Introduction: When One Mechanism Is No Longer Enough
Benign prostatic hyperplasia (BPH) is no longer viewed as a purely anatomical problem of prostate enlargement. It is a chronic, progressive condition characterized by a complex interaction of hormonal, vascular, neuromuscular, and inflammatory mechanisms. As life expectancy increases, so does the prevalence of BPH-associated lower urinary tract symptoms (LUTS), making effective long-term management a priority rather than a convenience.
For decades, medical therapy for BPH followed a relatively linear logic. Alpha-blockers were introduced to rapidly relieve symptoms by relaxing prostatic smooth muscle, while 5α-reductase inhibitors (5-ARIs) such as finasteride were later added to reduce prostate volume and slow disease progression. Combination therapy with these two classes became the standard of care for men with larger prostates and moderate to severe symptoms.
The emergence of tadalafil, a phosphodiesterase type 5 (PDE5) inhibitor, has challenged this paradigm. Originally developed for erectile dysfunction, tadalafil demonstrated consistent and clinically meaningful improvements in LUTS. Its combination with finasteride represents a logical extension of combination therapy—one that integrates symptom relief, disease modification, and preservation of sexual function within a single treatment strategy.
The Biological Rationale for Combining Tadalafil and Finasteride
Finasteride and tadalafil act on fundamentally different—but complementary—pathophysiological pathways. Finasteride inhibits the conversion of testosterone to dihydrotestosterone (DHT), the androgen primarily responsible for prostatic growth. Over time, this leads to a reduction in prostate volume, decreased risk of acute urinary retention, and lower likelihood of surgical intervention. However, finasteride’s clinical effects are slow to emerge, often requiring six months or more before meaningful symptom improvement is observed.
Tadalafil, in contrast, exerts its effects through enhancement of the nitric oxide (NO)–cyclic guanosine monophosphate (cGMP) pathway. PDE5 inhibition increases cGMP availability, resulting in smooth muscle relaxation within the prostate, bladder neck, urethra, and their supporting vasculature. This mechanism produces relatively rapid symptom relief without altering prostate size.
The combination of these agents therefore addresses both the functional and structural components of BPH. Finasteride modifies disease progression, while tadalafil improves dynamic obstruction and pelvic perfusion. Importantly, tadalafil may also counterbalance the sexual side effects commonly associated with finasteride, a feature that has substantial implications for long-term adherence and patient satisfaction.
Clinical Evidence Supporting the Combination Approach
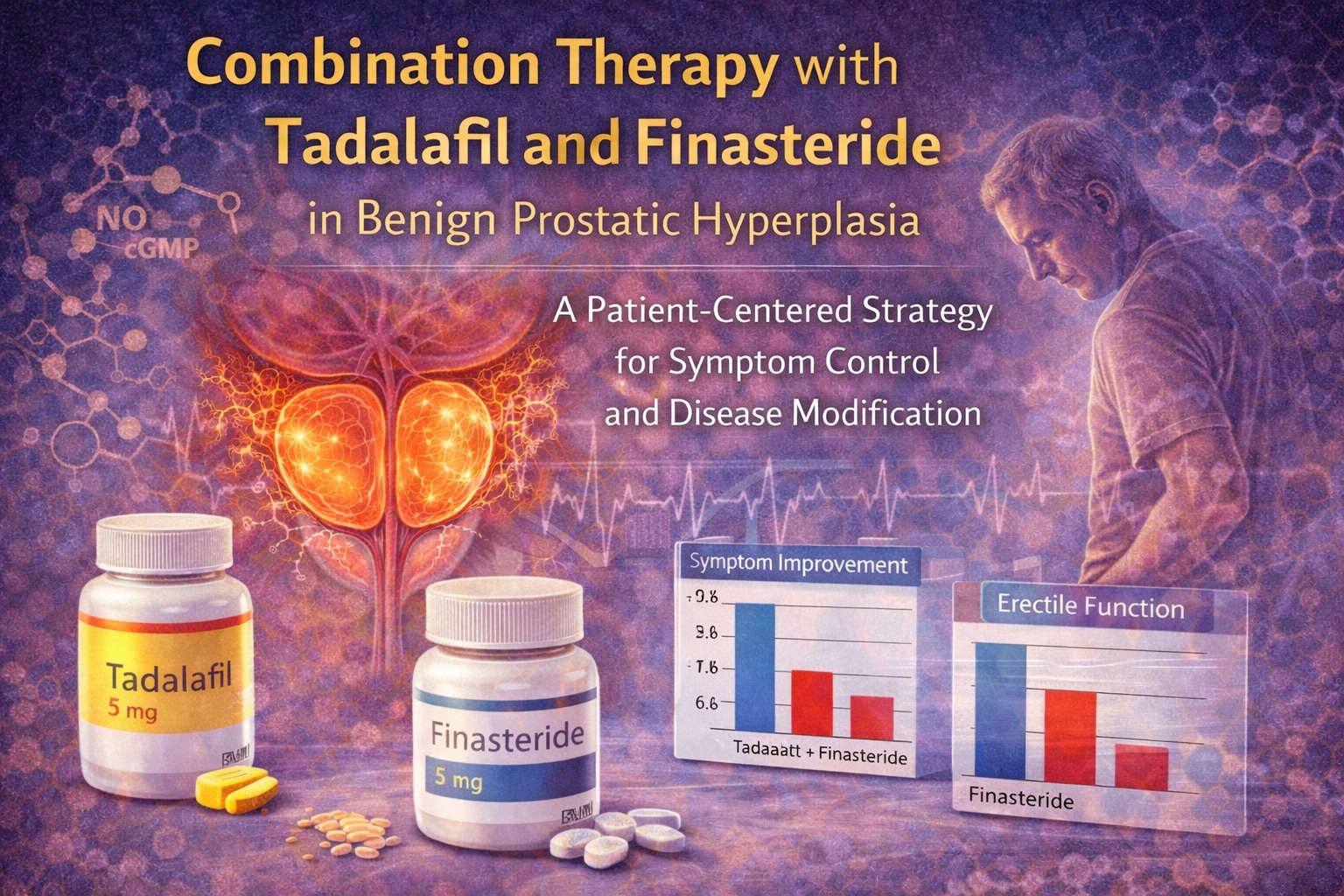
