Introduction: A Familiar Drug Class Enters Unfamiliar Territory
Modern neurology faces a paradox. Despite decades of research, Alzheimer’s disease and related dementias (ADRD) remain only partially understood and poorly treated. The therapeutic arsenal is limited, often focused on symptomatic relief rather than meaningful disease modification. Against this backdrop, the concept of drug repurposing has gained renewed attention—sometimes out of necessity, sometimes out of ingenuity.
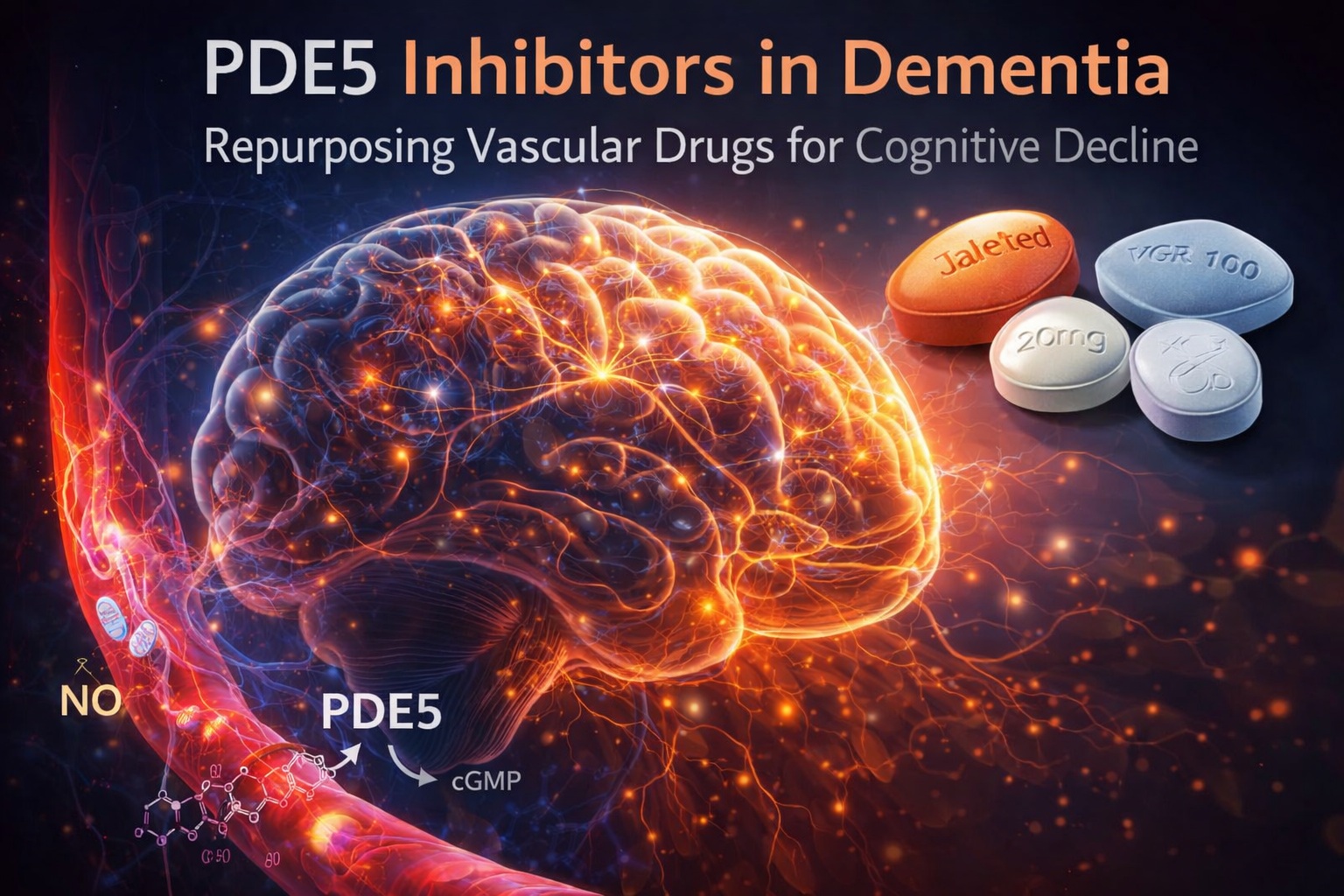
Phosphodiesterase-5 (PDE5) inhibitors—best known for their role in erectile dysfunction and pulmonary hypertension—have recently emerged as unexpected candidates in dementia research. Drugs such as sildenafil, vardenafil, and tadalafil, long associated with vascular pharmacology, are now being examined for their potential effects on brain function and cognition .
