Introduction
Adverse drug reactions represent one of the most intricate puzzles in modern medicine. While the majority of cutaneous drug reactions present as relatively benign rashes, there exists a distinct subset that is both diagnostically challenging and potentially life-threatening. Among these, the fixed drug eruption (FDE) occupies a unique niche, characterized by its recurring lesions at identical anatomical sites upon re-exposure to the offending agent. Though not the most common of drug-induced skin reactions, FDE is of particular interest because of its dramatic clinical presentation and the importance of precise etiological identification.
A fixed drug eruption manifests as erythematous or violaceous round-to-oval patches, often with a dusky center. In severe cases, the reaction progresses to form vesicles and bullae, creating diagnostic overlap with other blistering conditions such as erythema multiforme (EM) or bullous pemphigoid. For the clinician, navigating these diagnostic crossroads requires a nuanced understanding of clinical morphology, histopathology, and patient history.
The article at hand focuses on a rare but increasingly relevant trigger of FDE: tadalafil, a phosphodiesterase-5 inhibitor widely used for erectile dysfunction and benign prostatic hyperplasia. While tadalafil is FDA-approved and generally well tolerated, its misuse, particularly in unregulated over-the-counter sexual enhancement supplements, has resulted in unexpected dermatological complications. The reported case of a young male developing generalized bullous fixed drug eruption after consuming liquid tadalafil and honey-based supplements highlights the clinical and public health implications of this issue.
Understanding Fixed Drug Eruption
Fixed drug eruption is defined by its recurrent nature: once a lesion has appeared at a particular site, it will reliably reappear upon re-exposure to the causative medication. This “cutaneous memory” is not merely poetic—it reflects the underlying immunological basis of the condition. In FDE, CD8+ resident memory T cells persist in the epidermis and dermis at the previously affected sites. Upon re-exposure to the drug or its metabolites, these primed lymphocytes rapidly induce keratinocyte apoptosis, vascular damage, and the classical violaceous patches observed clinically.
The timing of lesion onset is instructive. While the first exposure to a drug may trigger FDE weeks after ingestion, subsequent exposures provoke reactions within hours, often startling patients with the speed and severity of recurrence. Lesions may be solitary or generalized, ranging from isolated patches on mucosal lips or genitals to widespread involvement of trunk and extremities. Following the acute phase, post-inflammatory hyperpigmentation remains as a characteristic reminder of the episode.
A particularly severe manifestation is the bullous fixed drug eruption, in which blister formation complicates the clinical picture. The tense bullae and erosions mimic other vesiculobullous diseases, raising the risk of misdiagnosis. Distinguishing FDE from erythema multiforme or Stevens-Johnson syndrome is crucial, as therapeutic approaches and prognoses differ substantially. In this regard, detailed patient history—especially concerning prior drug exposures and supplement intake—emerges as an indispensable diagnostic tool.