The clinical foundation for combining tadalafil and finasteride rests primarily on a landmark randomized, double-blind study in men with moderate to severe LUTS and prostate volumes greater than 30 g. In this trial, patients receiving combination therapy experienced significantly greater improvements in International Prostate Symptom Score (IPSS) compared with those receiving finasteride and placebo.
Symptom improvement was evident as early as four weeks, a clinically meaningful finding given the traditionally delayed onset of finasteride monotherapy. By 12 and 26 weeks, the superiority of combination therapy persisted, suggesting not only faster but also more robust symptom control.
Equally important was the effect on erectile function. While finasteride alone was associated with a decline in erectile function scores early in treatment, the addition of tadalafil resulted in consistent and sustained improvements across all measured domains of sexual function. This dual benefit distinguishes the tadalafil–finasteride combination from traditional BPH regimens.
Symptom Relief Versus Flow Rates: Understanding What Matters to Patients
One recurring observation in BPH research is the poor correlation between objective measures such as maximum urinary flow rate (Qmax) and patient-reported symptom severity. While finasteride and tadalafil may produce modest improvements in Qmax, these changes do not fully explain the substantial reductions in IPSS.
This discrepancy underscores a critical point: patients experience BPH primarily as a quality-of-life disorder rather than a urodynamic abnormality. Improvements in nocturia, urgency, weak stream, and sensation of incomplete emptying often matter more than numerical changes in flow rate.
The tadalafil–finasteride combination appears particularly effective in addressing these patient-centered outcomes. By reducing smooth muscle tone and improving pelvic blood flow, tadalafil alleviates irritative and obstructive symptoms that may persist despite reductions in prostate size alone.
Sexual Function as a Core Treatment Outcome
Sexual dysfunction is not a peripheral concern in BPH management—it is central to treatment acceptance and persistence. Finasteride is well known to cause erectile dysfunction, reduced libido, ejaculatory disorders, and decreased semen volume in a subset of patients. Even when these effects are statistically infrequent, their clinical impact can be profound.
The addition of tadalafil fundamentally alters this equation. Clinical data demonstrate that combination therapy not only prevents finasteride-associated sexual decline but often improves baseline sexual function, even in men without pre-existing erectile dysfunction. Improvements have been observed across multiple domains, including erectile rigidity, orgasmic function, sexual desire, and overall satisfaction.
