Introduction: When the First Repair Fails
Posterior urethral distraction injuries represent one of the most challenging scenarios in reconstructive urology. These injuries, typically associated with severe pelvic trauma, disrupt not only urethral continuity but also the delicate balance of continence, erectile function, and pelvic anatomy. While primary posterior urethroplasty has evolved into a highly successful procedure in experienced hands, failure still occurs—and when it does, the consequences are profound.
A failed bulbomembranous anastomosis is not simply a technical setback. It transforms a difficult operation into a complex salvage situation, often characterized by distorted anatomy, dense fibrosis, compromised vascular supply, and heightened risk to continence mechanisms. In this setting, repeating the same surgical strategy is rarely sufficient.
The concept of intrapelvic bulboprostatic repair emerges precisely at this intersection of failure and opportunity. Rather than abandoning function-preserving goals in favor of urinary diversion or palliative measures, this approach reasserts the possibility of anatomical and functional restoration—even after previous reconstruction has failed. The case under discussion provides a framework to explore how meticulous anatomical understanding, surgical humility, and strategic innovation can converge to reclaim outcomes once thought lost.
Posterior Urethral Distraction Injury: More Than a Broken Tube
Posterior urethral distraction injuries are fundamentally three-dimensional problems. Pelvic fracture forces displace the prostate, rupture the membranous urethra, and shear the surrounding sphincteric and neurovascular structures. The resulting defect is rarely linear; it is spatial, rotational, and often compounded by associated injuries to the bladder neck and pelvic floor.
Initial management priorities focus on avoiding further damage. Attempted transurethral catheterization in the acute phase risks converting a contained injury into a contaminated one. Suprapubic urinary diversion remains the safest initial strategy, allowing inflammation to subside and pelvic anatomy to stabilize before definitive reconstruction.
Definitive repair traditionally involves delayed end-to-end anastomotic urethroplasty. When performed correctly, success rates are high. However, this success depends on precise identification of the prostatic apex, tension-free alignment, and preservation of surrounding structures. Failure at this stage alters the biological and surgical landscape dramatically.
Why Posterior Urethroplasty Fails
Failure of posterior urethroplasty is rarely due to a single factor. More often, it reflects a cascade of small misjudgments compounded by challenging anatomy. Inadequate exposure, misidentification of the proximal urethral stump, or anastomosis to non-urethral tissue can result in immediate obstruction or early restenosis.
Scar formation following failed repair is particularly unforgiving. Fibrosis obliterates tissue planes, obscures anatomical landmarks, and compromises vascular supply. Each subsequent intervention increases the risk of sphincter damage and erectile dysfunction, making the surgeon’s margin for error progressively narrower.
Perhaps most importantly, failed repair erodes confidence—both patient and surgeon alike. This psychological dimension should not be underestimated, as it often drives premature acceptance of suboptimal outcomes rather than pursuit of technically demanding salvage options.
