Introduction
Few drugs in oncology embody both brilliance and betrayal as much as doxorubicin. This anthracycline antibiotic, celebrated for its wide-spectrum anticancer activity, has been a cornerstone of chemotherapy since the 1970s. Its power to intercalate into DNA and inhibit topoisomerase II revolutionized the treatment of malignancies ranging from breast and ovarian cancer to hematologic malignancies such as leukemia and lymphoma. Yet, its remarkable efficacy comes at a steep biological cost: severe off-target toxicity.
Among the collateral damages, hepatorenal toxicity stands out. The liver and kidneys, crucial guardians of detoxification and homeostasis, bear the brunt of oxidative stress and inflammatory cascades triggered by doxorubicin. As survival in oncology improves, so too does the urgency of mitigating these toxicities.
Enter tadalafil, a drug better known for improving quality of life in very different circumstances. As a phosphodiesterase-5 (PDE5) inhibitor, tadalafil has carved a reputation in the treatment of erectile dysfunction and pulmonary arterial hypertension. However, accumulating evidence suggests its pharmacological reach may extend much further. Its ability to modulate oxidative stress, inflammatory signaling, and endothelial function positions it as an unlikely yet promising candidate for chemoprevention in oncology.
This article explores findings from recent experimental research evaluating tadalafil’s potential to protect against doxorubicin-induced hepatorenal injury in Wistar rats. By weaving together mechanisms of oxidative stress, cytokine signaling, enzymatic defenses, and histopathology, we aim to illuminate a new frontier where a “cardiovascular drug” could be repurposed as a protector in chemotherapy.
The Paradox of Doxorubicin: Life-Saving Yet Organ-Toxic
Doxorubicin’s therapeutic appeal lies in its dual mechanism of DNA intercalation and topoisomerase II inhibition. These actions cripple tumor cell replication and survival. Unfortunately, the same mechanisms ignite collateral biochemical storms in healthy tissues.
The drug’s metabolism produces quinone-based free radicals, which in turn propagate reactive oxygen species (ROS). When antioxidant defenses are overwhelmed, the balance tilts toward oxidative stress, leading to lipid peroxidation, protein denaturation, and DNA fragmentation.
The liver, with its central role in drug metabolism and detoxification, is particularly vulnerable. Elevated serum aminotransferases and bilirubin levels are common harbingers of hepatic injury in doxorubicin-treated patients. The kidneys, guardians of electrolyte balance and excretion, similarly exhibit nephrotoxic profiles: rising serum creatinine, urea retention, and disrupted electrolyte homeostasis.
Ironically, while doxorubicin’s tumoricidal properties are lifesaving, its multi-organ toxicity can compromise the very survival it promises. Thus, oncological progress must be accompanied by strategies to shield non-cancerous tissues without diminishing therapeutic efficacy.
Tadalafil: Beyond Erectile Dysfunction
Phosphodiesterase-5 inhibitors such as tadalafil, sildenafil, and vardenafil were originally developed to target vascular smooth muscle relaxation through the cyclic guanosine monophosphate (cGMP) pathway. In erectile dysfunction and pulmonary hypertension, this translates into enhanced vasodilation and improved hemodynamics.
But biological curiosity has revealed unexpected therapeutic avenues. Preclinical studies have highlighted tadalafil’s protective roles in:
- Cisplatin-induced nephrotoxicity
- Adriamycin-induced cardiomyopathy
- Testicular toxicity
- Peripheral neuropathy and pain syndromes
- Benign prostatic hyperplasia
- Type 2 diabetes mellitus
The pharmacological threads connecting these indications are oxidative stress reduction, anti-inflammatory modulation, and endothelial protection. These attributes make tadalafil a candidate worth exploring in oncology’s toxicity paradox.
