Introduction
Liver cancer remains one of the most unforgiving malignancies in modern medicine. Despite decades of progress in oncology, hepatocellular carcinoma continues to claim lives with alarming efficiency, particularly in low- and middle-income countries where access to advanced therapies is limited. The clinical challenge is not merely biological aggressiveness, but also economics, pharmacokinetics, and the harsh realities of drug toxicity. In this context, innovation must be pragmatic, affordable, and biologically intelligent rather than extravagantly complex.
An emerging strategy that has gained serious scientific traction is drug repurposing, where approved non-oncology drugs are re-engineered to exert anticancer effects. This approach avoids the time, cost, and regulatory burden of developing new molecular entities. Yet, repurposing alone is not enough. Many promising drugs fail in oncology due to poor solubility, rapid metabolism, low tumor penetration, or systemic toxicity when administered using conventional delivery systems.
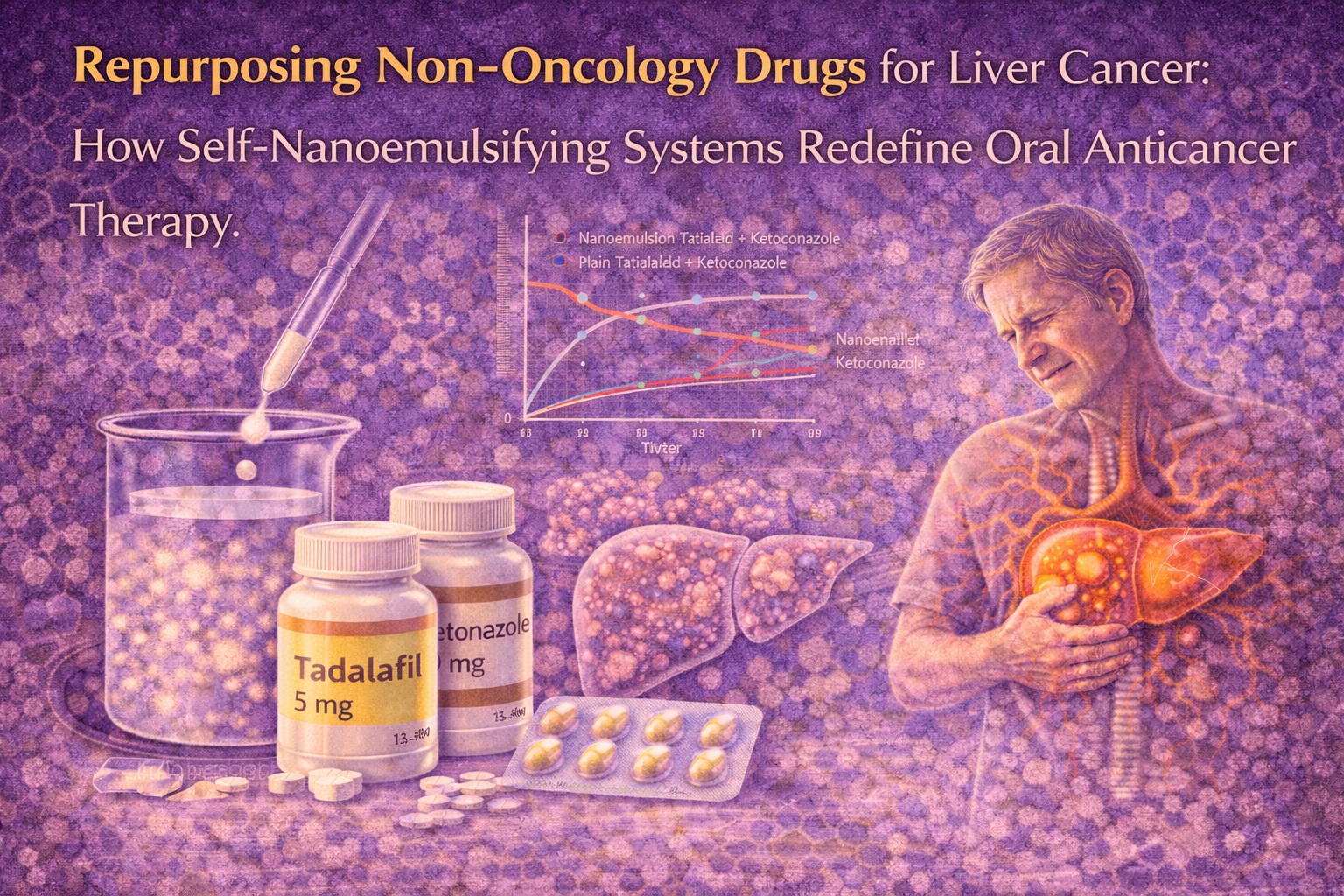
This is where self-nanoemulsifying drug delivery systems (SNEDDS) enter the conversation—not as a technological novelty, but as a clinically relevant solution. By redesigning how drugs are delivered rather than what drugs are delivered, SNEDDS bridge pharmacology and nanotechnology in a way that is both elegant and practical. The study at the center of this discussion demonstrates how two well-known non-oncology agents—tadalafil and ketoconazole—can be transformed into a viable anticancer strategy through nanoemulsion-based oral delivery .
