Introduction: When Antidepressant Therapy Meets Sexual Physiology
Selective serotonin reuptake inhibitors (SSRIs) have transformed psychiatric care. Major depressive disorder, anxiety disorders, obsessive-compulsive disorder, premature ejaculation—these and other conditions are now routinely treated with serotonergic agents. Among them, paroxetine stands out for both its therapeutic potency and its relatively high incidence of sexual adverse effects.
Erectile dysfunction (ED) affects up to 50% of men receiving SSRIs . For many patients, this adverse effect becomes the silent reason for non-adherence. Mood may improve, but sexual function deteriorates. The clinical dilemma is not trivial: discontinuing antidepressants risks psychiatric relapse; continuing them may impair intimate relationships and quality of life.
The experimental study published in Eurasian Journal of Medicine examined a mechanistic and potentially therapeutic question: can tadalafil, a phosphodiesterase type 5 (PDE5) inhibitor, preserve penile nitric oxide synthase (NOS) activity in the presence of paroxetine-induced suppression?
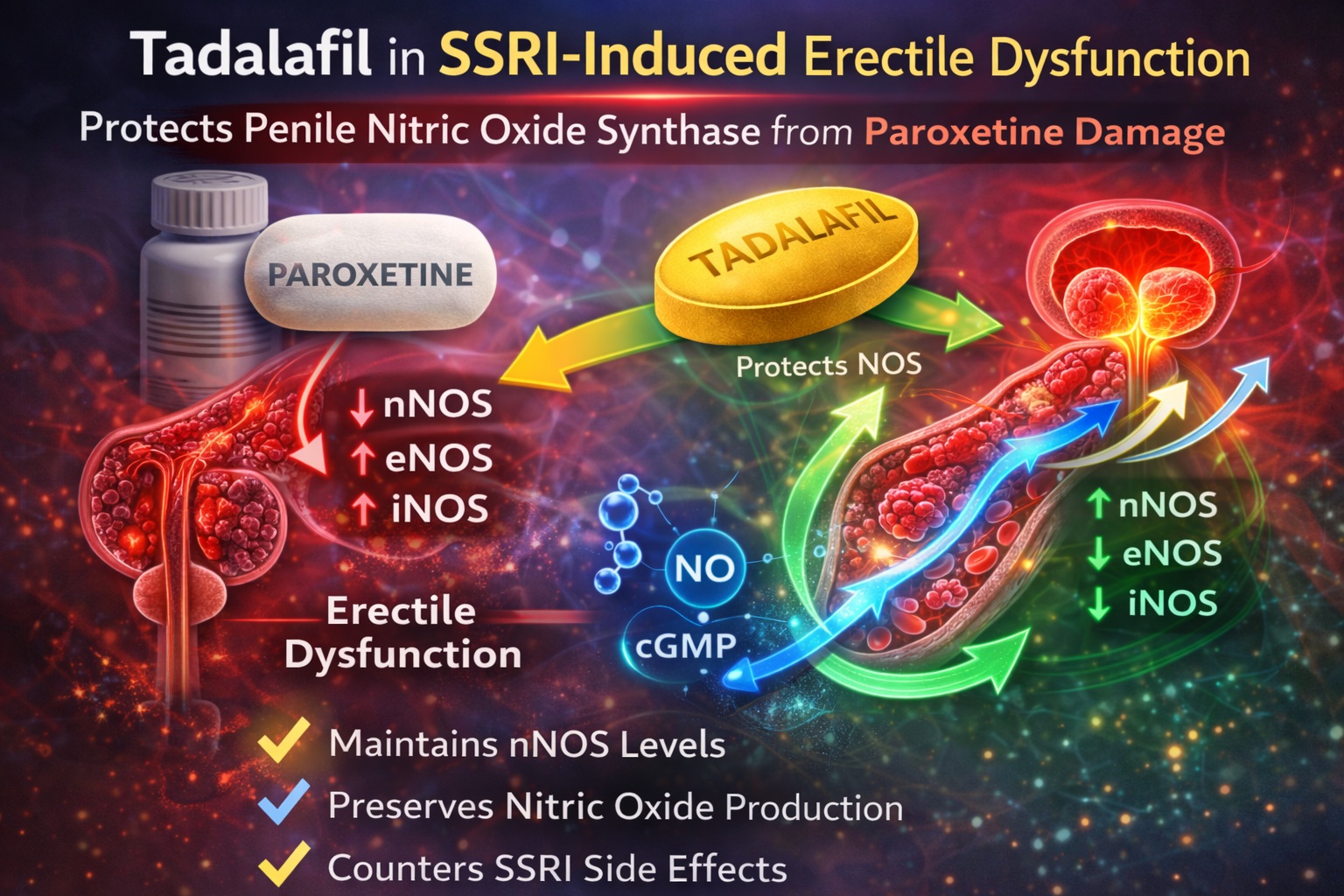
Using a controlled rat model, the investigators demonstrated that daily tadalafil prevented paroxetine-induced alterations in all three NOS isoforms—neuronal (nNOS), endothelial (eNOS), and inducible (iNOS). The findings illuminate the biochemical interplay between antidepressant therapy and erectile physiology, and offer translational relevance for clinical practice.
