Introduction
Disorders of the lower urinary tract (LUT) such as overactive bladder and detrusor overactivity are often discussed in the context of neural dysregulation, detrusor muscle dysfunction, or obstruction secondary to prostatic enlargement. Yet, beneath these familiar culprits lies a subtler and increasingly recognized factor: ischemia of the bladder vasculature. The bladder, like any organ, is only as resilient as its blood supply, and emerging data suggest that reduced perfusion plays a central role in the pathophysiology of LUT dysfunction.
Interestingly, pharmacological interventions originally designed for erectile dysfunction have been shown to improve urinary symptoms. Phosphodiesterase-5 inhibitors (PDE5i) such as tadalafil and sildenafil are now recognized not only for their role in sexual medicine but also for their potential to improve perfusion and modulate bladder vascular tone. How these drugs achieve such effects, however, has remained largely obscure.
The study at the heart of this discussion systematically dissects the vascular physiology of the superior vesical artery—the primary vessel supplying the bladder—using isolated porcine tissue. By employing electrical field stimulation (EFS), neurotransmitter blockade, nitric oxide (NO) donor assays, and selective pharmacological inhibitors, the investigators mapped the balance between vasoconstrictor and vasodilator influences. More importantly, they demonstrated how tadalafil and sildenafil potentiate nitrergic nerve-mediated relaxations, positioning these drugs as key modulators of bladder blood flow.
This article will explore the context, findings, and implications of this work, while also considering how such vascular pharmacology might translate into improved therapeutic strategies for patients suffering from LUT dysfunction.
Vascular Perfusion and the Lower Urinary Tract: Why Blood Flow Matters
The traditional explanation for LUT disorders—whether in men or women—often revolves around muscular dysfunction or hormonal changes. In men, benign prostatic hyperplasia (BPH) increases outlet resistance, whereas in women, postmenopausal changes alter detrusor and urothelial physiology. However, there is mounting evidence that ischemic injury to the bladder wall may be the unifying pathophysiological denominator.
Clinical and experimental studies have shown that:
- Bladder ischemia correlates with urinary frequency and nocturia, particularly in patients with vascular comorbidities.
- Animal models of chronic ischemia demonstrate detrusor hyperactivity, collagen deposition, oxidative stress, and heightened inflammatory cytokine activity.
- In both sexes, systemic vascular disease parallels LUT symptoms, suggesting that microvascular compromise is a critical shared risk factor.
Therefore, interventions that restore or potentiate bladder perfusion are not merely adjunctive but may address the very foundation of LUT dysfunction.
The Superior Vesical Artery: A Pharmacological Case Study
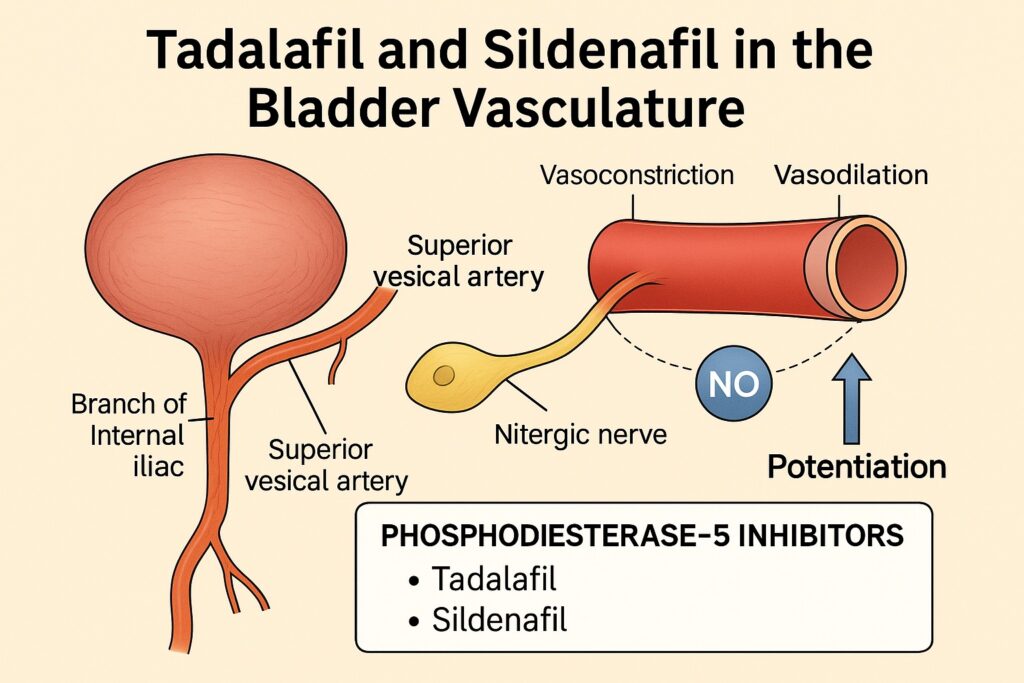