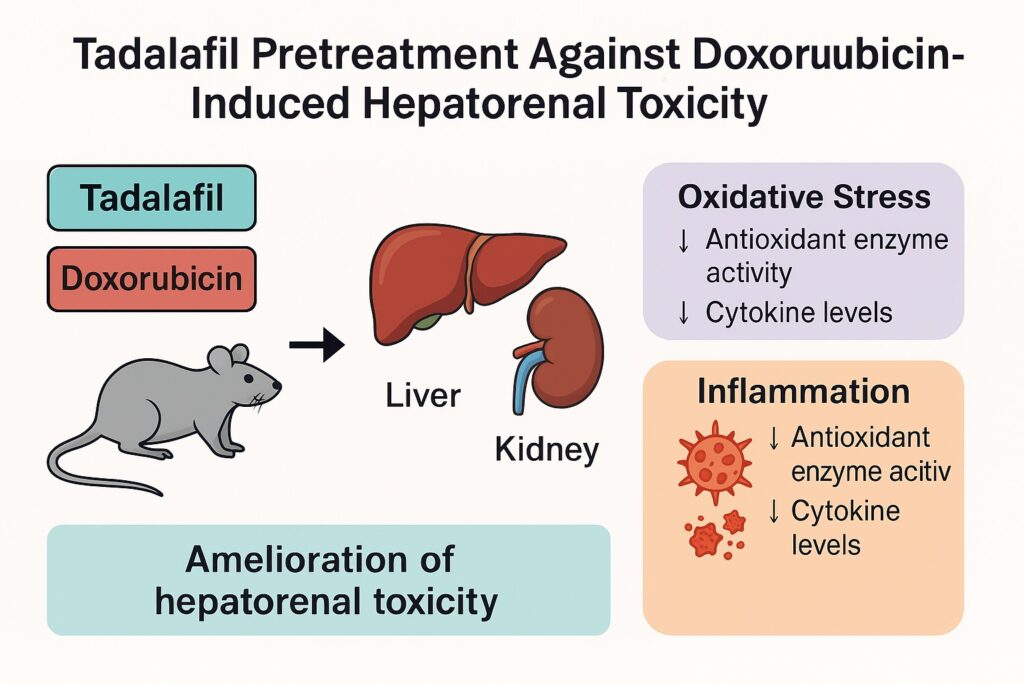
Study Design: Modeling Hepatorenal Protection
To evaluate tadalafil’s chemoprotective potential, investigators employed a controlled rat model of doxorubicin-induced hepatorenal injury.
Experimental Framework
- Subjects: Adult male Wistar rats, 10–12 weeks old, weighing 180–200 g.
- Groups: Eight groups of six rats each, including untreated controls, doxorubicin-only rats, and treatment cohorts pretreated with silymarin (a standard hepatoprotective reference drug) or graded doses of tadalafil (2.5, 5, and 10 mg/kg/day).
- Intervention: Oral pretreatment one hour before intraperitoneal doxorubicin injections (2.5 mg/kg on alternate days for 12 days).
- Endpoints:
- Serum liver and renal function tests.
- Antioxidant enzyme activity (SOD, CAT, GST, GPx, GSH, MDA).
- Pro-inflammatory cytokines (IL-1β, IL-6, TNF-α).
- Histopathological evaluation of liver and kidney tissues.
This experimental framework was designed to reflect both biochemical markers of organ function and structural pathology, providing a holistic assessment of tadalafil’s protective capacity.
Key Findings: Biochemistry Meets Histology
Body Weight and Organ Weights
As expected, doxorubicin-treated rats suffered significant weight loss, reflecting systemic toxicity and anorexia commonly reported in chemotherapy. While tadalafil did not reverse overall weight decline, it favorably influenced relative liver weight, mitigating disproportionate enlargement associated with hepatic injury.
Hepatic Function
Doxorubicin intoxication raised serum ALT, AST, and bilirubin, hallmarks of hepatocellular damage. Albumin and total protein levels fell, underscoring impaired synthetic capacity.
Pretreatment with tadalafil significantly reduced ALT and bilirubin, while restoring albumin and protein levels. Interestingly, AST levels remained elevated, possibly reflecting extrahepatic contributions such as muscle injury—an important nuance in interpreting hepatic panels.
Renal Function
Doxorubicin elevated serum potassium, creatinine, and urea, consistent with nephrotoxicity, while reducing bicarbonate, chloride, and calcium.
Tadalafil pretreatment ameliorated electrolyte imbalances and reduced markers of renal dysfunction, highlighting its renoprotective profile. Sodium levels, however, remained largely unaltered across groups.
Antioxidant Defense
Perhaps the most striking findings emerged from oxidative stress assays. Doxorubicin dramatically reduced SOD, CAT, GST, and GPx activity, while elevating malondialdehyde (MDA), a marker of lipid peroxidation.