Nitric Oxide and the Erection Cascade: A Molecular Prerequisite
Penile erection is fundamentally a nitric oxide–dependent process. Neuronal nitric oxide synthase (nNOS) initiates the erectile response following cavernous nerve stimulation. Endothelial nitric oxide synthase (eNOS) sustains the response by maintaining blood inflow. Inducible NOS (iNOS), typically associated with inflammatory states, may modulate local vascular tone under pathological conditions.
Nitric oxide (NO) is synthesized from L-arginine by NOS enzymes. Once released, NO activates guanylate cyclase, increasing intracellular cyclic guanosine monophosphate (cGMP). This cascade leads to smooth muscle relaxation in the corpus cavernosum, enhanced arterial inflow, and venous occlusion—producing erection .
Phosphodiesterase type 5 (PDE5) degrades cGMP. Tadalafil inhibits PDE5, thereby sustaining cGMP levels and enhancing the erectile response. Importantly, tadalafil does not generate NO; it amplifies the downstream signal. Therefore, preservation of NOS activity remains critical for optimal pharmacological efficacy.
If paroxetine impairs NOS, tadalafil’s capacity to restore erectile function may be partially limited—unless it also modulates NOS expression or stability.
Paroxetine and Erectile Dysfunction: A Mechanistic Hypothesis
While all SSRIs increase serotonin levels, not all produce sexual dysfunction at equivalent frequencies. Paroxetine, in particular, has been associated with a high incidence of ED .
Previous research suggests that paroxetine uniquely inhibits NOS activity. Structural similarities to certain cytochrome P450 isoenzymes may underlie this inhibitory effect. In animal models, paroxetine reduces intracavernosal pressure responses to nerve stimulation and decreases plasma nitrate and nitrite levels—biochemical proxies for NO availability .
The present study hypothesized that co-administration of tadalafil might counteract these NOS alterations. Rather than simply augmenting cGMP, tadalafil could potentially stabilize or preserve NOS isoform expression.
This is a subtle but clinically meaningful distinction: rescue of signaling versus preservation of upstream enzymatic integrity.
Experimental Design: A Controlled Rat Model
Thirty male Sprague-Dawley rats were randomly assigned to three groups:
- Control (Group C): placebo
- Paroxetine only (Group P): 20 mg/kg daily
- Paroxetine plus tadalafil (Group P+T): 20 mg/kg paroxetine + 5 mg/kg tadalafil daily
Treatment continued for 28 days . On day 29, animals were sacrificed, and penile tissue was harvested for analysis.
Two complementary methodologies were used:
- Immunohistochemical staining with semi-quantitative scoring (0–3 scale)
- Enzyme-linked immunosorbent assay (ELISA) to quantify protein levels of nNOS, eNOS, and iNOS
This dual approach enhanced methodological rigor, combining visual tissue-level analysis with quantitative biochemical measurement.
Histopathological Findings: Paroxetine Disrupts NOS Balance
Immunohistochemical analysis revealed striking differences between groups.
In the paroxetine-only group, nNOS levels were significantly decreased compared to controls (0.6 ± 0.69 vs. 2.2 ± 0.42; p<0.001) . This reduction is particularly important, as nNOS initiates erection.
Conversely, eNOS and iNOS levels were significantly increased in the paroxetine group relative to controls (p=0.003 for eNOS; p=0.012 for iNOS) . Such upregulation may represent a compensatory or inflammatory response rather than functional enhancement.
When tadalafil was co-administered, nNOS levels were preserved and approached control values (1.7 ± 0.48; p=0.122 vs. control) . Moreover, the elevations in eNOS and iNOS seen with paroxetine alone were normalized.
These data suggest that tadalafil stabilizes the NOS isoform profile rather than merely amplifying downstream cGMP.
ELISA Quantification: Confirming Molecular Preservation
Quantitative ELISA data reinforced the histological findings.