The Intrapelvic Bulboprostatic Concept: Rethinking the Target
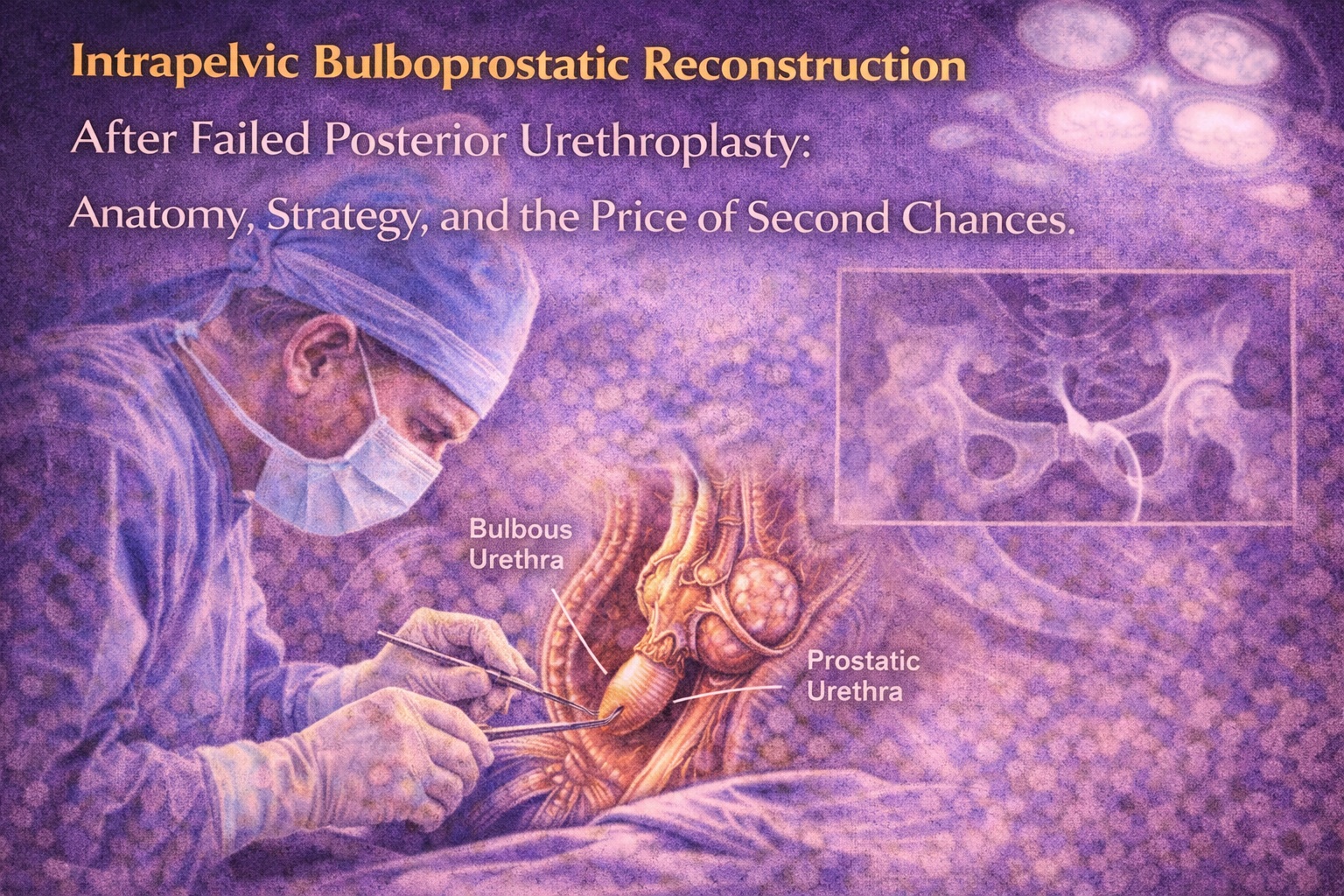
Traditional posterior urethroplasty focuses on reestablishing continuity between the bulbous and membranous urethra. In cases where the membranous segment is obliterated or inaccessible, this paradigm collapses. Intrapelvic bulboprostatic repair reframes the objective: instead of searching for a compromised membranous stump, the surgeon directly anastomoses the mobilized bulbous urethra to the prostatic urethra within the pelvis.
This strategy borrows conceptual elements from vesicourethral anastomosis after radical prostatectomy. The key difference lies in preserving the rhabdosphincter by intrasphincteric dissection, allowing the urethra to traverse the sphincter without destroying its functional integrity.
The result is not merely an anatomical shortcut, but a functional compromise designed to preserve continence while restoring urethral patency. It is an approach that accepts complexity rather than fighting it.
Intrasphincteric Dissection: Respecting the Last Line of Continence
The rhabdosphincter is the final guardian of urinary continence in posterior urethral reconstruction. Damage to this structure often condemns patients to lifelong incontinence, regardless of anastomotic success. Intrasphincteric dissection exploits a natural fascial plane within the sphincter, allowing safe passage of the urethra while preserving muscle fibers.
This maneuver demands anatomical precision and patience. The dissection plane is narrow, unforgiving, and invisible to the hurried hand. Yet when executed correctly, it allows the bulbous urethra to be advanced into the small pelvis without compromising sphincteric function.