The superior vesical artery, derived from the umbilical artery, is the principal supplier of blood to the bladder dome and body. It represents the anatomical and functional bottleneck through which perfusion is determined. Understanding its regulation is therefore essential for designing targeted therapies.
In the study, isolated porcine superior vesical artery rings were subjected to electrical field stimulation to mimic perivascular nerve activation. A biphasic response was consistently observed:
- Rapid vasoconstriction—mediated by release of ATP (≈55%) and noradrenaline (≈45%).
- Slower vasodilation—dominated by neuronal nitric oxide (nNO) signaling and, to a smaller extent, β-adrenoceptor activation.
Interestingly, endothelial removal did not abolish vasodilation, underscoring that nNOS-derived NO from perivascular nerves rather than endothelial nitric oxide synthase (eNOS) is the primary mediator of vascular relaxation in this setting.
This finding is more than an anatomical curiosity. It demonstrates that bladder perfusion is regulated by neurogenic NO release, which provides an obvious pharmacological target for PDE5 inhibitors.
Nitrergic Neurotransmission and the Role of Nitric Oxide
Nitric oxide (NO) remains the linchpin of vascular relaxation in nearly every organ system. In the bladder vasculature, the story is more nuanced:
- Noradrenaline, usually the star player in vasoconstriction, here contributes modestly compared to ATP.
- nNOS-derived NO is the predominant vasodilatory mediator, responsible for over 80% of the observed relaxation.
- The endothelium, surprisingly, plays only a minor role in EFS-induced vasodilation, which is largely neuronal in origin.
Thus, the superior vesical artery functions under a model where ATP and noradrenaline initiate constriction, while nitrergic nerves counterbalance with relaxation. The balance between these forces dictates perfusion, and disruption of nitrergic signaling could theoretically predispose to ischemia and LUT dysfunction.
This neurovascular arrangement presents an elegant but fragile equilibrium. Enhancing the nitrergic side of the equation without completely abolishing sympathetic tone is where PDE5 inhibitors appear to fit in seamlessly.
Tadalafil and Sildenafil: Mechanisms of Action in the Bladder Vasculature
Both tadalafil and sildenafil act as selective PDE5 inhibitors, preventing the breakdown of cyclic guanosine monophosphate (cGMP). Since cGMP is the second messenger of NO signaling, inhibiting PDE5 essentially amplifies the vasodilatory signal without directly releasing NO.
In the porcine superior vesical artery:
- Both drugs depressed noradrenaline-evoked vasoconstriction (by ≈27% with tadalafil and ≈36% with sildenafil).
- Both potentiated vasodilation induced by exogenous NO donors (sodium nitroprusside, SIN-1, SNAP), shifting dose-response curves leftward and increasing sensitivity without altering maximal relaxation.
- Both enhanced nerve-evoked vasodilation, with sildenafil showing a greater effect (≈52%) compared to tadalafil (≈28%).