The Case in Focus: Tadalafil as a Culprit
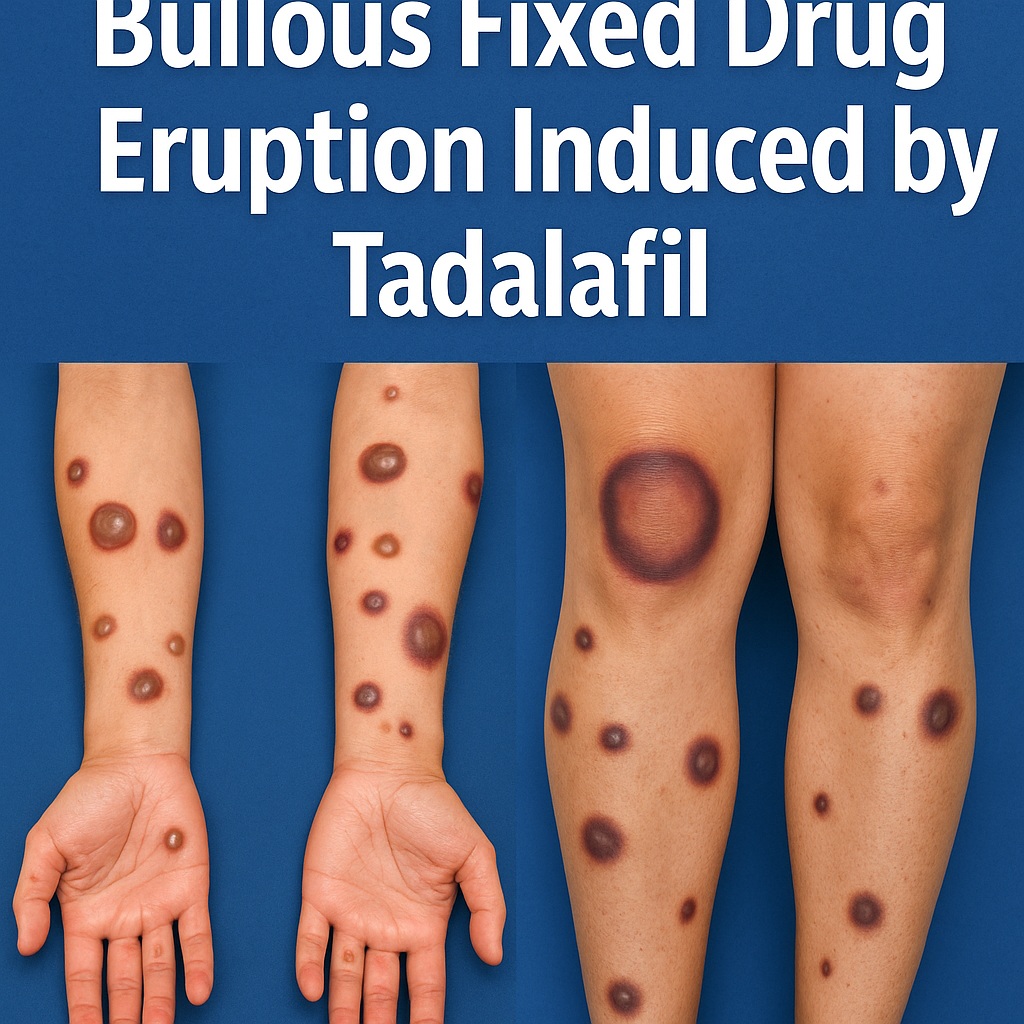
The published report describes a 29-year-old male who presented with painful bullae within an hour of ingesting liquid tadalafil obtained from a friend. His medical history revealed previous episodes of self-resolving lesions after consumption of honey-based sexual enhancement supplements, later determined to contain undeclared tadalafil. The clinical examination uncovered symmetric targetoid lesions with central bullae distributed across arms, legs, hands, feet, lips, and genitalia. Hyperpigmented patches persisted in previously affected areas.
Histopathological analysis revealed subepidermal bullae with vacuolar interface changes, necrosis, and rare eosinophils. Direct immunofluorescence demonstrated scattered cytoid bodies but ruled out other blistering conditions such as pemphigoid or collagen VII-related disorders. Laboratory investigations, including PCR for herpes simplex virus and ANA testing, were negative. These findings, combined with the clinical history and recurrent pattern, strongly supported a diagnosis of bullous fixed drug eruption induced by tadalafil.
Management included topical corticosteroids and prophylactic antiviral therapy in the differential diagnosis stage. Ultimately, the patient was counseled extensively on the need to avoid both prescription tadalafil and non-prescription supplements. Post-inflammatory hyperpigmentation persisted, but no new eruptions occurred after cessation of exposure.
Diagnostic Dilemmas: FDE Versus Other Blistering Disorders
The central challenge in diagnosing bullous FDE lies in its mimicry of other vesiculobullous diseases. Both erythema multiforme (EM) and bullous FDE can present with targetoid lesions, mucosal involvement, and acute blistering. Histologically, both conditions may demonstrate vacuolar interface dermatitis, necrosis, and apoptotic keratinocytes. Even for experienced dermatopathologists, the differentiation can be subtle.
Several features, however, tilt the balance toward FDE. Clinically, the recurrence of lesions at identical sites is strongly suggestive. Morphologically, the atypical oval targets and post-inflammatory pigmentation are more consistent with FDE than EM. Histologically, deeper inflammation with eosinophils and melanophages favors FDE, though no single pathological finding is definitive.
The diagnostic approach, therefore, must be multifactorial, integrating temporal relationships between drug exposure and lesion onset, anatomical distribution of lesions, recurrence pattern, histology, and exclusion of viral triggers. Failure to recognize FDE may lead to unnecessary treatments, prolonged morbidity, and recurrent exposures to the offending drug.
Tadalafil: From Therapy to Trigger
Tadalafil, marketed widely for erectile dysfunction, works by selectively inhibiting phosphodiesterase type 5 (PDE5), thereby enhancing cyclic GMP-mediated smooth muscle relaxation and increasing penile blood flow. Its safety profile in controlled clinical settings is well documented, with common adverse effects including headache, dyspepsia, flushing, and nasal congestion. Cutaneous hypersensitivity reactions are exceedingly rare in pre-marketing trials.