This shift may seem counterintuitive at first glance. Yet, when one considers the intricate relationship between cerebral blood flow, synaptic plasticity, and neurodegeneration, the rationale becomes less surprising. The brain, after all, is both an electrical and a vascular organ. And PDE5 inhibitors, it turns out, influence both domains.
Mechanistic Foundations: Beyond Vasodilation
At the heart of PDE5 inhibitor pharmacology lies the nitric oxide (NO)–cGMP signaling pathway. By inhibiting PDE5, these drugs prevent the breakdown of cyclic guanosine monophosphate (cGMP), thereby enhancing vasodilation. This mechanism is well established in peripheral tissues—but its implications in the brain are only now being fully appreciated.
PDE5 is expressed not only in vascular smooth muscle but also in neurons and glial cells. Within the central nervous system, cGMP signaling plays a crucial role in synaptic plasticity, particularly in processes such as long-term potentiation (LTP), which underpins memory formation . By preserving cGMP levels, PDE5 inhibitors may enhance these synaptic processes.
This dual mechanism—vascular and neuronal—creates a compelling therapeutic hypothesis. On one hand, improved cerebral perfusion may support metabolic demands of brain tissue. On the other, direct neuronal effects may strengthen synaptic connectivity. Whether one mechanism predominates remains an open question, and perhaps a false dichotomy; the most likely answer is that both contribute.
Tadalafil deserves particular mention in this context. Its longer half-life and demonstrated ability to cross the blood–brain barrier, albeit modestly, make it an attractive candidate for chronic neurological applications. Unlike shorter-acting agents, tadalafil offers sustained pharmacological activity, a feature that may be critical in diseases characterized by slow progression.
Preclinical Evidence: When Mice Remember Better Than Humans
Animal studies provide some of the most persuasive evidence supporting the cognitive potential of PDE5 inhibitors. Across multiple experimental models, these drugs have demonstrated consistent effects on learning, memory, and synaptic integrity.
In rodent models of Alzheimer’s disease, sildenafil has been shown to improve memory performance, reduce amyloid burden, and enhance neurogenesis. The experimental data illustrated on page 6 of the source document show enhanced synaptic strengthening and improved behavioral outcomes following PDE5 inhibition, reinforcing the biological plausibility of these findings .
Interestingly, these benefits are not limited to pathological models. Even in healthy animals, PDE5 inhibitors can enhance memory retention, suggesting a fundamental role in cognitive processing rather than merely disease correction. In primate studies, sildenafil improved executive function—a finding that brings the research one step closer to human relevance.
Yet, as is often the case in neuroscience, success in animal models does not guarantee clinical efficacy. The human brain is not merely a larger rodent brain; it is a far more complex and less forgiving system. Still, these findings provide a strong foundation for further investigation.
Human Data: Signals, Noise, and Interpretation
Human studies of PDE5 inhibitors in dementia present a more nuanced picture. On one hand, real-world data offer tantalizing hints of benefit. Large-scale analyses of insurance databases have shown that sildenafil use is associated with a significantly reduced risk of Alzheimer’s disease—by as much as 30% to 60% in some cohorts .
Such findings are difficult to ignore. They suggest that long-term exposure to PDE5 inhibitors may confer protective effects, potentially through mechanisms that are not fully captured in short-term clinical trials. However, these studies are observational and cannot establish causality. Confounding factors—such as socioeconomic status, baseline health, and access to care—may influence the results.
Controlled clinical studies, by contrast, have been less definitive. Trials examining acute administration of PDE5 inhibitors, including tadalafil, have generally failed to demonstrate significant changes in cerebral blood flow or cognitive performance. The PASTIS trial, for example, found no measurable difference between tadalafil and placebo in cerebral perfusion, despite a modest reduction in blood pressure .
The discrepancy between observational and experimental data raises important questions. Are the benefits of PDE5 inhibitors cumulative rather than immediate? Are current trial designs inadequate to capture subtle cognitive effects? Or are we simply observing statistical artifacts in large datasets? The answer likely lies somewhere in between.
Cerebral Blood Flow: A Necessary but Insufficient Explanation
One of the earliest hypotheses regarding PDE5 inhibitors in dementia focused on cerebral blood flow. Given their vasodilatory effects, it seemed reasonable to assume that these drugs might enhance perfusion in regions affected by small vessel disease.
However, the evidence is inconsistent. As shown in the graph on page 5, changes in cerebral blood flow after tadalafil administration were modest and variable, with no clear overall effect . Some subgroups, particularly older individuals, showed trends toward improvement, but these findings did not reach statistical significance.
