Introduction: The Limits of Success in Modern PAH Therapy
Pulmonary arterial hypertension (PAH) is no longer the uniformly fatal diagnosis it once was. Advances in targeted therapies have transformed the disease into a chronic, manageable condition for many patients. Among these therapies, phosphodiesterase type-5 (PDE5) inhibitors—such as tadalafil—have become a cornerstone of treatment, offering improved pulmonary hemodynamics and functional capacity.
Yet, despite these achievements, a persistent clinical reality remains: not all patients respond adequately. A substantial proportion of individuals treated with PDE5 inhibitors continue to experience symptoms, reduced exercise capacity, and progressive disease.
This gap between therapeutic potential and clinical reality has prompted a critical question in modern PAH management: What should be done when first-line therapy is insufficient?
The RESPITE study was designed to address precisely this dilemma. Rather than simply adding more medications or increasing doses, it explored a more nuanced strategy—switching from PDE5 inhibition to soluble guanylate cyclase (sGC) stimulation with riociguat.
This approach reflects a deeper understanding of the nitric oxide pathway and its dysfunction in PAH. It also challenges a common assumption in pharmacotherapy: that intensifying the same mechanism will always yield better results.
In this article, we explore the rationale, design, and implications of this strategy. Along the way, we will examine why some patients fail to respond to PDE5 inhibitors and how alternative pathways may offer a more effective route to treatment success.
The Nitric Oxide Pathway in PAH: A System Under Strain
The nitric oxide (NO)–cyclic guanosine monophosphate (cGMP) pathway plays a central role in regulating pulmonary vascular tone. Under normal conditions, endothelial cells produce nitric oxide, which diffuses into smooth muscle cells and activates soluble guanylate cyclase. This enzyme converts guanosine triphosphate (GTP) into cGMP, leading to vasodilation.
In pulmonary arterial hypertension, this pathway is profoundly disrupted. Endothelial dysfunction reduces nitric oxide production, while oxidative stress further impairs its bioavailability. The result is a state of relative NO deficiency.
PDE5 inhibitors such as tadalafil attempt to compensate for this deficiency by preventing the breakdown of cGMP. By preserving existing cGMP levels, they enhance vasodilation and improve blood flow within the pulmonary circulation.
However, this strategy assumes that sufficient cGMP is being produced in the first place. In patients with severe endothelial dysfunction, nitric oxide levels may be so low that PDE5 inhibition has limited substrate to act upon.
In other words, one cannot preserve what is not there.
This limitation helps explain why some patients exhibit an inadequate response to PDE5 inhibitors. Their underlying pathophysiology may simply render the mechanism less effective.
Clinical Reality: Inadequate Response to PDE5 Inhibitors
In clinical practice, inadequate response to PDE5 inhibitors is not uncommon. Patients may continue to experience symptoms such as dyspnea, fatigue, and reduced exercise tolerance despite ongoing therapy.
Functional assessments often reveal suboptimal improvement. Six-minute walk distance may remain limited, and biomarkers such as NT-proBNP may indicate persistent right ventricular strain.
Importantly, these patients are not necessarily treatment failures—they are partial responders. Their condition may improve to some extent, but not sufficiently to meet therapeutic goals.
Several factors contribute to this variability in response. These include disease severity, duration of illness, comorbid conditions, and individual differences in pharmacokinetics and pharmacodynamics.
From a mechanistic standpoint, insufficient nitric oxide availability is a key contributor. Without adequate NO signaling, the cGMP pathway cannot be effectively amplified by PDE5 inhibition alone.
This realization has led to the exploration of alternative strategies that bypass the need for endogenous nitric oxide.
Riociguat: A Different Approach to the Same Pathway
Riociguat represents a novel class of medication known as soluble guanylate cyclase stimulators. Unlike PDE5 inhibitors, which preserve cGMP, riociguat directly stimulates its production.
The drug acts through two complementary mechanisms. First, it sensitizes sGC to endogenous nitric oxide, enhancing its activity. Second, it directly stimulates sGC independently of nitric oxide.
This dual action allows riociguat to generate cGMP even in conditions of low NO availability. For patients with severe endothelial dysfunction, this represents a significant advantage.
By increasing cGMP production rather than merely preserving it, riociguat addresses a fundamental limitation of PDE5 inhibitors.