Liver Cancer and the Limitations of Conventional Chemotherapy
The burden of hepatic malignancies is disproportionately heavy in regions with limited healthcare resources. Conventional chemotherapy remains the backbone of treatment in many settings, yet it is plagued by fundamental pharmacological problems. Poor aqueous solubility, unpredictable absorption, and non-selective cytotoxicity often result in narrow therapeutic windows and unacceptable adverse effects.
Moreover, oral anticancer therapy—a long-sought goal for patient-friendly treatment—has repeatedly failed to deliver consistent outcomes. Drugs that look promising in vitro frequently disappoint in vivo because the gastrointestinal tract is an unforgiving environment. Enzymatic degradation, first-pass metabolism, and erratic absorption conspire to reduce systemic drug availability long before tumor cells are exposed.
Even when effective agents are identified, cost becomes a decisive barrier. Advanced biologics and targeted therapies remain financially inaccessible for a significant proportion of patients worldwide. As a result, there is a growing recognition that affordability is not a secondary outcome—it is a core therapeutic parameter. Any realistic solution for hepatic cancer must align scientific rigor with economic feasibility.
Drug Repurposing as a Strategic Anticancer Approach
Drug repurposing offers a rare convergence of speed, safety, and cost-effectiveness. Approved drugs come with well-characterized pharmacokinetic and safety profiles, allowing researchers to focus on efficacy rather than rediscovering toxicity thresholds. This strategy is particularly attractive in oncology, where the timeline from discovery to patient access can span decades.
Phosphodiesterase-5 inhibitors, traditionally used in cardiovascular and urological indications, have demonstrated unexpected anticancer properties. Tadalafil, in particular, modulates intracellular signaling pathways involved in tumor growth and immune evasion. However, its clinical potential in cancer has been limited by extensive hepatic metabolism mediated by CYP3A4 enzymes.
Ketoconazole, widely known as an antifungal agent, introduces an interesting pharmacological counterbalance. Beyond its own anticancer activity, it is a potent CYP3A4 inhibitor. When combined with tadalafil, it reduces metabolic degradation, effectively increasing intratumoral drug exposure. This dual-drug strategy is pharmacologically sound, but only if both agents can be delivered efficiently and safely—an obstacle that conventional formulations fail to overcome .
Why Self-Nanoemulsifying Drug Delivery Systems Matter
SNEDDS represent a fundamental shift in oral drug formulation. Instead of forcing poorly soluble drugs to dissolve in gastrointestinal fluids, SNEDDS exploit spontaneous nanoemulsion formation upon contact with aqueous media. The result is a fine dispersion of nanometer-sized droplets that dramatically increase the apparent solubility and absorption of lipophilic compounds.
From a clinical perspective, this translates into several meaningful advantages. First, enhanced absorption leads to more predictable plasma concentrations. Second, smaller droplet sizes facilitate improved cellular uptake and, potentially, enhanced tumor accumulation via the enhanced permeation and retention effect. Third, controlled release profiles reduce peak-related toxicity while maintaining sustained therapeutic exposure.
Equally important is manufacturability. SNEDDS are comparatively simple to produce and scale, requiring no exotic materials or prohibitively complex processes. This aligns perfectly with the goal of developing therapies suitable for widespread global use, not just elite research centers.
