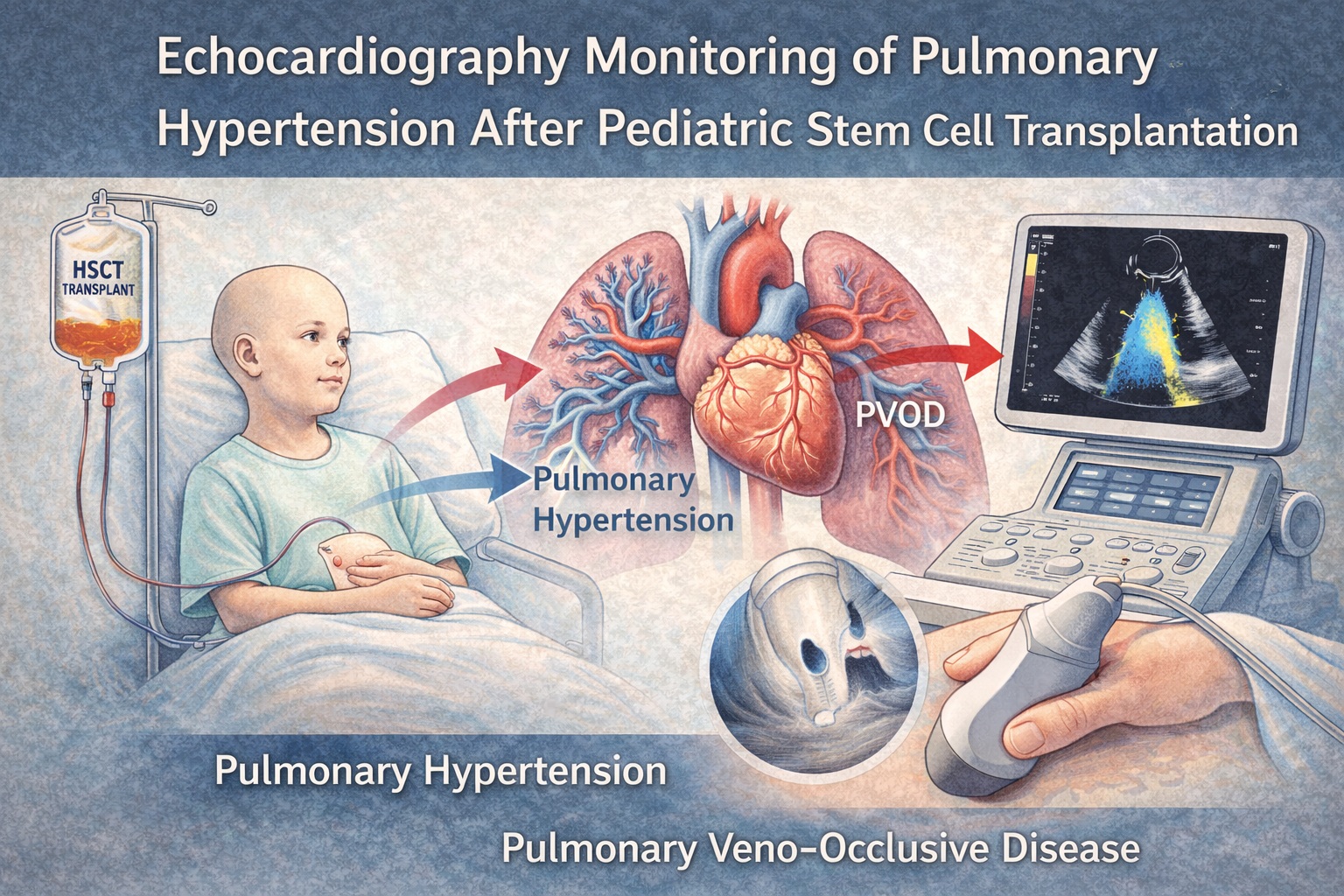
Pulmonary hypertension (PH) emerging after pediatric hematopoietic stem cell transplantation (HSCT) is a rare but devastating complication—an entity that tends to appear quietly yet progress aggressively. In clinical practice, it rarely announces itself early, instead hiding behind nonspecific respiratory complaints until right ventricular failure is already underway. The study underlying this article provides a detailed examination of PH and pulmonary veno-occlusive disease (PVOD) in pediatric HSCT survivors, highlighting the vital role of echocardiographic surveillance and the importance of early recognition.