The growing problem, however, is the non-regulated distribution of tadalafil in over-the-counter supplements, often marketed as natural “sexual performance enhancers.” Investigations by the U.S. Food and Drug Administration (FDA) have revealed several honey-based products adulterated with undisclosed PDE5 inhibitors. Consumers ingest these supplements believing them to be natural, safe, and free of prescription drugs, only to face unexpected pharmacological exposures and adverse effects.
This dual pathway of exposure—through legitimate prescription and illicit supplements—complicates both diagnosis and prevention. The case presented demonstrates how patients may inadvertently consume tadalafil in multiple forms, making the link to FDE elusive unless clinicians specifically inquire about supplement use. In essence, the unsuspecting consumer becomes a case study in the hidden dangers of supplement adulteration.
Public Health and Regulatory Implications
The implications of this case extend beyond dermatology into the broader realm of public health policy and drug regulation. The supplement industry often operates under looser regulatory frameworks compared to pharmaceuticals. Products labeled as “natural” may contain potent pharmacological agents without disclosure, bypassing safety evaluations, dosage standardization, and mandatory labeling.
For the individual patient, this lack of transparency creates an environment of risk. Adverse reactions may be dismissed or misattributed, and repeated exposures perpetuate morbidity. For clinicians, the task of obtaining a comprehensive history becomes more complex, requiring specific inquiry into non-prescription supplements, imported products, and unregulated enhancers.
The FDA has issued multiple warnings against companies illegally marketing adulterated honey-based sexual enhancers, but enforcement remains challenging in the face of online distribution and international manufacturing. The case underscores the need for greater clinician awareness, patient education, and regulatory vigilance. Without these measures, cases of drug-induced eruptions triggered by hidden ingredients are destined to recur.
Management Strategies in Bullous Fixed Drug Eruption
Treatment of bullous FDE is guided by two principles: elimination of the causative agent and symptomatic management of lesions. The cornerstone is patient education regarding avoidance of the offending drug, which in this case includes both prescription tadalafil and unregulated supplements containing PDE5 inhibitors.
Topical corticosteroids are commonly prescribed to reduce inflammation and hasten lesion resolution. In more extensive cases, systemic corticosteroids may be considered, though evidence remains limited. Supportive measures, including wound care for erosions, emollients for barrier restoration, and antihistamines for pruritus, complement therapy.
Perhaps the most challenging aspect is patient adherence. Even after clear counseling, individuals may underestimate the dangers of re-exposure, especially when supplements are marketed as natural and harmless. Long-term follow-up is critical, both to monitor for recurrence and to reinforce the message of strict avoidance.
Conclusion
The case of bullous fixed drug eruption induced by tadalafil exemplifies the intersection of dermatology, pharmacology, and public health. While the clinical diagnosis hinges on meticulous evaluation of lesion morphology, histology, and patient history, the broader narrative reflects the dangers of unregulated supplement markets and the importance of regulatory oversight.
For clinicians, the lessons are clear: always consider fixed drug eruption in the differential diagnosis of bullous eruptions, specifically inquire about supplement use, and educate patients about the risks of non-prescription enhancers. For regulators, the mandate is equally pressing: enforce transparency, monitor adulterated products, and safeguard the public from hidden pharmacological threats.
In the end, this case serves as both a medical cautionary tale and a public health call to action. The skin, after all, does not lie—it faithfully records the biochemical indiscretions of our choices, sometimes in the form of bullae.
FAQ
1. What makes fixed drug eruption different from other drug-induced skin reactions?
FDE is unique in its recurrence at identical anatomical sites upon re-exposure to the drug. This characteristic pattern, along with post-inflammatory hyperpigmentation, helps distinguish it from other reactions like urticaria or erythema multiforme.
2. Why is tadalafil implicated in fixed drug eruptions if it is generally considered safe?
While tadalafil is safe in regulated prescription use, unregulated supplements containing undisclosed tadalafil expose individuals to unpredictable doses and repeated exposures. These hidden exposures increase the risk of hypersensitivity reactions such as FDE.
3. How can patients protect themselves from adverse reactions linked to supplements?
Patients should avoid unregulated over-the-counter sexual enhancement products, especially those marketed as “herbal” or “natural.” Consulting healthcare providers before using any supplement and relying on regulated prescriptions significantly reduces the risk of hidden drug exposures and associated complications.