The case demonstrates that continence preservation is not an all-or-nothing proposition. With careful technique, functional outcomes can be salvaged even in revision settings that historically favored urinary diversion.
Combined Perineal and Suprapubic Access: Owning the Anatomy
Revision posterior urethroplasty rarely succeeds through a single incision. Combined perineal and suprapubic access provides the necessary exposure to address both distal mobilization and proximal identification of the prostatic urethra.
The suprapubic approach allows direct visualization of the bladder outlet and prostatic apex, eliminating guesswork. Endoscopic confirmation further ensures that the anastomosis targets true urethral tissue rather than scar or prostate capsule—an error that doomed the initial repair in the reported case.
This dual approach reflects a broader surgical philosophy: when anatomy is distorted, exposure is not a luxury but a necessity. Limited access may reduce operative time, but it increases the risk of irreversible mistakes.
Tension-Free Anastomosis: The Currency of Success
No principle in urethral reconstruction is more unforgiving than tension-free anastomosis. In revision cases, achieving this goal requires aggressive—but thoughtful—mobilization of the bulbous urethra, excision of all fibrotic tissue, and strategic routing through the pelvic floor.
Dissection of the proximal corpora cavernosa in the midline provides the additional length required for intrapelvic pull-through. This maneuver must be executed with extreme care to avoid injury to the dorsal neurovascular bundle, whose preservation is essential for erectile function.
The resulting anastomosis, performed intrapelvically, mirrors techniques familiar from radical prostatectomy, reinforcing the value of cross-disciplinary surgical literacy in complex reconstructions.
Functional Outcomes: Redefining “Success” After Failure
Success in salvage urethral reconstruction must be defined realistically. Absolute normality is rarely achievable; meaningful improvement is the true benchmark. In the presented case, the patient achieved acceptable continence using a single safety pad per day—an outcome that would have been dismissed as optimistic at the outset.
Erectile function, often sacrificed in salvage scenarios, was partially preserved. While erections sufficient for penetration were not achieved, the presence of orgasmic function and erectile response represents a significant quality-of-life victory in this context.
Adjunctive low-dose tadalafil therapy, although not definitively proven in post-urethroplasty settings, reflects a proactive approach to penile rehabilitation grounded in emerging experimental evidence.
The Role of Experience and Case Selection
It must be emphasized that intrapelvic bulboprostatic repair is not a universal solution. It is a technically demanding procedure best reserved for high-volume reconstructive centers with extensive experience in posterior urethral surgery.
Patient selection is equally critical. Candidates must have intact bladder neck function, preserved sphincter anatomy, and sufficient urethral length for mobilization. Without these prerequisites, the risk–benefit balance shifts unfavorably.
When applied judiciously, however, this approach expands the therapeutic horizon for patients otherwise relegated to permanent diversion.
Ethical and Psychological Dimensions of Salvage Surgery
Salvage reconstruction occupies a unique ethical space. Patients must be counseled honestly about risks, limitations, and realistic expectations. Overpromising functional outcomes is as harmful as denying the possibility of improvement.
At the same time, offering revision surgery acknowledges patient dignity and the value of urinary and sexual function beyond mere survival. This balance between surgical ambition and ethical restraint defines excellence in reconstructive urology.
Conclusion
Intrapelvic bulboprostatic repair after failed posterior urethroplasty exemplifies the evolution of reconstructive surgery from rigid algorithms to adaptive problem-solving. By combining deep anatomical knowledge with surgical creativity, it reclaims function in situations once considered irreparable.
Failure, in this context, is not an endpoint but a diagnostic tool—revealing what must be done differently the second time. For selected patients, this approach transforms surgical salvage into meaningful recovery.
FAQ
Is revision urethroplasty possible after a failed posterior repair?
Yes. In selected patients, revision surgery using intrapelvic bulboprostatic anastomosis can restore urethral patency and preserve continence.
Does intrasphincteric dissection increase the risk of incontinence?
When performed correctly, it actually preserves sphincter function by respecting internal fascial planes.
What level of expertise is required for this procedure?
This surgery should be performed only in specialized reconstructive centers due to its technical complexity and anatomical risks.