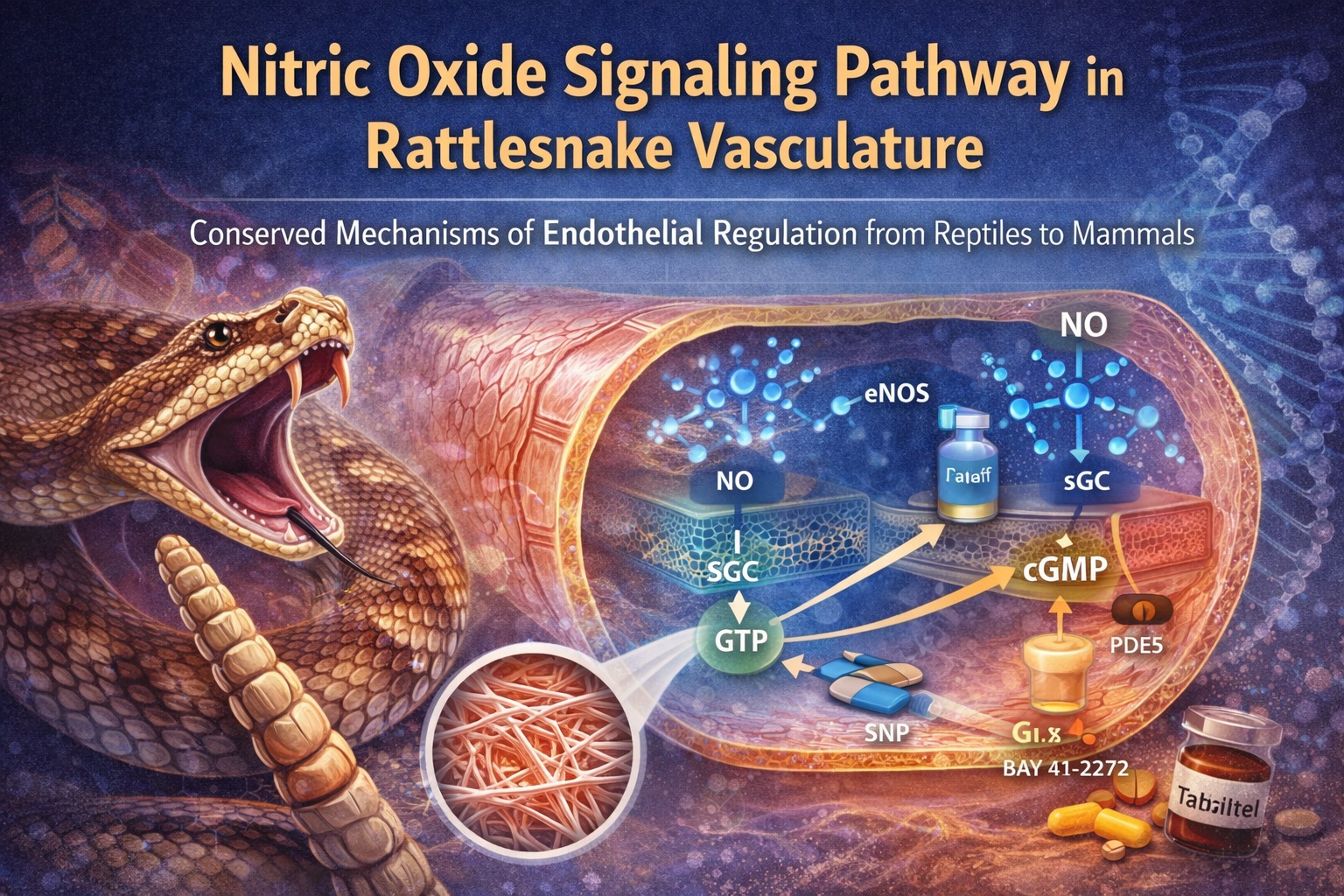
Introduction: Nitric Oxide Beyond Mammals
Nitric oxide (NO) occupies a privileged position in cardiovascular physiology. In mammals, it is almost impossible to discuss vascular tone, endothelial function, or smooth muscle relaxation without invoking the NO–cGMP signaling cascade. Over decades, this pathway has become a cornerstone of both basic science and clinical pharmacology, underpinning therapies ranging from nitrates to phosphodiesterase type 5 (PDE5) inhibitors.
Yet the evolutionary origins of this signaling system are often taken for granted. It is tempting to assume that such a refined molecular mechanism is a relatively recent innovation, perfected in mammals and merely adapted elsewhere. The reality, as increasingly revealed by comparative physiology, is far more intriguing. Elements of the NO signaling pathway appear across a wide phylogenetic spectrum, suggesting deep evolutionary roots rather than superficial convergence.
