Importantly, PDE5 is highly expressed in lung tissue, making it an ideal therapeutic target. This tissue specificity allows for relatively selective pulmonary vasodilation without excessive systemic hypotension.
In essence, tadalafil restores a pathway that PAH has disrupted.
Pharmacology of Tadalafil: Designed for Continuity
Among PDE5 inhibitors, tadalafil possesses a distinctive pharmacokinetic profile.
Its elimination half-life of approximately 17.5 hours allows for prolonged activity and supports once-daily dosing . This contrasts with agents such as sildenafil, which require multiple daily doses.
From a clinical perspective, this difference is more than convenient—it is strategic.
Adherence is a critical issue in chronic diseases. Medications requiring frequent dosing are associated with higher rates of nonadherence. By simplifying the regimen, tadalafil addresses this challenge directly.
Additionally, its slower onset and sustained effect provide a more stable hemodynamic profile, avoiding the peaks and troughs associated with shorter-acting agents.
In PAH, where consistency matters, this stability may be particularly advantageous.
Clinical Evidence: Early Signals of Efficacy
While large-scale randomized trials are still evolving, early clinical evidence for tadalafil in PAH is encouraging.
Small pilot studies and case series have demonstrated:
- Reduction in pulmonary vascular resistance
- Improvement in functional class
- Increased exercise capacity (6-minute walk distance)
In one observational study, patients treated with tadalafil showed significant improvement in WHO functional class and walking distance after 12 weeks of therapy. These changes, while modest numerically, translate into meaningful clinical benefits.
Another case series reported that patients transitioning from sildenafil to tadalafil maintained their clinical improvements, suggesting comparable efficacy.
These findings align with the known effects of PDE5 inhibition and reinforce the biological plausibility of tadalafil’s role in PAH.
Comparative Insights: Tadalafil Versus Other PDE5 Inhibitors
Sildenafil remains the most extensively studied PDE5 inhibitor in PAH and is currently approved for this indication. Its efficacy is well established, with demonstrated improvements in exercise capacity and pulmonary hemodynamics.
Tadalafil shares the same fundamental mechanism but differs in pharmacodynamic nuances.
For example, studies comparing PDE5 inhibitors in pulmonary artery preparations suggest that tadalafil may have unique effects on hypoxia-induced vasoconstriction and inflammatory signaling .
In clinical settings, all three major PDE5 inhibitors—sildenafil, vardenafil, and tadalafil—reduce pulmonary vascular resistance. However, tadalafil’s longer duration of action may offer advantages in chronic management.
It is not necessarily superior—but it is different in ways that matter.
Integration into Current Treatment Strategies
Modern PAH management is multifaceted. It includes anticoagulation, diuretics, oxygen therapy, and targeted pharmacological agents such as prostacyclins and endothelin receptor antagonists.
PDE5 inhibitors fit into this framework as oral vasodilators that are relatively easy to administer.
Tadalafil’s role may be particularly valuable in combination therapy. Preliminary evidence suggests that combining agents with different mechanisms can produce additive benefits.
For patients who struggle with complex regimens or invasive therapies, tadalafil offers a simpler alternative.
Its potential lies not only in efficacy, but in accessibility.
Patient-Centered Outcomes: Beyond Hemodynamics
Clinical success in PAH is not defined solely by pressure measurements.
Patients care about how they feel—how far they can walk, how easily they can breathe, and whether they can participate in daily life.
Tadalafil has shown promise in improving functional capacity and quality of life. Patients report increased exercise tolerance and reduced symptoms, reflecting real-world benefit .
These improvements may seem modest in clinical terms, but they are significant for individuals living with a chronic, progressive disease.
In PAH, small gains often translate into meaningful changes in daily life.
Limitations of Current Evidence
Despite promising findings, the evidence base for tadalafil in PAH remains limited.
Most studies are small, non-randomized, or observational. Large-scale randomized controlled trials are needed to establish definitive efficacy and safety.
The ongoing PHIRST study represents an important step in this direction, aiming to provide robust data on clinical outcomes.
Additionally, PAH is a heterogeneous disease. Different etiologies may respond differently to therapy, complicating interpretation of results.
These limitations do not diminish the potential of tadalafil—but they underscore the need for continued research.
Future Perspectives: Expanding the Role of PDE5 Inhibition
The potential applications of tadalafil extend beyond PAH.
Preliminary studies suggest benefits in heart failure, endothelial dysfunction, and even conditions such as Raynaud’s phenomenon .
This raises an intriguing possibility: PDE5 inhibition as a broader cardiovascular strategy.
If confirmed, tadalafil could become part of a new generation of therapies that target fundamental signaling pathways rather than isolated symptoms.
In this sense, PAH may be just the beginning.
Conclusion: A Drug Reimagined
Tadalafil’s journey from erectile dysfunction to pulmonary hypertension illustrates the power of translational medicine.
By targeting a key molecular pathway, it offers a rational and potentially effective approach to a complex disease. Its pharmacokinetic advantages and emerging clinical evidence make it a strong candidate for inclusion in PAH treatment strategies.
While further research is needed, the trajectory is clear: tadalafil is no longer confined to its original indication.
It is becoming a versatile therapeutic tool—one that bridges disciplines and redefines expectations.
FAQ: Tadalafil in Pulmonary Arterial Hypertension
1. How does tadalafil help in PAH?
It enhances nitric oxide signaling by inhibiting PDE5, leading to pulmonary vasodilation.
2. Is tadalafil as effective as sildenafil?
Early evidence suggests comparable efficacy, with the advantage of once-daily dosing.
3. Can tadalafil improve exercise capacity?
Yes. Studies show improved 6-minute walk distance and functional class.
4. Is tadalafil safe in PAH patients?
It is generally well tolerated, with a safety profile similar to other PDE5 inhibitors.
5. What makes tadalafil unique?
Its long half-life allows for sustained effect and simplified dosing, improving adherence.