This suggests that vascular effects alone cannot fully explain the potential cognitive benefits of PDE5 inhibitors. The brain is not simply a hydraulic system; it is a network of highly specialized cells whose function depends on precise biochemical signaling.
It is therefore increasingly likely that any therapeutic effect of PDE5 inhibitors in dementia involves a combination of vascular support and direct neuronal modulation. This dual action may be subtle, requiring prolonged exposure to manifest clinically meaningful outcomes.
Synaptic Plasticity: The Hidden Target
If vascular effects are only part of the story, synaptic plasticity may be the missing piece. The NO–cGMP–PKG–CREB signaling cascade, influenced by PDE5 inhibition, plays a central role in memory formation and consolidation.
In experimental models, PDE5 inhibitors enhance long-term potentiation, a process often described as the cellular basis of learning. By amplifying this signaling pathway, these drugs may counteract the synaptic deficits characteristic of Alzheimer’s disease.
This mechanism also aligns with findings from cellular studies. PDE5 inhibition has been shown to promote neurite outgrowth and reduce pathological tau phosphorylation—both key features of neurodegenerative processes .
There is a certain irony here. Drugs originally developed to improve vascular function in peripheral tissues may exert their most meaningful effects at the level of synaptic transmission in the brain. It is a reminder that pharmacology often reveals its full potential only after years of clinical use.
Clinical Limitations: Why Evidence Remains Inconclusive
Despite promising signals, several limitations hinder the translation of PDE5 inhibitors into dementia therapy. The most obvious is the lack of large, long-term randomized controlled trials. Most existing studies are either small, short in duration, or focused on surrogate endpoints.
Another challenge is drug selection. Not all PDE5 inhibitors are created equal. Differences in half-life, blood–brain barrier penetration, and off-target effects may significantly influence clinical outcomes. Tadalafil, with its longer duration of action, may offer advantages over shorter-acting agents, but this remains to be proven.
Patient selection is equally important. Dementia is a heterogeneous condition, often involving both neurodegenerative and vascular components. Identifying the subgroups most likely to benefit from PDE5 inhibition will be critical for future research.
Finally, there is the issue of dosing. Many studies may have used doses insufficient to achieve meaningful central nervous system effects. As noted in the source text, brain concentrations of PDE5 inhibitors can vary widely, and subtherapeutic exposure may lead to false-negative results .
Future Directions: Designing the Right Trial
If PDE5 inhibitors are to find a place in dementia treatment, future studies must address these limitations with precision. The design of clinical trials will be crucial, requiring careful consideration of duration, endpoints, and patient populations.
Key priorities include:
- Long-term treatment protocols, likely extending beyond 12 months
- Inclusion of older patients with mixed vascular and neurodegenerative pathology
- Use of biomarkers to assess both vascular and synaptic effects
- Selection of agents with optimal brain penetration and tolerability
Equally important is the integration of mechanistic and clinical data. Understanding how PDE5 inhibitors influence brain biology will help refine their therapeutic application and avoid the pitfalls of empirical treatment.
Conclusion: A Modest Drug with Ambitious Potential
PDE5 inhibitors occupy a unique position in modern medicine. Familiar, widely used, and generally well tolerated, they now stand at the threshold of a new therapeutic frontier. Their potential role in dementia reflects a broader shift toward systems-based thinking, where vascular and neuronal processes are understood as interconnected rather than separate.
The evidence to date is intriguing but incomplete. Animal studies are consistently positive, observational human data are encouraging, and clinical trials are cautiously neutral. This combination does not provide definitive answers—but it does justify further investigation.
Tadalafil and its pharmacological relatives may not be the long-awaited cure for dementia. But they offer something equally valuable: a new direction, grounded in biology and supported by real-world evidence. In a field often characterized by disappointment, that alone is worth pursuing.
FAQ: Key Questions About PDE5 Inhibitors in Dementia
1. Can PDE5 inhibitors like tadalafil improve memory?
There is promising preclinical evidence, but human data are still inconclusive. Long-term effects may differ from short-term trial results.
2. Do these drugs increase blood flow to the brain?
They can influence cerebral blood flow, but effects are modest and inconsistent. Cognitive benefits likely involve additional mechanisms.
3. Are PDE5 inhibitors currently approved for dementia treatment?
No. Their use in dementia is experimental and under investigation.
4. Why is tadalafil considered a strong candidate?
Its long half-life and ability to cross the blood–brain barrier make it suitable for chronic neurological applications.
5. What is the biggest challenge in this research area?
Designing large, long-term clinical trials that can detect subtle but meaningful cognitive changes in diverse patient populations.