This unexpected enhancement of sexual function may reflect broader effects of NO pathway activation on genital blood flow and neural responsiveness. While these findings should be interpreted cautiously, they highlight an important advantage of the combination strategy: treatment of LUTS no longer requires sacrificing sexual health.
Safety and Tolerability of Combination Therapy
From a safety perspective, the tadalafil–finasteride combination demonstrates a favorable and predictable profile. Adverse events associated with tadalafil, such as headache, dyspepsia, back pain, and flushing, occur at low frequencies and are generally mild. Finasteride-related adverse effects are not increased by the addition of tadalafil.
Importantly, the combination does not appear to exacerbate cardiovascular risk in appropriately selected patients. However, tadalafil remains contraindicated in men using nitrates or those with significant hypersensitivity to PDE5 inhibitors. Careful patient selection and counseling are therefore essential.
Notably, combination therapy is not indicated for men whose primary complaint is nocturia. This symptom often reflects bladder dysfunction or sleep disorders rather than outlet obstruction, and alternative management strategies may be more appropriate.
Identifying the Ideal Patient for Tadalafil–Finasteride Therapy
The success of combination therapy depends on appropriate patient selection. Based on available evidence, the ideal candidate is a man with moderate to severe LUTS, a prostate volume greater than 30 g, and a desire for more comprehensive symptom control than finasteride alone can provide.
Men with coexisting erectile dysfunction represent a particularly strong indication. In this population, tadalafil not only improves urinary symptoms but also enhances sexual function, addressing two interrelated conditions with a single therapeutic approach.
Combination therapy may also be considered in treatment-naïve patients with large prostates who seek rapid symptom relief while simultaneously reducing long-term progression risk. In such cases, tadalafil provides early benefit while finasteride exerts its disease-modifying effects over time.
Comparison with Alpha-Blocker–Based Combinations
Alpha-blocker–finasteride combinations are well established and effective, but they are not without limitations. Alpha-blockers are associated with orthostatic hypotension, dizziness, and a high incidence of ejaculatory dysfunction, particularly with more selective agents such as silodosin.
Compared with alpha-blocker combinations, tadalafil–finasteride therapy offers several advantages. It avoids adrenergic side effects, has a lower risk of ejaculatory disorders, and provides additional sexual benefits. While direct comparative trials are limited, the overall risk–benefit profile favors tadalafil-based combinations in appropriately selected patients.
That said, alpha-blockers remain valuable options, particularly for patients who cannot use PDE5 inhibitors. The choice between these strategies should be individualized rather than dogmatic.
Long-Term Disease Management and Patient Adherence
BPH is a chronic condition, and long-term adherence to therapy is essential for sustained benefit. Sexual side effects are a leading cause of treatment discontinuation with finasteride and alpha-blockers. By mitigating or reversing these effects, tadalafil may significantly improve adherence.
Improved adherence has downstream consequences. Consistent finasteride use is necessary to achieve prostate volume reduction and reduce the risk of acute urinary retention and surgery. In this context, tadalafil indirectly enhances the disease-modifying potential of finasteride by making long-term therapy more tolerable.
This patient-focused perspective is central to modern BPH management. Effective therapy is not defined solely by symptom scores, but by how well it aligns with patient priorities and expectations.
Conclusion
The combination of tadalafil and finasteride represents a rational, evidence-based evolution in the medical management of benign prostatic hyperplasia. By integrating rapid functional symptom relief with long-term disease modification and preservation of sexual function, this strategy addresses the multifaceted nature of BPH more comprehensively than monotherapy.
For men with moderate to severe LUTS and enlarged prostates—particularly those with concomitant erectile dysfunction—tadalafil–finasteride combination therapy offers meaningful clinical advantages. Its favorable safety profile and patient-centered benefits position it as a valuable option in contemporary urological practice.
As BPH management continues to shift toward individualized, quality-of-life–driven care, combination strategies that respect both urinary and sexual health are likely to play an increasingly prominent role.
FAQ
1. Does tadalafil replace alpha-blockers in BPH treatment?
No. Tadalafil offers an alternative mechanism of action and may be preferable in selected patients, but alpha-blockers remain effective options.
2. How quickly does symptom improvement occur with combination therapy?
Symptom relief may begin within weeks due to tadalafil, while finasteride provides longer-term benefits over several months.
3. Is combination therapy suitable for all men with BPH?
No. It is best suited for men with larger prostates, moderate to severe symptoms, and no contraindications to PDE5 inhibitors.