Introduction: A Quiet Drug Interaction With Loud Clinical Consequences
Modern medicine is built on combinations. Antibiotics are prescribed alongside cardiovascular drugs, metabolic agents, analgesics, and—more often than many clinicians openly acknowledge—phosphodiesterase type-5 inhibitors. Sildenafil, tadalafil, and vardenafil have long escaped the narrow label of “erectile dysfunction drugs” and now reside comfortably in everyday pharmacotherapy, particularly in aging male populations.
Ciprofloxacin, on the other hand, remains a cornerstone antibiotic. It is effective, familiar, and widely prescribed for urinary tract infections, prostatitis, respiratory infections, and complicated Gram-negative disease. For decades, it has been trusted to do its job decisively.
The problem begins when trust becomes habit.
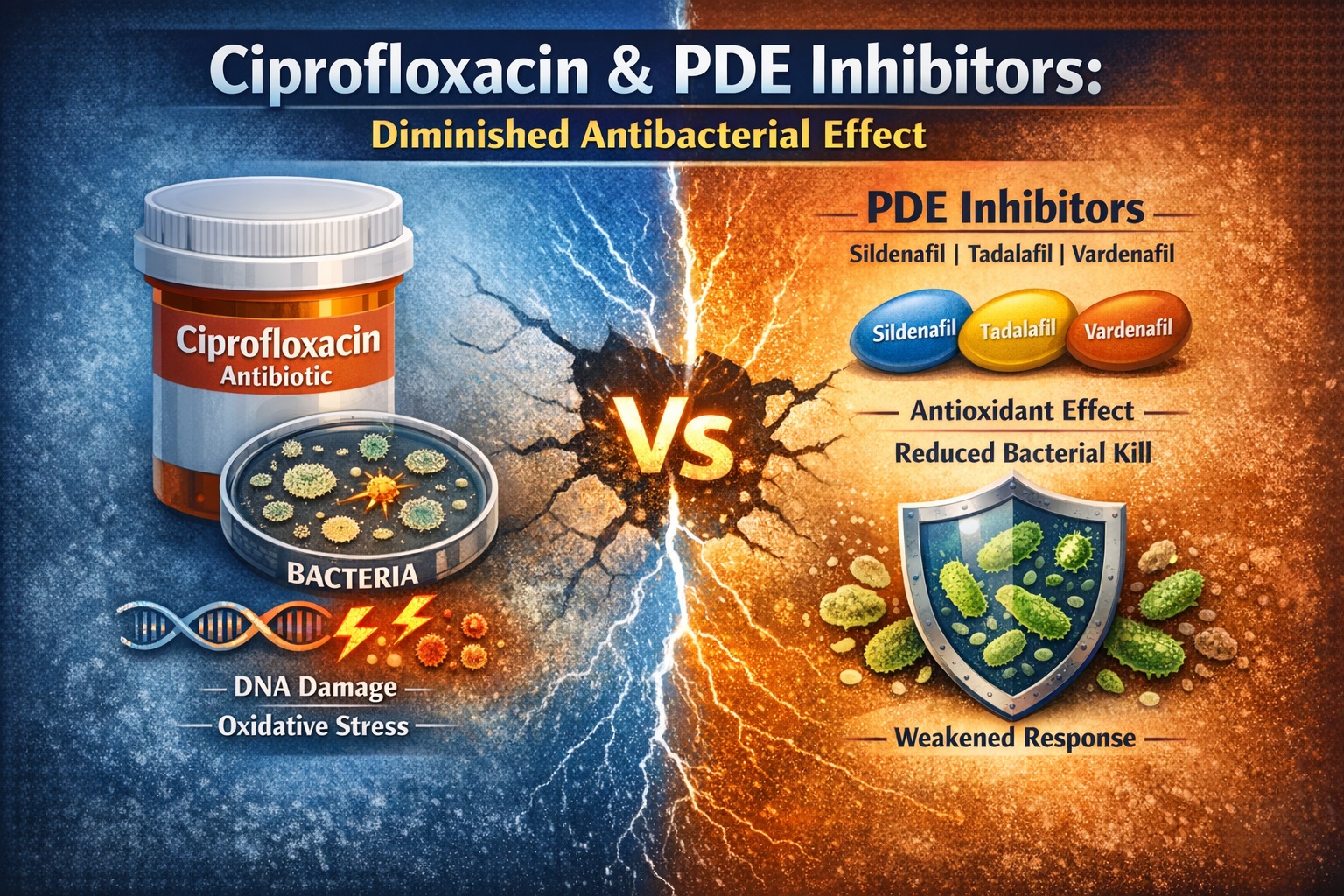
Emerging experimental evidence demonstrates that phosphodiesterase inhibitors (PDE-5 inhibitors) can significantly attenuate the antibacterial activity of ciprofloxacin. This interaction is subtle, clinically silent at first glance, and therefore dangerous. No dramatic toxicity alerts the clinician. No obvious adverse reaction warns the patient. Instead, the antibiotic simply works less well.