The study under consideration explores this idea with unusual clarity. By examining the aorta of Crotalus durissus terrificus, a South American rattlesnake, it demonstrates that the NO–soluble guanylyl cyclase (sGC)–PDE5 pathway is not only present, but functionally robust and strikingly similar to that found in mammals. This article expands on those findings, placing them into a broader biomedical and evolutionary context.
The Nitric Oxide–cGMP Axis: A Universal Language of Vascular Control
In mammalian vessels, NO is synthesized primarily by endothelial nitric oxide synthase (eNOS), diffuses into adjacent smooth muscle cells, and activates sGC. This activation triggers the conversion of guanosine triphosphate into cyclic guanosine monophosphate (cGMP), which in turn initiates a cascade leading to smooth muscle relaxation. PDE5 serves as the main brake in this system, hydrolyzing cGMP and terminating the signal.
What makes this pathway so biologically elegant is its modularity. Each component—NO synthesis, cGMP generation, and cGMP degradation—can be regulated independently. This modularity allows for fine-tuned control of vascular tone in response to neural, hormonal, and mechanical stimuli. Clinically, it also provides multiple pharmacological targets.
The relevance of this system outside mammals has been debated for years. Some studies in lower vertebrates suggested incomplete or atypical NO signaling, raising doubts about its universality. The current work challenges those doubts decisively by demonstrating a complete and functional NO–cGMP–PDE5 axis in reptilian vasculature.
Experimental Approach: Functional Physiology Meets Molecular Validation
One of the strengths of the study lies in its integrated methodology. Functional vascular assays, immunohistochemistry, and transcriptomic analysis are combined to create a coherent picture rather than isolated observations. This triangulation is particularly important in comparative biology, where functional similarity does not always imply molecular identity.
Isolated aortic rings from Crotalus durissus terrificus were used to assess vasomotor responses. Phenylephrine-induced contraction established a standardized baseline, against which relaxation responses could be measured. Endothelium-intact and endothelium-denuded preparations allowed clear discrimination between endothelial and smooth muscle contributions.
Pharmacological tools were chosen with precision. Acetylcholine tested endothelium-dependent NO release, sodium nitroprusside served as a direct NO donor, BAY 41–2272 and BAY 60–2770 probed distinct activation states of sGC, and tadalafil selectively inhibited PDE5. This systematic approach ensured that each node of the pathway was interrogated rather than inferred.
Endothelial Function in the Rattlesnake Aorta
Acetylcholine-induced relaxation is often considered the gold standard for assessing endothelial integrity. In the rattlesnake aorta, acetylcholine produced clear, concentration-dependent relaxation in endothelium-intact rings, an effect abolished by endothelial removal. This alone suggests a functional endothelial signaling system.
The inhibition of acetylcholine-induced relaxation by L-NAME, a nitric oxide synthase inhibitor, further confirms that NO mediates this response. Importantly, the absence of relaxation in denuded vessels rules out significant compensatory mechanisms originating from smooth muscle or perivascular nerves under these conditions.
Immunohistochemical analysis provides the anatomical correlate: eNOS expression is confined to endothelial cells, mirroring the mammalian pattern. This localization is not merely symbolic; it explains the functional dependence on an intact endothelium and supports the conclusion that endothelial NO production is the primary driver of acetylcholine-induced vasodilation in this species.
Neural Nitric Oxide: A Supporting but Distinct Role
While eNOS dominates endothelial signaling, neuronal nitric oxide synthase (nNOS) plays a complementary role in vascular regulation. In mammals, nNOS is often associated with perivascular nerves, contributing to neurogenic modulation of vascular tone.
The rattlesnake aorta displays a remarkably similar arrangement. nNOS immunoreactivity is detected exclusively in perivascular nerve fibers located in the adventitial layer. This spatial segregation suggests functional specialization rather than redundancy. Endothelial NO governs basal and receptor-mediated responses, while neural NO likely modulates tone in response to autonomic input.
This distinction matters. It implies that the division of labor between endothelial and neural sources of NO predates the divergence of reptiles and mammals. In evolutionary terms, this argues for conservation rather than independent innovation.
Soluble Guanylyl Cyclase: Conserved Structure, Conserved Function
Soluble guanylyl cyclase is the molecular linchpin of NO signaling. Its ability to sense NO and translate it into cGMP production underlies the entire pathway. The presence of functional sGC in reptilian vessels had previously been inferred indirectly, but not demonstrated comprehensively.
The study shows that both heme-dependent and heme-independent sGC activation mechanisms operate in the rattlesnake aorta. BAY 41–2272, which stimulates sGC in a heme-dependent manner, produces potent relaxation that is sensitive to both NOS inhibition and sGC blockade. This indicates that endogenous NO contributes to its effect.
Conversely, BAY 60–2770, a heme-independent sGC activator, produces even more potent relaxation, particularly under conditions where NO synthesis or heme availability is compromised. The dramatic potentiation of BAY 60–2770 responses by ODQ highlights the presence of oxidized or heme-free sGC in the tissue, a phenomenon previously characterized in mammals.