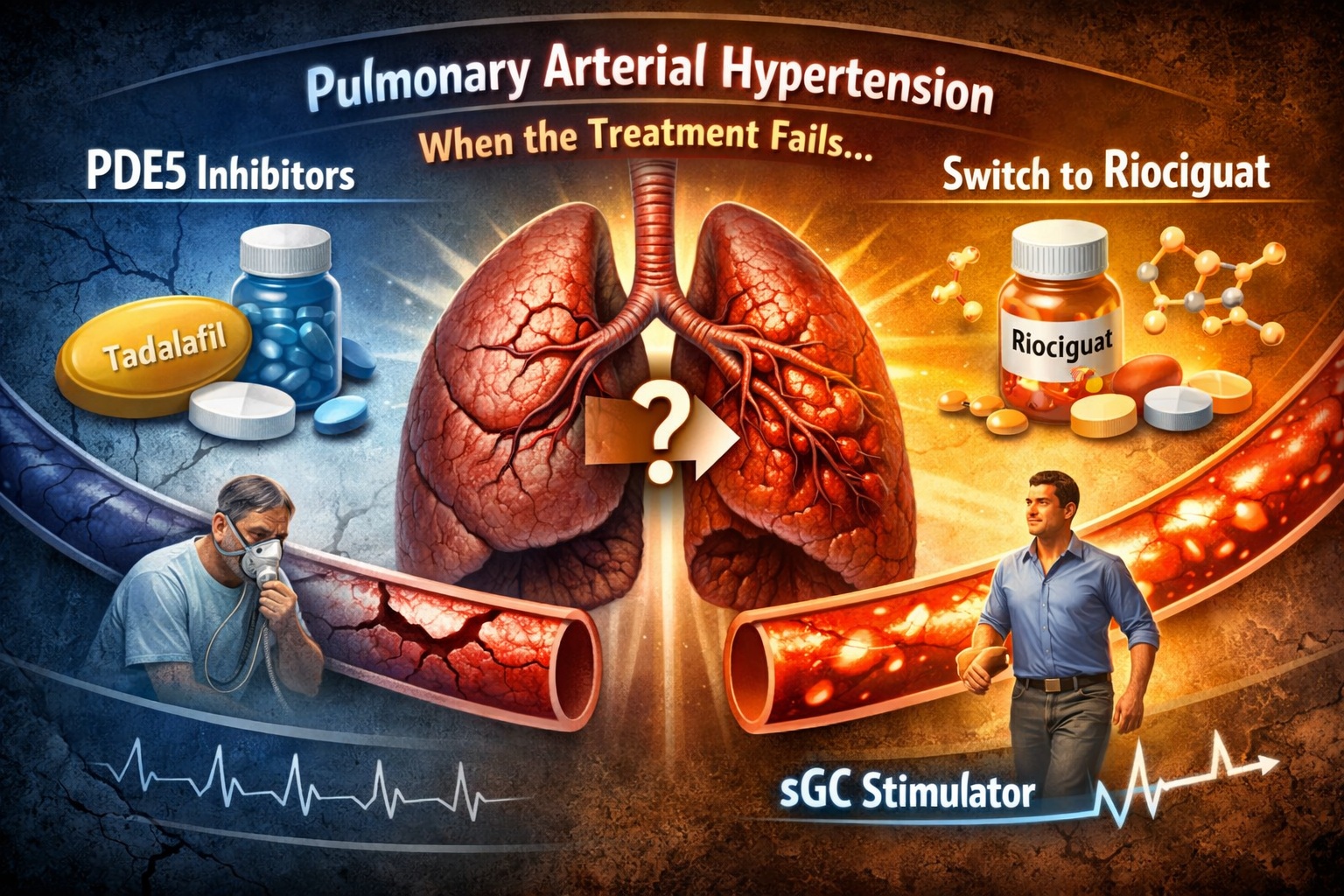
However, this pharmacological overlap also introduces a challenge: PDE5 inhibitors and riociguat cannot be used together safely due to the risk of excessive hypotension.
This constraint necessitates a strategic decision—whether to continue PDE5 inhibition or switch to sGC stimulation.
The RESPITE Study: Rationale and Design
The RESPITE study was conceived to evaluate the feasibility and clinical impact of switching patients from PDE5 inhibitors to riociguat.
The study focused on patients with pulmonary arterial hypertension who demonstrated an insufficient response to PDE5 inhibitor therapy. These individuals represented a clinically relevant population—patients who were not failing therapy outright, but who were not achieving optimal outcomes.