This article explores the pharmacological meaning of this interaction, explains why it matters far beyond the laboratory, and outlines what clinicians must reconsider when prescribing ciprofloxacin in patients using PDE inhibitors.
Ciprofloxacin: More Than a DNA Gyrase Inhibitor
Ciprofloxacin belongs to the fluoroquinolone class, and its primary antibacterial mechanism is well known. It inhibits bacterial DNA gyrase and topoisomerase IV, enzymes essential for DNA replication and transcription. This leads to stalled replication forks, DNA damage, and ultimately bacterial death.
However, focusing exclusively on DNA gyrase inhibition is an oversimplification.
Ciprofloxacin is also a redox-active antibiotic. Its bactericidal activity involves the generation of reactive oxygen species (ROS) within bacterial cells. These oxidative radicals damage proteins, lipids, and nucleic acids, amplifying lethal stress beyond topoisomerase inhibition. In susceptible bacteria, this oxidative burst is not incidental—it is central to killing efficiency.
This dual mechanism explains why ciprofloxacin can be rapidly bactericidal and why antioxidants have repeatedly been shown to blunt its efficacy. Any agent capable of dampening oxidative stress inside bacterial cells has the theoretical potential to weaken ciprofloxacin’s action.
Phosphodiesterase inhibitors fall precisely into that category.
Phosphodiesterase Inhibitors: Systemic Drugs With Unexpected Microbial Effects
PDE-5 inhibitors were developed to enhance cyclic guanosine monophosphate (cGMP) signaling in smooth muscle. By inhibiting cGMP degradation, they promote vasodilation, improved blood flow, and sustained smooth-muscle relaxation.
Yet these drugs are pharmacologically promiscuous.
Beyond vascular effects, PDE inhibitors demonstrate antioxidant properties, immunomodulatory behavior, and measurable effects on cellular energy metabolism and mitochondrial function. These properties are well documented in mammalian systems and increasingly recognized in microbial contexts.
Importantly, bacteria possess cyclic nucleotide signaling systems. While simpler than mammalian pathways, bacterial phosphodiesterase activity exists and participates in metabolic regulation and stress responses. When PDE inhibitors enter bacterial environments—particularly in the urinary tract—they may interfere with these systems in ways never intended by drug designers.
The result is not bacterial death, but bacterial protection.
Experimental Evidence: Ciprofloxacin Loses Its Edge
In controlled in-vitro experiments using standard reference strains, ciprofloxacin demonstrates strong antibacterial activity across multiple Gram-negative and Gram-positive organisms. Zones of inhibition are robust, and minimum inhibitory concentrations (MICs) remain predictably low.
That picture changes dramatically when PDE inhibitors are introduced.
When bacteria are pretreated with sildenafil, tadalafil, or vardenafil, ciprofloxacin’s effectiveness is sharply reduced. Zones of inhibition shrink significantly. MIC values increase by several orders of magnitude. In practical terms, bacteria that were previously sensitive behave as if they were approaching resistance.
This effect is consistent across diverse organisms, including Escherichia coli, Staphylococcus aureus, Pseudomonas aeruginosa, Acinetobacter baumannii, and Klebsiella pneumoniae. The phenomenon is not strain-specific and cannot be dismissed as experimental noise.
Crucially, this attenuation occurs without any direct interference with ciprofloxacin’s ability to inhibit DNA gyrase. Enzymatic assays confirm that PDE inhibitors do not block ciprofloxacin’s primary molecular target.
The implication is clear: the interaction occurs downstream, at the level of bacterial stress handling, oxidative balance, or energy metabolism.
Oxidative Stress: The Missing Link
To understand this interaction, one must abandon the outdated notion that antibiotics work through single, isolated targets. Ciprofloxacin kills bacteria not only by freezing DNA replication, but by overwhelming cellular defense systems.
Reactive oxygen species play a pivotal role in this process. Superoxide radicals and hydrogen peroxide accumulate rapidly after ciprofloxacin exposure, pushing bacterial cells beyond repair capacity. Sensitive bacteria succumb; resistant ones often possess superior antioxidant defenses.
Phosphodiesterase inhibitors, by virtue of their antioxidant and cytoprotective effects, appear to tip this balance.