As the paper demonstrates, PH in post-transplant children rarely follows the classical trajectory seen in idiopathic pulmonary arterial hypertension (PAH). Instead, it arises from a convergence of risk factors: endothelial injury from conditioning regimens, infections, graft-versus-host disease (GVHD), thrombotic microangiopathy, and direct toxicity from chemotherapeutic agents. The article emphasizes that diagnosis is particularly challenging in children, whose clinical presentation may be atypical and whose symptoms can easily be attributed to the complex post-HSCT recovery process.
Echo-based screening therefore becomes the clinician’s indispensable tool. Using sensitive parameters—including tricuspid regurgitant velocity (TRV), right ventricular systolic pressure (RVSP), right atrial dimensions, septal flattening, and qualitative assessments of right ventricular function—it is possible to detect early hemodynamic derangements long before overt PH emerges. The images and flow diagrams in the PDF (pages 3–5) clarify the echocardiographic features that distinguish PH, PH with left-sided involvement, and PVOD .
In the sections that follow, we explore the pathophysiology, clinical manifestations, and imaging strategies outlined in the manuscript, with special emphasis on the unique features of pediatric post-HSCT PH.
Why Pulmonary Hypertension Appears After Pediatric Stem Cell Transplantation
HSCT induces profound systemic changes. Conditioning regimens—including busulfan, cyclophosphamide, fludarabine, and total body irradiation—trigger endothelial damage, alter vascular reactivity, and cause pulmonary microangiopathy. The study underscores that endothelial injury is the central driver of PH in these patients. Damaged endothelial cells lose the ability to regulate vascular tone, produce nitric oxide, or maintain thromboresistant surfaces. Combined with inflammatory cytokines and donor–recipient immune interactions, this creates a fertile ground for progressive pulmonary vasculopathy.
Post-transplant PH often emerges in the setting of ongoing inflammation. Acute and chronic GVHD contribute to persistent immune-mediated endothelial injury, while infections—especially viral infections such as adenovirus or CMV—introduce additional layers of vascular stress. Thrombotic microangiopathy (TMA), frequently observed in the early post-transplant period, further narrows small pulmonary vessels and increases pulmonary vascular resistance. Importantly, children may develop PH even when systemic TMA manifestations are minor, making it a largely pulmonary event.
Another unique factor is the vulnerability of children’s developing lungs. While adult lungs are structurally mature at the time of HSCT, pediatric patients may experience disrupted alveolar and vascular growth, predisposing them to long-term ventilation–perfusion mismatch. The study notes that younger patients—particularly those transplanted in infancy—appear to have a higher risk of severe PH, possibly due to incomplete alveolarization and heightened sensitivity to endothelial injury.
The pdf’s data tables reflect that PH after pediatric HSCT may present within weeks or months of transplant, but cases several years later are also documented. This delayed manifestation complicates screening strategies and reinforces the need for long-term surveillance.
The Challenge of Diagnosing PH and PVOD in Children: Clinical Clues and Diagnostic Pitfalls
Diagnosis remains challenging because symptoms are nonspecific. Dyspnea, fatigue, tachypnea, and exercise intolerance—canonical signs of PH—may easily overlap with post-transplant complications such as anemia, infection, GVHD, or poor conditioning recovery. Younger children, particularly infants, often cannot articulate symptoms, relying instead on behavioral changes, feeding difficulties, or subtle respiratory distress.
As the article emphasizes, PVOD is particularly insidious. Unlike classical PAH, where vasoconstriction predominates, PVOD is a disease of the pulmonary venules and capillaries. Occlusion of post-capillary vessels increases pulmonary venous pressure without necessarily elevating TRV early on. Thus, echocardiography may initially appear deceptively normal. Clues highlighted in the manuscript include septal thickening, interlobular septal lines on CT, and significant hypoxemia disproportionate to apparent PH severity. High-resolution CT findings (page 4) may suggest PVOD even before overt hemodynamic collapse occurs .
