Introduction
Phosphodiesterase type 5 (PDE5) inhibitors have long been a cornerstone in the management of erectile dysfunction (ED) and pulmonary arterial hypertension (PAH). Among these, tadalafil has gained prominence for its extended half-life, once-daily dosing, and therapeutic versatility. Yet, despite its established role, the oral delivery of tadalafil poses persistent challenges. Poor aqueous solubility, variable bioavailability, and first-pass hepatic metabolism restrict its pharmacokinetic profile, often necessitating higher doses and delaying therapeutic onset.
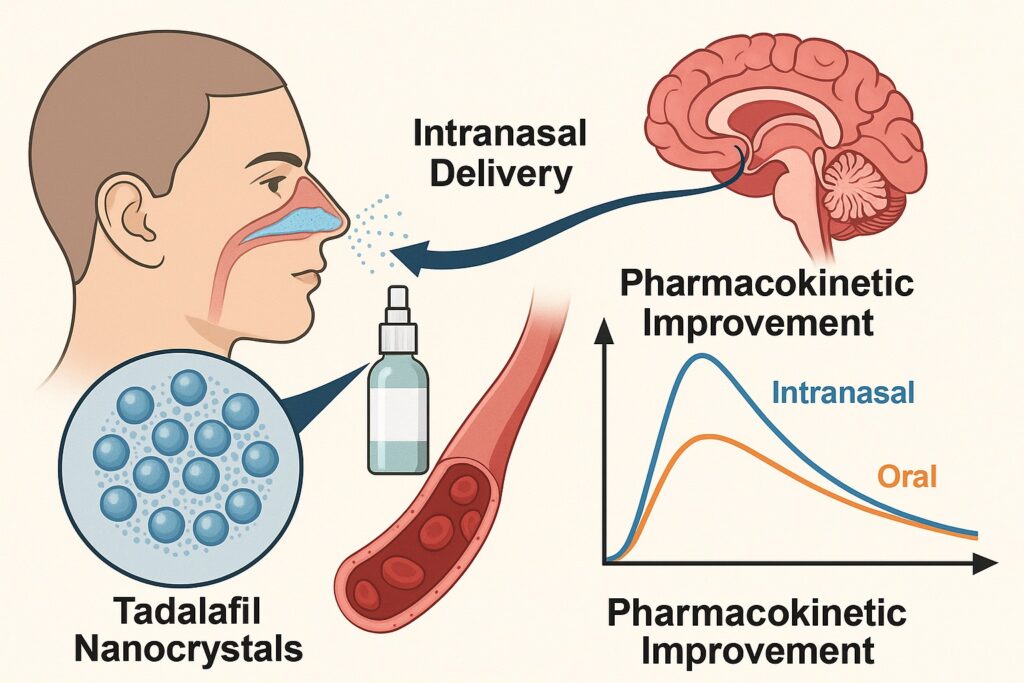
The pharmaceutical sciences, never content with stagnation, have continuously sought solutions to such limitations. One of the most promising directions in recent years has been the design of nanocrystal formulations. By engineering drug particles into the nanometer range, solubility and dissolution rates can be enhanced dramatically, potentially revolutionizing pharmacotherapy. This study of intranasal tadalafil nanocrystals exemplifies this innovation, exploring whether a drug traditionally confined to oral dosing can find new life through a novel delivery route.
Intranasal administration is particularly attractive for lipophilic drugs like tadalafil. The nasal mucosa provides a highly vascularized surface for rapid systemic absorption, circumventing the gastrointestinal tract and hepatic first-pass effect. In theory, this should translate into faster onset of action, higher bioavailability, and reduced interpatient variability. When combined with nanocrystal technology, intranasal delivery promises not only efficiency but also precision, offering patients a therapeutic option that is both rapid and reliable.
This article delves into the rationale, methodology, and outcomes of developing tadalafil intranasal nanocrystals, placing the findings into a broader clinical and pharmacological context. It highlights the interplay between pharmaceutical technology, pharmacokinetics, and patient-centered care, illustrating how a century-old aspiration—to overcome the limitations of oral drug delivery—is gradually materializing.
