Modern urology increasingly favors minimally invasive solutions. Prostatic artery embolization (PAE) has emerged as an attractive alternative to traditional surgical interventions for benign prostatic hyperplasia (BPH), offering symptom relief without tissue resection and with a lower risk of sexual dysfunction. Yet, as with all endovascular procedures, technical elegance does not eliminate biological risk. Rare complications still occur, and when they do, they tend to be dramatic, distressing, and poorly addressed by existing guidelines.
Glans penis necrosis following PAE is one such complication. It is infrequently reported, lacks standardized management, and carries profound physical and psychological implications for patients. The case described in the source article illustrates not only the vulnerability of pelvic vascular anatomy but also the therapeutic potential of hyperbaric oxygen therapy (HBOT) in salvaging ischemic penile tissue .
This article explores the clinical significance of penile ischemia after PAE, the mechanisms that underlie non-target embolization, and the rationale for hyperbaric oxygen therapy as a limb- and organ-preserving intervention. More importantly, it offers practical insight into how clinicians should recognize, interpret, and manage this rare but serious complication.
Prostatic Artery Embolization and the Price of Precision
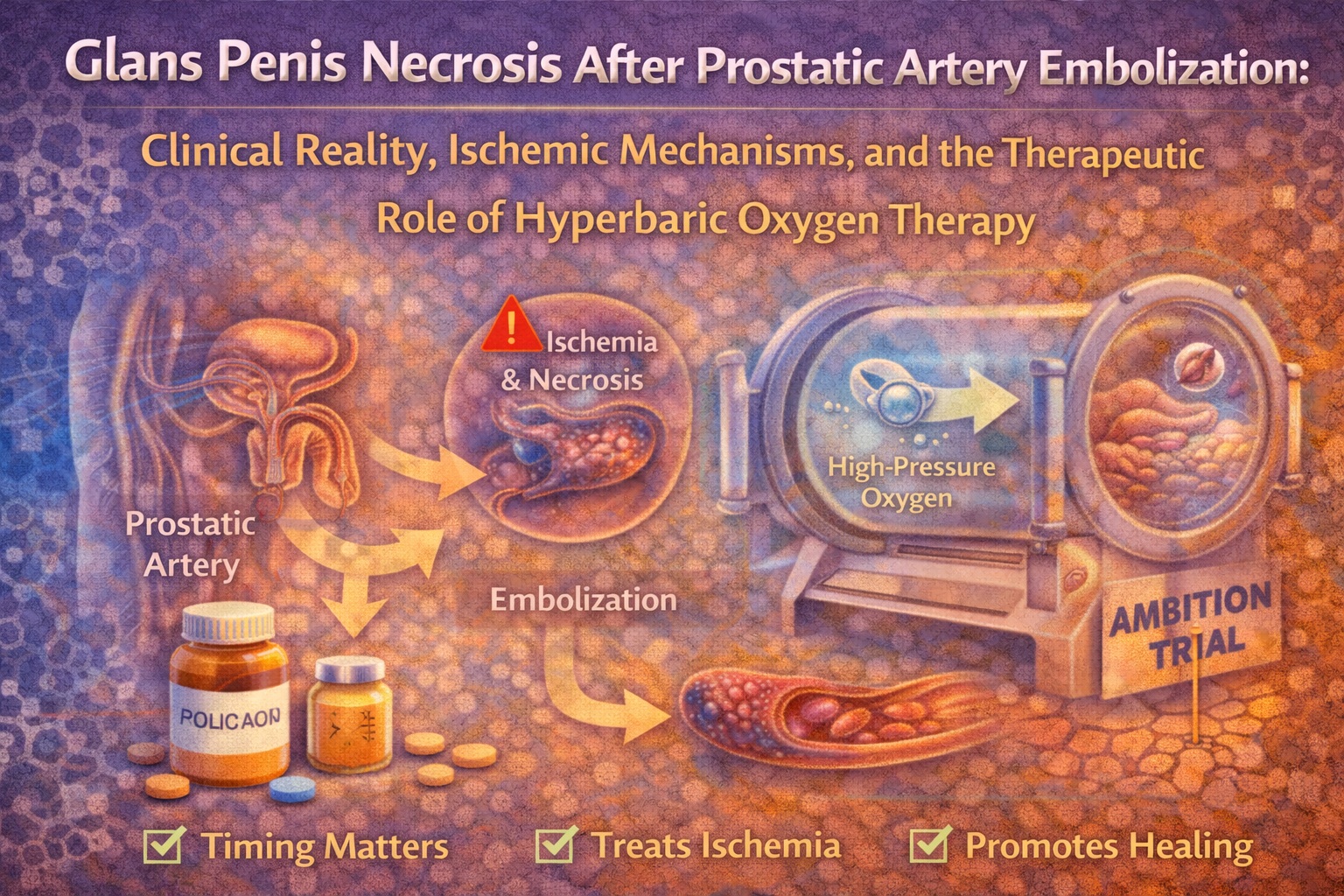
Prostatic artery embolization represents a paradigm shift in the management of BPH. By selectively occluding prostatic arterial supply, the procedure induces ischemic shrinkage of prostatic tissue, relieving obstruction while avoiding the morbidity associated with transurethral resection. From a conceptual standpoint, the appeal is obvious: less invasiveness, fewer sexual side effects, and faster recovery.
However, pelvic vascular anatomy is rarely cooperative. The arterial supply to the prostate frequently includes anastomoses with branches serving the bladder, rectum, and penis. Even when embolic particles are delivered slowly and with meticulous angiographic guidance, non-target embolization remains a recognized risk. The penis, with its end-arterial characteristics and limited collateral circulation, is particularly vulnerable to ischemic injury.
In the reported case, embolization of the left prostatic artery using microspheres was technically successful, while the right prostatic artery could not be cannulated. Despite procedural caution, embolic material reached penile vasculature, resulting in acute ischemia of the glans. This outcome underscores an uncomfortable truth: in endovascular medicine, “minimally invasive” does not equate to “minimally consequential.”