Another diagnostic challenge is the overlap between PH and pulmonary complications such as idiopathic pneumonia syndrome, bronchiolitis obliterans, and infectious pneumonias. In the early post-transplant period, pulmonary edema can mimic PH by raising pulmonary pressures transiently. The study emphasizes that PH should be suspected when presumed respiratory issues fail to improve or when right ventricular dysfunction becomes apparent despite adequate pulmonary management.
Ultimately, echocardiography emerges as the frontline tool—but only when interpreted with pediatric-specific nuance and transplant-related context.
Echocardiography as the Primary Monitoring Tool: Parameters That Matter Most
Echocardiography offers a safe, non-invasive, and repeatable method for screening and monitoring PH. In children who may undergo dozens of imaging sessions over months or years, its portability and lack of radiation make it indispensable. The article outlines a structured echocardiographic approach, emphasizing parameters that provide early warning signs of PH or RV dysfunction.
TRV remains one of the most widely used markers, as elevated TRV indirectly estimates right ventricular systolic pressure (RVSP). However, in pediatric HSCT patients, TRV may be absent or unreliable due to poor acoustic windows or minimal tricuspid regurgitation. Thus, the study stresses alternative indicators: septal flattening, right ventricular hypertrophy, right atrial enlargement, and pulmonary artery acceleration time (PAAT). A shortened PAAT often precedes measurable TRV elevation.
Echocardiographic images on page 5 present typical findings in PH: interventricular septal shift, dilated RV outflow tract, and reduced tricuspid annular plane systolic excursion (TAPSE). TAPSE, while widely used in adults, requires pediatric Z-scores for proper interpretation. A low TAPSE in a child may reflect severe RV dysfunction or simply age-related normal variation, underscoring the need for careful calibration.
Interestingly, the study highlights that many children with early PH will exhibit preserved TAPSE because the RV’s longitudinal fibers compensate until late stages. Thus, clinicians must rely on global assessments—including fractional area change (FAC) and qualitative evaluation of RV wall motion—to detect early dysfunction.
The article also discusses the potential use of strain imaging (speckle-tracking echocardiography). Although not routinely used in all centers, longitudinal RV strain may detect dysfunction earlier than TAPSE or FAC. The research suggests that children with early post-HSCT PH demonstrate decreased strain even when conventional metrics remain normal.
Pulmonary Veno-Occlusive Disease: The Hidden Danger After HSCT
PVOD represents the most lethal form of post-HSCT pulmonary vascular disease. The study dedicates a significant portion of its discussion to PVOD, emphasizing that early recognition is crucial because standard PAH therapies—particularly vasodilators—can precipitate pulmonary edema and catastrophic respiratory failure.
What makes PVOD so challenging is that echocardiographic signs often lag behind clinical symptoms. Children may exhibit profound hypoxemia, tachypnea, and refractory respiratory distress while echocardiographic pulmonary pressures remain only mildly elevated. This mismatch arises because PVOD primarily increases post-capillary resistance, causing pulmonary edema and impaired oxygenation without immediate RV pressure overload.
As described in the text, CT imaging reveals centrilobular ground-glass opacities, septal thickening, and lymphadenopathy—findings that, while suggestive, are not specific. Bronchoalveolar lavage, rarely performed due to fragility of these patients, may show hemosiderin-laden macrophages, consistent with venous congestion.
The authors highlight that PVOD after HSCT often develops rapidly and may be misdiagnosed as infection or GVHD. Importantly, the PDF summarizes several case reports where early echocardiographic abnormalities—such as subtle RV dilation and borderline TRV elevation—were retrospectively recognized as early PVOD indicators.
Therefore, clinicians must approach unexplained hypoxemia with a high index of suspicion, using echocardiography not as a definitive diagnostic tool but as a continuous monitoring instrument to track hemodynamic deterioration.