The Rationale for Nanocrystal Technology
The solubility issue with tadalafil is emblematic of a broader challenge in drug development. Nearly 40% of newly discovered drug candidates are classified as poorly water-soluble, which severely limits their oral bioavailability. Tadalafil falls squarely into this category, with low dissolution rates restricting absorption in the gastrointestinal tract.
Nanocrystal technology addresses this limitation by reducing drug particles to nanometer dimensions, thereby increasing their surface area-to-volume ratio. According to the Noyes–Whitney equation, dissolution rate is directly proportional to surface area; hence, smaller particles dissolve faster and more completely. Furthermore, nanocrystals possess high saturation solubility due to increased surface free energy, enabling improved dissolution even in limited fluid volumes such as those present in nasal mucosa.
Another distinct advantage lies in the stabilization of these nanosystems. By selecting appropriate stabilizers and surfactants, nanocrystals can resist agglomeration, maintain their size distribution, and remain effective over extended storage periods. Thus, when formulated correctly, nanocrystals provide not only improved pharmacokinetics but also practical pharmaceutical stability, ensuring translation from bench to bedside.
Why Intranasal Delivery?
The intranasal route offers unique pharmacological benefits that oral tablets cannot match. First, the nasal cavity’s rich vascular network facilitates direct entry into systemic circulation, eliminating hepatic first-pass metabolism. This feature alone increases the fraction of the drug that reaches circulation intact, often lowering the necessary dose.
Second, intranasal delivery yields a rapid onset of action, a characteristic especially relevant for conditions like erectile dysfunction where timing is crucial. Patients often prioritize spontaneity and minimal delay between dosing and effect. A formulation that can shorten onset from hours to minutes offers not only clinical but also psychological benefits, improving satisfaction and adherence.
Third, the intranasal route may expand the therapeutic applications of tadalafil. In pulmonary hypertension, rapid systemic vasodilation is desirable, and bypassing gastrointestinal variability could make treatment more predictable. Similarly, for patients with dysphagia or gastrointestinal disorders, nasal delivery provides a practical alternative.
Nonetheless, intranasal administration also presents challenges. The limited volume capacity of the nasal cavity restricts the dose deliverable at one time, necessitating high-potency and efficiently absorbed formulations. Tadalafil’s low solubility would typically disqualify it from such a route, but nanocrystal engineering reopens the door, enabling sufficient concentrations in small volumes to achieve therapeutic plasma levels.
In Vitro Characterisation of Tadalafil Nanocrystals
The study employed established methodologies to prepare tadalafil nanocrystals, utilizing techniques such as high-pressure homogenization. Critical attributes including particle size, polydispersity index (PDI), zeta potential, and crystallinity were rigorously analyzed, as these parameters collectively determine stability and performance.
Particle size analysis revealed that nanocrystals consistently fell within the optimal sub-300 nm range, ensuring not only rapid dissolution but also mucosal permeability. A low PDI indicated uniform distribution, essential for predictable pharmacokinetics and reduced aggregation risk. Meanwhile, zeta potential measurements provided insight into colloidal stability, with sufficiently negative values minimizing particle agglomeration during storage and application.
Differential scanning calorimetry and X-ray diffraction analyses confirmed that tadalafil retained its crystalline state post-processing, avoiding undesirable amorphization that could destabilize the formulation. Importantly, dissolution studies demonstrated a marked increase in tadalafil’s solubility and dissolution rate compared to conventional crystalline forms. Together, these findings validated the scientific rationale of combining nanocrystal engineering with intranasal delivery.
In Vivo Pharmacokinetic Findings
The pharmacokinetic evaluation represented the critical test of this innovation. Animal studies compared intranasal nanocrystal formulations with conventional oral tadalafil, assessing parameters such as maximum plasma concentration (Cmax), time to maximum concentration (Tmax), area under the curve (AUC), and half-life (t1/2).
Results were striking. Intranasal delivery achieved significantly higher Cmax values within a markedly shorter Tmax, confirming both faster absorption and greater systemic exposure. The AUC values, reflecting total drug exposure, were substantially improved, indicating enhanced bioavailability. Importantly, the half-life remained consistent with tadalafil’s known pharmacology, preserving its advantage of prolonged action despite altered delivery.