By reducing oxidative stress or enhancing bacterial tolerance to it, PDE inhibitors allow bacteria to survive ciprofloxacin exposure that would otherwise be lethal. The antibiotic is still present. The DNA gyrase is still inhibited. But the cell does not cross the threshold into irreversible damage.
In effect, the drug becomes bacteriostatic where it was once bactericidal.
Why This Matters Clinically: The Illusion of Adequate Therapy
From a clinician’s perspective, this interaction is particularly insidious.
There is no warning label suggesting that sildenafil or tadalafil may compromise antibiotic therapy. There is no immediate clinical failure in every patient. Instead, there is delayed bacterial clearance, prolonged infection, and increased opportunity for resistance development.
The scenario is especially relevant in older male patients, where urinary tract infections and PDE-5 inhibitor use frequently overlap. A patient receiving ciprofloxacin for prostatitis while using tadalafil may appear compliant and adequately treated, yet experience suboptimal bacterial eradication.
This is not a pharmacokinetic interaction in the classic sense. Ciprofloxacin plasma levels remain unchanged. There is no overt toxicity. The failure occurs silently, at the microbial level.
In an era where antimicrobial resistance is already a global crisis, silent antagonism between commonly prescribed drugs is the last thing clinicians can afford to ignore.
Implications for Antimicrobial Stewardship
Antibiotic stewardship traditionally focuses on dose, duration, and spectrum. Drug-drug interactions that reduce antibacterial efficacy without causing toxicity have received far less attention.
The interaction between ciprofloxacin and PDE inhibitors highlights a blind spot in current prescribing logic. Clinicians routinely consider cytochrome P450 interactions, QT prolongation, and renal clearance. Rarely do they consider whether a non-antibiotic drug might protect bacteria from oxidative damage.
This oversight has consequences.
Repeated exposure of bacteria to sublethal antibiotic stress accelerates adaptive responses. What begins as pharmacological attenuation may end as genuine resistance. The patient receives a standard dose, the pathogen survives, and future options narrow.
Stewardship must evolve beyond pharmacokinetics into pharmacodynamic compatibility.
Are All PDE Inhibitors the Same?
Interestingly, the attenuating effect appears consistent across sildenafil, tadalafil, and vardenafil, despite their differing pharmacokinetic profiles. This suggests a class effect rather than a molecule-specific phenomenon.
While differences in half-life and tissue distribution may influence the magnitude of interaction in vivo, the underlying mechanism—interference with oxidative stress-mediated killing—appears shared.
This raises important questions for future research. Are other PDE inhibitors implicated? Do lower doses produce the same effect? Is the interaction clinically significant in all infection sites, or primarily in the urinary tract where drug concentrations overlap?
At present, caution is warranted rather than speculation.
Practical Considerations for Clinicians
Clinicians should not interpret these findings as a reason to abandon ciprofloxacin or PDE inhibitors. Rather, they should prompt thoughtful prescribing.
When possible, temporary discontinuation of PDE inhibitors during fluoroquinolone therapy may be reasonable, particularly in severe infections. Alternatively, selecting antibiotics whose killing mechanisms do not rely heavily on oxidative stress may reduce risk.
Most importantly, clinicians should remain vigilant in cases of unexpected treatment failure. When ciprofloxacin appears less effective than anticipated, concurrent medications should be reviewed—not only for toxicity, but for antagonism.
Medicine advances not only by discovering new drugs, but by understanding how old ones behave together.
Conclusion: Small Molecules, Big Consequences
The interaction between ciprofloxacin and phosphodiesterase inhibitors challenges comfortable assumptions about drug compatibility. It reminds us that bacteria are not passive targets, but dynamic systems capable of exploiting pharmacological gaps.
Ciprofloxacin remains a powerful antibiotic. PDE inhibitors remain valuable therapeutic agents. But when combined, their interaction may quietly undermine treatment success.
In the age of precision medicine, ignorance is no longer an acceptable defense. Understanding such interactions is not academic curiosity—it is clinical responsibility.
FAQ
1. Does taking sildenafil make ciprofloxacin completely ineffective?
No. Ciprofloxacin retains activity, but its antibacterial potency is significantly reduced. This may lead to slower bacterial clearance or treatment failure in some patients.
2. Is this interaction proven in humans or only in laboratory studies?
Current evidence is experimental, but the biological mechanisms involved are well established. Given the widespread use of both drug classes, the findings are clinically concerning and warrant caution.
3. Should patients stop PDE inhibitors when prescribed ciprofloxacin?
This decision should be individualized. In serious infections, temporary discontinuation may be considered. Patients should never stop prescribed medications without consulting their physician.