Penile Ischemia as a Clinical Emergency in Disguise
Penile ischemia does not announce itself politely. Initial symptoms may be subtle—distal penile pain, mild discoloration, or sensory changes—easily dismissed in the immediate post-procedural period. Yet beneath these early signs lies a time-sensitive vascular catastrophe. Without prompt recognition and intervention, ischemia progresses rapidly to necrosis.
In the described patient, pain developed within hours of PAE and escalated over days, culminating in visible necrosis of the glans. Imaging confirmed absent arterial flow, a finding that transforms clinical suspicion into diagnostic certainty. At this point, the goal shifts from prevention to salvage.
What makes penile ischemia particularly challenging is the absence of standardized treatment pathways. Unlike myocardial or cerebral ischemia, there is no universally accepted algorithm. Management is often extrapolated from principles of peripheral arterial disease, wound care, and reconstructive surgery—fields that rarely intersect in a single patient.
Pathophysiology of Non-Target Embolization
To understand why hyperbaric oxygen therapy can be effective, one must first appreciate the pathophysiology of non-target embolization. Embolic particles obstruct small arterial branches, abruptly halting oxygen delivery to dependent tissues. In the penis, this results in acute hypoxia, endothelial dysfunction, inflammatory cascades, and eventual cell death.
The glans penis is uniquely susceptible. Its vascular supply is terminal, its metabolic demands are high, and its tolerance for ischemia is low. Unlike muscle tissue, which can endure hypoxia for hours, penile tissue progresses quickly from reversible ischemia to irreversible necrosis.
This pathophysiological cascade explains why conservative observation alone is risky. While spontaneous resolution has been reported, it is unpredictable. In many cases, delay simply allows ischemia to consolidate into tissue loss, increasing the likelihood of surgical intervention and permanent functional impairment.
Hyperbaric Oxygen Therapy: From Theory to Clinical Application
Hyperbaric oxygen therapy occupies an unusual position in modern medicine. It is widely available, physiologically elegant, and supported by strong mechanistic rationale—yet often underutilized outside a narrow range of indications. In the context of acute arterial insufficiency, its relevance is particularly compelling.