Engineering a Stable Nanoemulsion for Anticancer Repurposing
The formulation described in the study was not an exercise in trial-and-error. A systematic design approach was employed to optimize surfactant concentration, lipid content, and sonication parameters. The result was a stable nanoemulsion with a mean particle size of approximately 40 nanometers—a size range that balances stability with biological interaction.
Stability, often overlooked in academic discussions, is critical for real-world applicability. The nanoemulsion demonstrated excellent physical stability over extended storage periods without phase separation or creaming. This is not merely a technical success; it is a prerequisite for clinical translation and commercial viability.
The formulation also achieved high drug loading efficiency for both tadalafil and ketoconazole. This ensured that therapeutic concentrations could be delivered without excessive excipient burden—a subtle but important factor when considering long-term administration.
Controlled Drug Release and Blood Compatibility
One of the more compelling aspects of the nanoemulsion system is its controlled release profile. Unlike plain drug solutions that exhibit rapid and uncontrolled dissolution, the nanoemulsion released both drugs gradually over 24 hours. This sustained release pattern aligns well with oncological treatment paradigms, where prolonged exposure often yields better therapeutic outcomes than sharp concentration spikes.
Blood compatibility is another critical consideration, particularly for formulations that may eventually be administered intravenously or reach systemic circulation in significant amounts. Hemolysis studies demonstrated only moderate red blood cell disruption, comparable to that observed with free drug solutions. This finding suggests that the nanoemulsion does not introduce disproportionate hematological risk—a key safety signal in early formulation development .
Enhanced Cytotoxicity and Cellular Uptake
In vitro studies using HepG2 liver cancer cells revealed a striking enhancement in cytotoxicity when drugs were delivered via the nanoemulsion. The combination formulation achieved significantly lower IC50 values compared to either drug alone, indicating a synergistic or at least additive anticancer effect.
Equally important was the observed increase in cellular uptake. The nanoemulsion facilitated several-fold higher intracellular concentrations of both drugs compared to plain solutions. This phenomenon likely reflects improved membrane interaction mediated by lipid-based nanocarriers, reinforcing the argument that delivery systems can be as influential as pharmacological targets.
From a mechanistic standpoint, the inhibition of CYP3A4-mediated metabolism by ketoconazole likely played a role in sustaining intracellular tadalafil levels. Together, these effects create a pharmacological environment that favors tumor cell apoptosis while sparing normal tissues—a long-standing goal in oncology.
Clinical and Translational Implications
While in vitro success does not guarantee clinical efficacy, the translational potential of this approach is difficult to ignore. The strategy combines three elements rarely aligned in cancer therapy: affordability, biological plausibility, and formulation simplicity. For healthcare systems under economic pressure, such combinations are not luxuries—they are necessities.
Oral nanoemulsion-based therapy also carries profound implications for patient quality of life. Reduced hospital visits, simplified dosing regimens, and improved tolerability can significantly enhance treatment adherence. In oncology, where therapeutic fatigue is a genuine concern, these factors matter as much as tumor response rates.
Future investigations must focus on in vivo validation, pharmacokinetic profiling, and long-term safety assessment. However, the foundational work already demonstrates that repurposed drugs, when paired with intelligent delivery systems, can challenge conventional assumptions about anticancer therapy.
Conclusion
The development of a stable self-nanoemulsifying system for delivering repurposed non-oncology drugs represents more than a formulation success—it represents a philosophical shift in cancer treatment design. Instead of chasing ever more complex molecules, this approach optimizes what is already known, trusted, and accessible.
By combining tadalafil and ketoconazole within a nanoemulsion framework, researchers have demonstrated a credible pathway toward affordable, effective oral therapy for hepatic cancer. The implications extend beyond liver malignancies, opening doors to similar strategies across oncology and other therapeutic domains.
Innovation, it turns out, does not always require reinvention. Sometimes, it simply requires delivering old ideas in smarter ways.
FAQ
Can non-oncology drugs really be effective against cancer?
Yes. Many approved drugs influence cellular pathways relevant to cancer biology. When combined with appropriate delivery systems, their therapeutic potential can be significantly enhanced.
Why is nanoemulsion-based delivery superior to conventional oral formulations?
Nanoemulsions improve solubility, absorption, cellular uptake, and release control, overcoming key limitations of traditional oral drug delivery.
Is this approach ready for clinical use?
Not yet. While the preclinical data are promising, in vivo studies and clinical trials are required before translation into routine medical practice.