Mean nNOS levels (ng/ml) were:
- Control: 3.578 ± 0.276
- Paroxetine: 1.963 ± 0.187
- Paroxetine + tadalafil: 3.508 ± 0.186
The decline in nNOS with paroxetine was statistically significant (p<0.001), and tadalafil co-treatment restored nNOS to near-control levels (p<0.001 vs. paroxetine alone).
Similarly, eNOS and iNOS were elevated with paroxetine and normalized with tadalafil. Importantly, there was no significant difference between control and paroxetine+tadalafil groups for these isoforms.
This pattern indicates that tadalafil does not simply “boost” NOS indiscriminately; it appears to prevent maladaptive alterations induced by paroxetine.
Mechanistic Interpretation: Beyond cGMP Amplification
How might tadalafil preserve NOS expression?
Several possibilities merit consideration:
- Improved penile hemodynamics may reduce oxidative stress and prevent NOS downregulation.
- Sustained cGMP signaling may exert feedback effects on NOS transcription.
- Tadalafil may modulate inflammatory pathways affecting iNOS expression.
- Enhanced endothelial function may stabilize eNOS activity.
While the study did not examine phosphorylation status or upstream transcriptional regulation, the consistent normalization of all three isoforms suggests a regulatory influence beyond mere enzymatic inhibition.
It is tempting to speculate that PDE5 inhibition may indirectly support NOS homeostasis by maintaining vascular integrity.
Clinical Translation: What Does This Mean for Patients?
Paroxetine remains widely prescribed for depression, anxiety disorders, premature ejaculation, and chronic pain syndromes . Sexual adverse effects remain a leading cause of treatment discontinuation.
The study’s conclusion—that daily tadalafil prevented chronic paroxetine-induced NOS alterations—carries clear translational implications .
In clinical practice, tadalafil may serve not only as symptomatic therapy for SSRI-associated ED but potentially as a prophylactic adjunct. Rather than waiting for erectile dysfunction to develop, early combination therapy might preserve physiological signaling pathways.
However, caution is warranted. Animal models provide mechanistic clarity but not direct human equivalence. Prospective clinical trials are necessary to confirm whether NOS preservation translates into durable functional benefit.
Limitations and Future Directions
The investigators acknowledge several limitations .
Intracavernosal pressure (ICP) measurements were not performed, which would have provided functional correlation. Phosphorylation states of NOS isoforms were not analyzed. Cell-specific localization of NOS changes was not separately quantified.
Future studies should:
- Assess functional erectile responses via ICP monitoring
- Examine oxidative stress markers
- Explore transcriptional regulation of NOS isoforms
- Conduct human translational trials in men receiving paroxetine
Despite these limitations, the study offers compelling biochemical evidence.
Broader Perspective: Integrating Psychiatry and Sexual Medicine
The intersection between psychiatry and sexual medicine is clinically unavoidable. Depression itself may impair libido and erection; antidepressants may compound the problem.
Rather than framing SSRI-induced ED as an unavoidable trade-off, emerging data support proactive management. Tadalafil—long recognized for its efficacy in ED—may possess additional biological benefits, including preservation of NOS integrity.
This perspective shifts the conversation from reactive symptom control to protective co-therapy.
Conclusion: Tadalafil as a Biochemical Safeguard
The study demonstrates that paroxetine significantly reduces penile nNOS levels while increasing eNOS and iNOS, disrupting the delicate nitric oxide balance necessary for erection .
Co-administration of tadalafil prevents these changes, restoring NOS isoform levels to near-normal values. The data suggest that tadalafil may preserve the molecular foundation of erectile function under SSRI exposure.
While translation to human practice requires further investigation, the concept is compelling: tadalafil does not merely treat ED—it may help protect the biochemical machinery that enables erection.
In a therapeutic landscape where mental health and sexual health often collide, such protective strategies deserve serious consideration.
FAQ
1. Why does paroxetine cause erectile dysfunction?
Paroxetine appears to inhibit neuronal nitric oxide synthase (nNOS), reducing nitric oxide production in penile tissue. This diminishes the NO-cGMP pathway necessary for erection.
2. How does tadalafil help in SSRI-induced erectile dysfunction?
Tadalafil inhibits PDE5, sustaining cGMP levels. In this study, it also preserved nNOS, eNOS, and iNOS levels in penile tissue, preventing paroxetine-induced molecular changes.
3. Can tadalafil be used preventively with paroxetine?
The animal data suggest potential protective effects. However, clinical trials in humans are needed before routine prophylactic co-administration can be recommended.