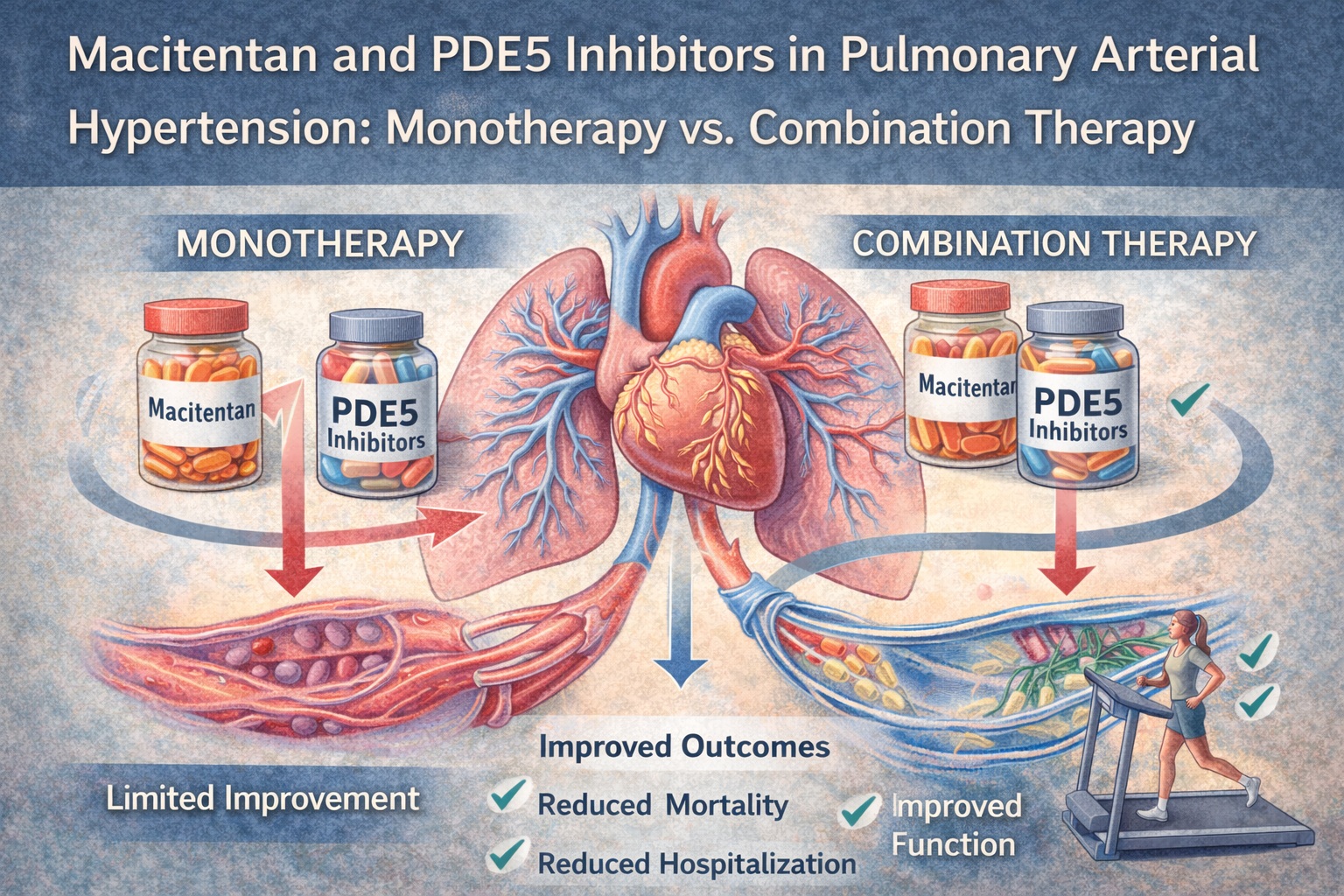
Pulmonary arterial hypertension (PAH) remains one of the most challenging chronic cardiovascular disorders to manage in modern medicine. Its progression is insidious, its clinical course unforgiving, and its therapeutic demands increasingly complex. Despite the considerable evolution in treatment strategies over the past two decades, clinicians continue to debate the optimal timing and composition of initial therapy—particularly whether monotherapy remains viable or whether combination therapy should be the default approach for newly diagnosed patients.
The pooled analysis underlying this article synthesizes available evidence on macitentan, a dual endothelin receptor antagonist (ERA), and phosphodiesterase-5 inhibitors (PDE5i) such as sildenafil or tadalafil. While both drug classes have demonstrated meaningful improvements in hemodynamic parameters and clinical outcomes, their combined use may offer synergistic benefits that monotherapy alone cannot consistently provide. The analysis integrates multiple datasets, compares trajectories of clinical worsening, evaluates hospitalization risk, and explores functional capacity across therapeutic strategies.
This article reframes the core insights of that analysis into a structured, readable narrative aimed at clinicians and researchers who must make evidence-based decisions in the face of PAH’s biological complexity. The data, when interpreted through a mechanistic lens, support a paradigm shift toward early combination therapy—particularly macitentan with a PDE5 inhibitor—as a rational, biologically coherent, and prognostically advantageous strategy.