Pathophysiology: Why PH Progresses Faster in Post-Transplant Children
Underlying molecular pathways align with the rapid progression seen in pediatric post-HSCT PH. Endothelial injury triggers a cascade of vasoconstrictive and proliferative signals, including:
- reduced NO production, impairing vasodilation,
- increased endothelin-1, driving smooth muscle proliferation,
- inflammatory cytokines activating fibroblasts and macrophages,
- microthrombi narrowing pulmonary vessels.
Children display heightened susceptibility due to more reactive pulmonary vasculature and increased metabolic demands. The study emphasizes that pulmonary artery smooth muscle cells (PASMCs) in children proliferate faster than in adults when exposed to inflammatory mediators, accelerating vascular remodeling.
Another important factor is mitochondrial injury. Conditioning regimens damage mitochondrial DNA and impair oxidative metabolism. The resulting energy deficit renders pulmonary endothelial cells vulnerable to apoptosis, endothelial detachment, and capillary dropout. The manuscript highlights evidence that mitochondrial dysfunction correlates strongly with PVOD development.
There is also growing recognition of genetic vulnerabilities. Mutations in EIF2AK4—which classically cause hereditary PVOD—may predispose some children to venous occlusion after HSCT, though the evidence remains preliminary.
These biological considerations underscore why early identification is essential: once remodeling is established, reversal becomes exceedingly unlikely in pediatric vasculature.
Clinical Management: Why Timing Is Everything
Once PH is detected, therapeutic decision-making becomes complex. The paper stresses that while standard PAH therapies—PDE5 inhibitors, endothelin receptor antagonists, prostacyclin analogues—may be beneficial in selected HSCT survivors, they carry significant risks in patients with occult PVOD. Vasodilation in PVOD can precipitate pulmonary edema by increasing capillary hydrostatic pressure in vessels already obstructed by fibrotic venular lesions.
Thus, management must be individualized. The article suggests beginning with cautious evaluation of echocardiographic findings, oxygenation status, imaging abnormalities, and laboratory markers. Vasodilators should not be initiated until PVOD is excluded or considered unlikely.
Supportive therapies, such as oxygen supplementation, diuretics, infection control, and treatment of underlying GVHD or TMA, remain essential. Extracorporeal membrane oxygenation (ECMO) may be required in severe cases, and lung transplantation is considered the ultimate option for refractory PVOD or rapidly progressive PH.
Importantly, the manuscript advocates for routine echocardiographic screening throughout the first year after HSCT, with additional evaluations triggered by symptoms, oxygen desaturation, or abnormalities in pulmonary imaging.
Conclusion
The reviewed article underscores a crucial truth: pulmonary hypertension and PVOD after pediatric stem cell transplantation are not rare anomalies but under-recognized, high-risk complications requiring vigilant monitoring. Echocardiography stands at the center of early detection—accessible, repeatable, and capable of revealing subtle shifts in right ventricular performance or pulmonary vascular resistance long before clinical collapse occurs.
Understanding the interplay between endothelial injury, immune dysregulation, mitochondrial dysfunction, and vascular remodeling provides the clinical rationale for close surveillance. Early detection is not merely desirable—it is life-saving. Adequate screening, cautious interpretation, and awareness of PVOD’s unique risks are essential to delivering safe, effective post-transplant care.
FAQ
1. Why is pulmonary hypertension so common after pediatric HSCT?
Because conditioning regimens, infections, inflammation, and GVHD cause extensive endothelial injury—making pediatric lungs particularly vulnerable to pulmonary vascular remodeling.
2. Can echocardiography detect PVOD early?
It can detect indirect signs, but early PVOD often presents with normal or mildly elevated pulmonary pressures. Hypoxemia out of proportion to echo findings should raise suspicion.
3. Are standard PAH medications safe in HSCT-related PH?
They can be beneficial in selected patients, but in PVOD they may cause pulmonary edema. Thus, vasodilator use requires careful clinical judgment and exclusion of venous occlusion.