These results highlight that PDE5 inhibition works not by inventing new pathways, but by tilting the balance in favor of nitrergic relaxation. The implication is profound: if chronic ischemia drives LUT symptoms, then amplifying nitrergic vasodilation offers a rational, mechanism-based therapy.
Clinical Implications: From Erectile Dysfunction to Urology Clinics
Tadalafil and sildenafil have long since proven their worth in erectile dysfunction, where penile cavernosal perfusion depends on NO-cGMP signaling. Their introduction into LUT therapeutics was initially serendipitous, with men reporting improved urinary symptoms during treatment for erectile dysfunction.
Subsequent trials demonstrated:
- Improved International Prostate Symptom Scores (IPSS) in men with BPH-related LUT symptoms.
- Reduced nocturia and urgency episodes in overactive bladder cohorts.
- Enhanced endothelial function in patients with vascular risk factors.
The mechanistic insight from the porcine bladder vasculature now provides a physiological explanation: these drugs improve LUT symptoms by potentiating nitrergic vasodilation of the superior vesical artery, thereby improving bladder perfusion.
The story, however, is not entirely without caution. Extrapolating porcine data to humans requires consideration of sex differences, hormonal influences, and the altered vascular milieu in patients with established disease. Still, the physiological plausibility makes PDE5 inhibitors attractive candidates for repurposing in LUT disorders, particularly those with an ischemic basis.
Limitations and Future Directions
Every study illuminates a piece of the puzzle while leaving shadows elsewhere. This work, though rigorous, also invites further inquiry:
- Species differences: Porcine models approximate human physiology well but are not identical, particularly in sex-specific vascular regulation.
- Normal versus diseased tissue: The experiments used healthy arteries; in patients with LUT dysfunction, vascular remodeling, fibrosis, and endothelial dysfunction may alter drug responsiveness.
- Drug selectivity: Tadalafil and sildenafil differ in their secondary targets (PDE11 vs PDE6, respectively), which could subtly influence bladder vascular responses.
- Clinical endpoints: Biochemical potentiation of NO is compelling, but real-world outcomes must ultimately be measured in symptom relief, quality of life, and long-term safety.
Future research should explore whether daily low-dose PDE5 inhibitors can serve not just as symptomatic therapy but as disease-modifying agents that reverse or prevent ischemic remodeling in the bladder wall.
Conclusion
The superior vesical artery may not be a household name in medicine, but it represents the frontline of bladder health. Its perfusion determines whether detrusor muscle and urothelium thrive or suffocate under ischemic stress.
This study demonstrates that tadalafil and sildenafil, by potentiating nitrergic nerve-mediated relaxations, provide a pharmacological lever to tilt the balance toward improved perfusion. In doing so, they not only validate the vascular hypothesis of LUT dysfunction but also carve a new niche for PDE5 inhibitors in urology.
What began as a revolution in sexual medicine may evolve into a cornerstone of vascular urology, reminding us that sometimes the most effective therapies come from repurposing rather than reinventing.
FAQ
1. How do PDE5 inhibitors improve bladder function if they were designed for erectile dysfunction?
They amplify nitric oxide signaling by preventing the breakdown of cGMP, thereby enhancing nitrergic nerve-mediated vasodilation in the bladder vasculature. This improves bladder perfusion and helps alleviate LUT symptoms.
2. Are tadalafil and sildenafil equally effective for urinary symptoms?
Both show significant benefits, but subtle differences exist. Sildenafil enhanced nerve-mediated vasodilation more strongly in the study, while tadalafil’s longer half-life may provide steadier therapeutic coverage. Clinical choice often depends on patient profile and comorbidities.
3. Could PDE5 inhibitors become a standard therapy for overactive bladder or LUT dysfunction?
They already show clinical efficacy in selected populations, especially men with BPH. Ongoing research is needed to validate their role in women, in non-obstructive LUT dysfunction, and as long-term disease-modifying therapies targeting bladder ischemia.