The Pathophysiological Logic Behind Combination Therapy
The central argument for combination therapy emerges from a simple yet powerful physiological principle: PAH is a multidimensional disease, driven not by a single dysregulated pathway but by a network of dysfunctional signaling systems involving vasoconstriction, inflammation, endothelial impairment, smooth muscle proliferation, and remodeling of the distal pulmonary arteries. Thus, targeting only one pathway is akin to trying to slow a runaway train by applying a single brake when several are failing simultaneously.
Macitentan antagonizes both ETA and ETB receptors, blocking endothelin-1-mediated vasoconstriction and cell proliferation. PDE5 inhibitors, by contrast, enhance nitric oxide-mediated vasodilation and reduce right ventricular afterload through elevation of intracellular cGMP. Their combined action therefore counterbalances two of the most potent and mutually reinforcing drivers of PAH: excessive vasoconstriction and inadequate vasodilation. Mechanistically, this approach harmonizes with what molecular biology teaches us about vascular remodeling: it is easier to slow pathological proliferation when vasoconstrictive tone is controlled, and easier to enhance vasodilation when cGMP signaling is unopposed by endothelin.
The pooled analysis makes this synergy palpable. Patients receiving the combination exhibited a sharper and earlier reduction in pulmonary vascular resistance and a more pronounced improvement in 6-minute walk distance (6MWD) than those on monotherapy. Importantly, the combination also produced fewer early clinical worsening events, signaling not only symptomatic benefit but also genuine modification of disease trajectory.
From a translational standpoint, these findings place combination therapy exactly where it belongs: not as an escalation tool reserved for therapeutic failure but as a strategic starting point for disease interception.
Macitentan Monotherapy: Effective, But Often Insufficient
Macitentan’s approval represented a significant advancement in endothelin receptor blockade, offering improved receptor affinity, superior tissue penetration, and more sustained pathway inhibition compared with earlier ERAs. Its benefits in reducing morbidity and delaying clinical worsening are well documented, making it a cornerstone in PAH guidelines.
Yet the pooled analysis reveals several limitations when macitentan is used alone. Although many patients experienced symptomatic improvement, a substantial subset demonstrated incomplete response—manifesting as persistent dyspnea, inadequate improvement in exercise capacity, or progression to hospitalization within the first year. This heterogeneity in response highlights a biological truth: endothelin blockade alone cannot fully reverse or stabilize the complex remodeling processes characteristic of PAH.
Macitentan monotherapy does provide clear value in patients who cannot tolerate PDE5 inhibitors or who present with milder disease. However, even in these contexts, the analysis suggests that monotherapy performs best as a bridge toward combination therapy rather than as a long-term stand-alone approach. This echoes broader PAH literature, which increasingly favors layering therapies early for maximal disease control.
Thus, while macitentan remains indispensable, the pooled data remind us that it should rarely be used in isolation in newly diagnosed patients unless there are compelling clinical contraindications.
PDE5 Inhibitors Alone: Helpful, But Vulnerable to Biological Escape Mechanisms
Sildenafil and tadalafil, the most widely used PDE5 inhibitors, have transformed PAH management since their introduction. By enhancing cGMP signaling and potentiating nitric oxide-mediated vasodilation, they reduce pulmonary vascular resistance, improve quality of life, and enhance right ventricular efficiency. Their effects on exercise capacity are especially robust, making them a popular initial choice.
However, like macitentan, PDE5 inhibitors demonstrate variable effectiveness when used alone. The pooled analysis indicates that while many patients show early improvement, the durability of this response is inconsistent. Over time, endothelial dysfunction, reduced NO bioavailability, and increased oxidative stress undermine the physiological pathways that PDE5 inhibitors rely upon. As a result, their vasodilatory effect wanes, and disease progression resumes.
The analysis also notes that patients on PDE5 monotherapy had a higher risk of hospitalization and clinical worsening compared with those on macitentan monotherapy or combination therapy. This highlights a crucial limitation: PDE5 inhibitors do not address proliferative pathways, smooth muscle hyperplasia, or endothelin-driven fibrosis. Without counteracting these processes, vasodilatory therapy alone becomes a temporary reprieve rather than a long-term strategy.
In essence, PDE5 inhibitors remain valuable but incomplete: they improve symptoms but do not sufficiently intercept disease mechanisms.
Combination Therapy: A Biologically Synergistic and Clinically Superior Approach
The strongest and most clinically relevant conclusion of the pooled analysis is that initial macitentan–PDE5i combination therapy outperforms either monotherapy across virtually all major clinical domains. Whether examining exercise capacity, hemodynamic parameters, rate of hospitalization, or incidence of clinical worsening, combination therapy repeatedly demonstrates superiority.
One of the most striking findings is the reduction in early clinical worsening events. Newly diagnosed patients on combination therapy showed significantly fewer episodes of rapid decompensation, reflecting not merely improved vasodilation but enhanced vascular stability. These benefits appeared early—often within the first months of treatment—suggesting that early pathway suppression is crucial before remodeling becomes irreversible.