Transcriptomic analysis reinforces these functional findings. High sequence identity between rattlesnake and human sGC subunits confirms that structural conservation accompanies functional similarity. This molecular continuity strengthens the argument for evolutionary preservation of the pathway.
Phosphodiesterase Type 5: The Brake Is Also Conserved
No signaling pathway is complete without a mechanism for termination. In the NO–cGMP axis, PDE5 fulfills this role by hydrolyzing cGMP and limiting signal duration. The clinical importance of PDE5 is well established, given the widespread use of its inhibitors in cardiovascular and urological medicine.
In the rattlesnake aorta, tadalafil induces robust, concentration-dependent relaxation in endothelium-intact vessels. This effect is attenuated by endothelial removal and by sGC inhibition, indicating that PDE5 inhibition amplifies endogenous NO–cGMP signaling rather than acting through an independent mechanism.
Immunohistochemistry localizes PDE5 predominantly to vascular smooth muscle, consistent with its role in modulating intracellular cGMP levels. Transcriptomic data reveal substantial sequence homology with mammalian PDE5, underscoring evolutionary conservation at both functional and genetic levels.
The implication is subtle but profound: the pharmacological logic exploited in human medicine operates on a molecular framework that predates mammals by hundreds of millions of years.
Functional Integration of the NO–cGMP–PDE5 Pathway
Perhaps the most compelling aspect of the study is not the identification of individual components, but the demonstration that they function as an integrated system. Endothelial NO production, sGC activation, cGMP accumulation, and PDE5-mediated degradation all interact dynamically to regulate vascular tone.
This integration is evident in pharmacological cross-modulation. PDE5 inhibition enhances sGC-mediated relaxation, sGC activation bypasses NO deficiency, and NO synthesis inhibition shifts the balance toward NO-independent mechanisms. Such interactions are hallmarks of a mature signaling network rather than a rudimentary system.
From a physiological standpoint, this integration allows the rattlesnake to regulate vascular tone under diverse environmental and metabolic conditions. From an evolutionary standpoint, it suggests that the core architecture of vascular signaling was established early and refined rather than reinvented.
Evolutionary Perspective: Conservation Over Innovation
The presence of a fully functional NO–cGMP–PDE5 pathway in reptilian vessels challenges simplistic narratives of evolutionary progression. Rather than viewing mammalian cardiovascular regulation as a unique achievement, it may be more accurate to see it as a sophisticated variation on an ancient theme.
Differences between species likely reflect variations in sensitivity, regulatory inputs, or expression levels rather than wholesale changes in signaling logic. This perspective aligns with the study’s conclusion that evolutionary divergence in vascular reactivity may be driven by modulatory factors rather than fundamental transduction mechanisms.
Such insights have implications beyond comparative physiology. They inform how we interpret animal models, how we extrapolate pharmacological data across species, and how we conceptualize the evolutionary origins of drug targets.
Clinical and Pharmacological Implications
Although rattlesnakes are unlikely patients, the relevance of these findings to human medicine should not be underestimated. The demonstration of conserved NO signaling validates the use of non-mammalian models for studying fundamental vascular mechanisms and testing pharmacological principles.
Moreover, understanding how sGC activators and PDE5 inhibitors function in diverse biological contexts may inform the development of next-generation therapeutics. The distinction between heme-dependent and heme-independent sGC activation, in particular, has growing relevance in conditions characterized by oxidative stress and endothelial dysfunction.
In a broader sense, the study reminds us that successful drugs often exploit deeply conserved biological pathways. Their efficacy is not an accident of modern physiology, but a reflection of evolutionary robustness.
Conclusion
The nitric oxide–cGMP–PDE5 signaling pathway in the aorta of Crotalus durissus terrificus is functionally complete, molecularly conserved, and physiologically sophisticated. Endothelial NO production, neural modulation, sGC activation, and PDE5-mediated regulation operate in concert to control vascular tone, closely mirroring mammalian systems.
These findings reinforce the concept that key mechanisms of cardiovascular regulation are ancient and resilient. Evolution has modified their expression and regulation, but not their fundamental logic. For medicine and pharmacology, this continuity offers both reassurance and opportunity: reassurance that our models rest on solid biological foundations, and opportunity to explore new therapeutic strategies informed by evolutionary insight.
FAQ
1. Why study nitric oxide signaling in reptiles?
Because it reveals whether fundamental vascular mechanisms are evolutionarily conserved, improving our understanding of both physiology and pharmacology.
2. Is the NO–cGMP–PDE5 pathway identical in reptiles and mammals?
Not identical, but remarkably similar in structure and function, with differences mainly in regulation rather than core mechanisms.
3. Do these findings have clinical relevance?
Yes. They support the evolutionary robustness of drug targets such as sGC and PDE5, strengthening confidence in their translational relevance.