RESPITE was designed as an open-label, phase 3b study, reflecting its exploratory nature. Rather than comparing two treatments in a randomized fashion, the study examined the effects of a deliberate therapeutic switch.
Patients enrolled in the study underwent a washout period to discontinue PDE5 inhibitors such as tadalafil before initiating riociguat. This step was essential to avoid pharmacodynamic interactions.
The study then evaluated multiple clinical endpoints, including:
- Exercise capacity (six-minute walk distance)
- Functional class
- Biomarkers of cardiac stress
- Hemodynamic parameters
By assessing these outcomes, researchers aimed to determine whether switching therapy could provide meaningful clinical benefit.
Clinical Outcomes: What Happens After the Switch?
Results from the RESPITE study suggested that switching to riociguat may improve clinical outcomes in selected patients.
Many participants demonstrated increased exercise capacity, reflected in improvements in six-minute walk distance. This change is particularly meaningful in PAH, where exercise tolerance closely correlates with quality of life.
Functional class also improved in a proportion of patients, indicating reduced symptom burden. These improvements suggest that riociguat may provide additional benefit beyond what was achieved with PDE5 inhibitors.
Biomarker analysis revealed reductions in NT-proBNP levels, reflecting decreased right ventricular strain. This finding supports the idea that improved pulmonary hemodynamics translate into reduced cardiac stress.
Importantly, the safety profile of the switch was acceptable, with manageable adverse effects. However, careful monitoring remained essential, particularly during the transition period.
These results highlight a key principle: when one mechanism is insufficient, targeting the same pathway through a different mechanism may yield better outcomes.
Practical Considerations: Switching Therapy in Clinical Practice
The decision to switch from a PDE5 inhibitor such as tadalafil to riociguat should not be taken lightly. It requires careful patient selection, thorough evaluation, and close monitoring.
Patients considered for switching typically exhibit persistent symptoms despite optimized therapy. They may have stable disease but fail to achieve treatment goals.
The transition process involves a washout period to eliminate the PDE5 inhibitor before initiating riociguat. This step is critical to prevent hypotension and other adverse effects.
Clinicians must also consider individual patient factors, including blood pressure, comorbid conditions, and tolerance of previous therapies.
In practice, switching therapy represents a strategic adjustment rather than an escalation. It reflects a shift in mechanism rather than an increase in pharmacological intensity.
Rethinking Treatment Strategy: Precision Over Persistence
The RESPITE study underscores an important lesson in modern medicine: persistence with a single therapeutic mechanism is not always the optimal strategy.
In pulmonary arterial hypertension, where multiple pathways contribute to disease progression, treatment must be adaptable. When one approach reaches its limits, alternative mechanisms should be considered.
This concept aligns with the broader trend toward precision medicine, where therapy is tailored to the individual patient’s pathophysiology.
For some patients, PDE5 inhibitors like tadalafil remain highly effective. For others, particularly those with advanced endothelial dysfunction, alternative approaches such as sGC stimulation may provide greater benefit.
The key lies in recognizing when to change course.
Conclusion: A Strategic Shift in PAH Management
The RESPITE study offers a compelling example of how thoughtful therapeutic adjustment can improve patient outcomes.
By shifting from PDE5 inhibition to sGC stimulation, clinicians can address limitations in the nitric oxide pathway and provide a more effective treatment for selected patients.
This approach does not diminish the value of tadalafil or other PDE5 inhibitors. Rather, it highlights the importance of using these tools within a broader, flexible treatment strategy.
In pulmonary arterial hypertension, success is rarely achieved through a single intervention. It requires a dynamic, patient-centered approach that adapts to the evolving nature of the disease.
And sometimes, the most effective step forward is not to push harder in the same direction—but to take a different path altogether.
FAQ
Why might tadalafil not work for some PAH patients?
Tadalafil relies on existing nitric oxide signaling to enhance cGMP levels. In patients with severe endothelial dysfunction, nitric oxide production may be too low for the drug to be fully effective.
What is riociguat and how is it different?
Riociguat is a soluble guanylate cyclase stimulator that directly increases cGMP production, even in the absence of nitric oxide. This makes it effective in patients who do not respond well to PDE5 inhibitors.
Can tadalafil and riociguat be used together?
No. Combining these drugs can cause excessive vasodilation and dangerous hypotension. Patients must discontinue PDE5 inhibitors before starting riociguat.