The improvement in 6-minute walk distance was also notably greater in the combination cohort. This metric, although simple, remains one of the most powerful predictors of long-term survival in PAH. Its responsiveness to therapy reflects not only enhanced pulmonary blood flow but also improved right ventricular output—a critical determinant of overall prognosis.
Furthermore, combination therapy patients demonstrated superior WHO functional class stabilization. This indicates better symptom control, reduced dyspnea, and improved day-to-day functioning—outcomes that matter deeply to patients and caregivers.
The pooled data collectively support a shift toward early, aggressive therapy—one that targets multiple pathways simultaneously rather than sequentially.
Why Combination Therapy Works: Mechanistic Precision and Prevention of Pathway Drift
The physiological synergy between macitentan and PDE5 inhibitors is more than additive; it is corrective. Each drug counteracts the compensatory mechanisms that weaken the effect of the other. Endothelin-mediated vasoconstriction and smooth muscle proliferation are weakened by macitentan, allowing PDE5i-mediated vasodilation to be more effective. Meanwhile, enhanced NO-cGMP signaling reduces vasoconstrictive tone and mitigates right ventricular strain, making endothelin receptor blockade more efficient.
This combination also limits “pathway drift,” a phenomenon where single-pathway inhibition leads the disease to reroute its progression through alternate mechanisms. By targeting both the endothelin and NO–cGMP pathways from the outset, combination therapy closes off multiple escape routes simultaneously.
The pooled analysis also suggests that combination therapy may reduce biomarker surrogates of endothelial injury—such as NT-proBNP—more effectively than monotherapy. As NT-proBNP closely tracks right ventricular stress, this finding reinforces the notion that combination therapy confers physiologic resilience rather than merely symptomatic relief.
Thus, combination therapy is not simply two drugs administered together—it is an integrated strategy that restores balance to a system overwhelmed by pathological signaling.
Safety and Tolerability: A Balanced Perspective
The pooled analysis confirms that macitentan–PDE5i combination therapy is generally well tolerated. Adverse events such as edema, headache, or nasal congestion occurred at expected rates and rarely resulted in discontinuation. Liver toxicity, a historical concern with early ERAs, remained low with macitentan. PDE5 inhibitors performed predictably, with side effects largely mild and transient.
Importantly, combination therapy did not significantly increase adverse events compared with monotherapy, suggesting that the incremental therapeutic gain does not come at the cost of safety. This aligns with findings from larger trials that have used similar dual approaches.
Safety matters greatly in PAH, where patients often require lifelong therapy. The reassuring tolerability of this combination makes it suitable not only for advanced disease but also for early intervention.
Clinical Implications: Redefining the Standard for Newly Diagnosed PAH
The pooled analysis supports a decisive shift in how clinicians approach newly diagnosed PAH. The evidence argues compellingly for initial combination therapy—not as an escalation tool but as a starting point. Healthcare providers should consider this strategy particularly for:
- patients in WHO functional class II–III at diagnosis,
- individuals with elevated biomarkers, early right ventricular dysfunction, or high-risk clinical features,
- younger patients, in whom long-term disease modification is essential.
Conversely, monotherapy remains appropriate for carefully selected patients with mild disease or contraindications. Yet even in these cases, early reassessment is crucial, with low thresholds for escalation.
Ultimately, the message is clear: dual pathway inhibition delivers superior disease control, reduces early deterioration, and provides a more stable functional trajectory for patients beginning their PAH journey.
Conclusion
The pooled analysis of macitentan and PDE5 inhibitors provides a coherent, biologically grounded, and clinically persuasive case for early combination therapy in newly diagnosed pulmonary arterial hypertension. While macitentan and PDE5 inhibitors each deliver meaningful benefits individually, their combined use produces deeper, faster, and more durable improvements across key metrics of disease progression.
Given PAH’s aggressive nature and the limited window during which interventions can truly alter disease trajectory, early combination therapy emerges not as an optional enhancement but as a strategically necessary approach. It restores vascular balance, reduces right ventricular strain, prevents early clinical worsening, and aligns with the mechanistic complexity of PAH pathophysiology.
The evidence is strong, the logic is compelling, and the therapeutic implications are profound: start early, target multiple pathways, and treat PAH with the respect its complexity demands.
FAQ
1. Why is combination therapy superior to monotherapy in newly diagnosed PAH?
Because PAH involves multiple dysregulated pathways. Targeting endothelin signaling and NO–cGMP pathways simultaneously produces synergistic effects that monotherapy cannot achieve.
2. Is macitentan–PDE5 inhibitor therapy safe to start immediately?
Yes. The pooled analysis demonstrates favorable tolerability, with adverse events similar to monotherapy and manageable in most patients.
3. Does every newly diagnosed patient require combination therapy?
Most benefit from it, particularly WHO class II–III patients. However, monotherapy may still be appropriate in select cases with mild disease or specific contraindications.