Tadalafil pretreatment reversed these trends, significantly enhancing antioxidant enzyme activity and reducing MDA accumulation. In essence, tadalafil restored the redox balance crucial for cellular survival.
Cytokine Modulation
In doxorubicin-only groups, hepatic and renal tissues showed surges in IL-1β, IL-6, and TNF-α, underscoring an inflammatory cascade compounding oxidative damage.
Tadalafil pretreatment suppressed these cytokines, bringing levels closer to baseline. This anti-inflammatory property is particularly relevant given the role of cytokines in chemotherapy-induced cachexia and multi-organ injury.
Histopathology
Under the microscope, the narrative became vivid.
- Liver: Doxorubicin produced central venous congestion, hepatocyte vacuolation, and lymphocytic infiltration. Tadalafil-pretreated rats exhibited markedly improved architecture, with reduced congestion and preserved hepatocytes, especially at higher doses.
- Kidneys: Doxorubicin caused glomerular atrophy and tubulointerstitial congestion. Tadalafil-pretreated kidneys showed remarkable preservation of normal glomeruli and tubules, with significantly less congestion and atrophy.
Histology confirmed what biochemistry suggested: tadalafil guards cellular integrity against doxorubicin assault.
Mechanistic Insights: How Does Tadalafil Work Here?
The study supports several interconnected mechanisms through which tadalafil confers hepatorenal protection:
- Antioxidant Restoration: By upregulating endogenous enzymes (SOD, CAT, GPx, GST), tadalafil curbs oxidative stress and reduces lipid peroxidation.
- Anti-Inflammatory Modulation: Suppression of IL-1β, IL-6, and TNF-α blunts inflammatory injury to parenchymal cells.
- Endothelial Protection: Through PDE5 inhibition and cGMP signaling, tadalafil enhances microvascular function, improving perfusion and reducing ischemic damage.
- Cellular Preservation: Histological evidence suggests tadalafil prevents necrotic cascades and maintains tissue structure.
Taken together, these mechanisms reveal tadalafil not as a miracle cure but as a biochemical stabilizer, tilting the balance back toward homeostasis in an environment otherwise overwhelmed by free radicals and inflammatory cytokines.
Clinical Implications and Future Directions
The translation from rat models to clinical oncology is never straightforward. Yet the findings warrant optimism and careful pursuit.
- Adjunctive Chemoprotection: If replicated in humans, tadalafil could join the armamentarium of supportive care, shielding patients from doxorubicin’s hepatorenal fallout.
- Repurposing an Established Drug: Tadalafil’s established safety profile in cardiovascular and urological diseases accelerates the pathway to potential clinical application in oncology.
- Dose Optimization: Determining the most effective protective dose without interfering with anticancer efficacy will be crucial.
- Combinatorial Strategies: Pairing tadalafil with other antioxidants (e.g., silymarin) may yield synergistic benefits.
- Patient-Centered Outcomes: Ultimately, the goal is not merely biochemical normalization but improved quality of life and tolerability of life-saving chemotherapy.
Conclusion
Doxorubicin remains a paradox: a lifesaver shadowed by toxicity. This experimental evidence positions tadalafil as a promising chemoprotective adjuvant, capable of attenuating doxorubicin-induced hepatorenal injury through modulation of oxidative stress and inflammatory pathways.
What was once a drug to treat erectile dysfunction may soon find a role in oncology wards, not for enhancing intimacy, but for safeguarding livers and kidneys as patients battle cancer. The irony is poetic—and potentially life-changing.
FAQ
1. Does tadalafil reduce the anticancer efficacy of doxorubicin?
Current preclinical evidence focuses on toxicity mitigation, not anticancer efficacy. There is no indication that tadalafil diminishes doxorubicin’s tumoricidal action, but human studies are required to confirm this balance.
2. Could tadalafil be used in cancer patients already on PDE5 inhibitors for other reasons?
Yes, theoretically. Patients already receiving tadalafil for erectile dysfunction or pulmonary hypertension may experience protective effects during doxorubicin therapy, though clinical validation is essential before formal recommendations.
3. Is tadalafil superior to standard hepatoprotective agents like silymarin?
In the study, both silymarin and tadalafil showed protective effects. Tadalafil’s added benefits include anti-inflammatory and endothelial actions, suggesting it may complement, rather than replace, existing agents.