These pharmacokinetic gains translate into tangible clinical implications. A patient receiving intranasal tadalafil nanocrystals could expect more rapid onset of action, stronger therapeutic effects at lower doses, and reduced variability compared with oral administration. This is not merely a laboratory curiosity but a potential shift in how tadalafil and similar drugs are administered in clinical practice.
Clinical Implications and Therapeutic Potential
The development of intranasal tadalafil nanocrystals holds implications beyond incremental pharmacological improvements. It challenges clinicians and pharmaceutical developers to rethink drug delivery for established molecules, demonstrating that innovation does not always require inventing new drugs but rather re-engineering existing ones for better performance.
For erectile dysfunction, the benefits are clear. Faster onset of action addresses one of the most frequently cited patient concerns regarding oral PDE5 inhibitors: the delay between dosing and effect. Improved predictability may reduce anxiety, enhance sexual confidence, and increase adherence.
In pulmonary arterial hypertension, a condition requiring precise hemodynamic modulation, intranasal delivery could optimize therapeutic onset while minimizing systemic side effects linked to variable oral absorption. Moreover, in populations with polypharmacy or hepatic impairment, bypassing first-pass metabolism could reduce drug–drug interactions and hepatotoxic risk.
There is also scope for extending intranasal nanocrystal technology to other PDE5 inhibitors or drugs with similar solubility constraints. By addressing fundamental pharmacokinetic limitations, this platform could serve as a model for the re-engineering of multiple therapeutic agents across diverse fields.
Challenges and Future Perspectives
While the study presents compelling evidence, clinical translation is not without obstacles. Regulatory pathways for nanomedicines remain stringent, requiring extensive evaluation of safety, stability, and long-term mucosal tolerability. The nasal cavity is a delicate anatomical site, and chronic administration raises questions about local irritation, mucociliary clearance, and patient acceptance.
Manufacturing scalability is another consideration. Laboratory-scale nanocrystal preparation must be translated into robust industrial processes capable of consistent quality and stability. Stability testing over prolonged periods, under varied storage conditions, will be crucial for regulatory approval and market viability.
Future research should also explore patient-reported outcomes, comparing satisfaction, adherence, and quality of life with intranasal versus oral tadalafil. Furthermore, pharmacodynamic studies in clinical populations—not just animal models—are essential to confirm whether the pharmacokinetic advantages translate into superior therapeutic outcomes.
Despite these hurdles, the trajectory is promising. As precision medicine advances, drug delivery innovations like intranasal nanocrystals will become increasingly important, aligning therapeutic strategies with patient needs, physiology, and lifestyle.
Conclusion
The investigation into intranasal tadalafil nanocrystals represents more than an academic exercise. It is a proof-of-concept that drug delivery can be reimagined, even for well-established molecules. By leveraging nanotechnology and alternative routes of administration, limitations once accepted as inevitable can be overcome.
For tadalafil, this means transforming a drug already valued for its duration of action into one that also offers rapid onset, improved bioavailability, and potentially lower dosing requirements. Such innovation could elevate patient satisfaction, optimize clinical outcomes, and broaden therapeutic utility across multiple indications.
In the broader sense, this research underscores a central truth of pharmaceutical science: innovation lies not only in discovering new drugs but in refining how we use the ones we already have. The story of intranasal tadalafil nanocrystals may well be the beginning of a new chapter in precision drug delivery, where pharmacology meets patient-centered design to achieve outcomes once thought unattainable.
FAQ
1. How do intranasal nanocrystals improve tadalafil absorption compared to oral tablets?
They bypass gastrointestinal dissolution and first-pass metabolism, dissolve more rapidly due to increased surface area, and are absorbed directly through the nasal mucosa into systemic circulation, leading to higher and faster drug levels.
2. Is intranasal tadalafil expected to replace oral tablets?
Not entirely. Oral tablets remain convenient for many patients. Intranasal formulations are more likely to serve as complementary options, particularly for those who require rapid onset, have gastrointestinal issues, or need reduced dosing variability.
3. What are the main hurdles before intranasal tadalafil nanocrystals can reach clinical practice?
The key challenges include demonstrating long-term safety in humans, ensuring large-scale manufacturing stability, navigating regulatory approval processes, and conducting robust clinical trials to confirm pharmacodynamic and patient-reported benefits.