HBOT works by delivering 100% oxygen at increased atmospheric pressure, dramatically increasing dissolved oxygen content in plasma. This allows oxygen delivery to ischemic tissues even in the absence of normal arterial flow. In effect, it buys time—time for collateral circulation to develop, inflammation to subside, and tissue viability to be restored.
In the reported case, HBOT was initiated nearly two weeks after the embolization event. Despite this delay, the patient experienced rapid pain relief and progressive resolution of necrosis. This outcome highlights two important points: penile tissue retains surprising regenerative potential, and hyperbaric oxygen can meaningfully influence ischemic recovery even beyond the acute phase.
Multimodal Therapy and the Logic of Combination Treatment
Hyperbaric oxygen therapy did not act in isolation. The patient received a combination of systemic analgesia, vasodilatory therapy with tadalafil, and topical anesthetics. Each component addressed a different aspect of the ischemic process: pain control, microvascular dilation, and symptomatic relief.
This multimodal approach reflects clinical pragmatism rather than protocol. In the absence of evidence-based guidelines, clinicians must construct rational treatment strategies grounded in physiology. The success of this case suggests that HBOT functions best not as a standalone solution, but as a central pillar within a broader therapeutic framework.
Importantly, HBOT was well tolerated. Adverse effects were absent, and treatment was completed without complication. This safety profile is consistent with broader HBOT experience and strengthens the argument for its early consideration in similar cases.
Practical Considerations for Clinicians
For urologists, interventional radiologists, and wound specialists, penile necrosis after PAE represents a shared responsibility. Early recognition depends on vigilance, patient education, and a low threshold for vascular imaging when symptoms arise.
From a therapeutic standpoint, clinicians should consider the following principles:
- Penile pain or discoloration after PAE is never “normal” and warrants urgent evaluation
- Imaging confirmation of ischemia should prompt immediate multidisciplinary discussion
- Hyperbaric oxygen therapy should be considered early, not as a last resort
These principles are not radical; they simply apply established ischemia management concepts to an anatomical region that is often overlooked in vascular medicine.
Ethical and Psychological Dimensions
Beyond physiology and therapy lies an often-ignored dimension: psychological impact. Penile necrosis is profoundly distressing. It affects body image, sexual identity, and quality of life. Even transient ischemic injury can produce lasting anxiety.
Successful resolution, therefore, carries benefits far beyond tissue preservation. It restores dignity, autonomy, and trust in medical care. From this perspective, HBOT is not merely a wound-healing tool—it is an intervention with psychosocial significance.
Future Directions and the Need for Clinical Awareness
The rarity of reported cases should not breed complacency. As PAE becomes more widely adopted, absolute numbers of complications will rise, even if relative risk remains low. Without awareness, these cases may be misdiagnosed, undertreated, or dismissed as inevitable.
Future research should aim to define optimal treatment timing, session number, and combination strategies for HBOT in penile ischemia. Until such data exist, case-based reasoning remains our best guide.
Conclusion
Glans penis necrosis after prostatic artery embolization is a rare but serious complication that demands prompt recognition and decisive management. The case analyzed here demonstrates that hyperbaric oxygen therapy can play a critical role in reversing ischemic injury, relieving pain, and preserving tissue integrity.
In an era that prizes minimally invasive solutions, clinicians must remember that complications, when they occur, require maximal clinical attention. Hyperbaric oxygen therapy offers a physiologically sound, clinically effective, and ethically compelling option for managing penile ischemia after PAE—and deserves a place in the modern urological toolkit.
FAQ
1. How soon should hyperbaric oxygen therapy be started after penile ischemia is suspected?
Ideally as early as possible after diagnosis, but meaningful benefit may still occur even when therapy is initiated days later.
2. Is hyperbaric oxygen therapy safe for elderly patients?
Yes. When properly screened and monitored, HBOT is generally well tolerated, including in older patients.
3. Can penile necrosis after PAE resolve without treatment?
Spontaneous resolution has been reported, but it is unpredictable. Early intervention significantly improves the likelihood of full recovery.