Introduction
The twenty-first century has reshaped expectations for pharmaceutical analysis. Gone are the days when an analytical chemist could rely on a bulky spectrophotometer and a slow chromatographic column to keep counterfeiters and dosage errors at bay. Today’s clinicians, regulators, and patients demand rapid, precise, and sustainable methods to ensure the authenticity and quality of medicines. Among the many drugs that require rigorous monitoring, tadalafil—a widely prescribed phosphodiesterase type 5 (PDE5) inhibitor used for erectile dysfunction (ED) and benign prostatic hyperplasia (BPH)—has gained particular importance.
The global market for PDE5 inhibitors is vast, and, unfortunately, so is the counterfeit drug trade. Poor-quality formulations may contain no active drug, the wrong concentration, or even harmful adulterants. Inaccurate dosing compromises efficacy and may increase adverse effects. For this reason, developing reliable analytical techniques for tadalafil detection is not merely an academic exercise—it is a matter of public health.
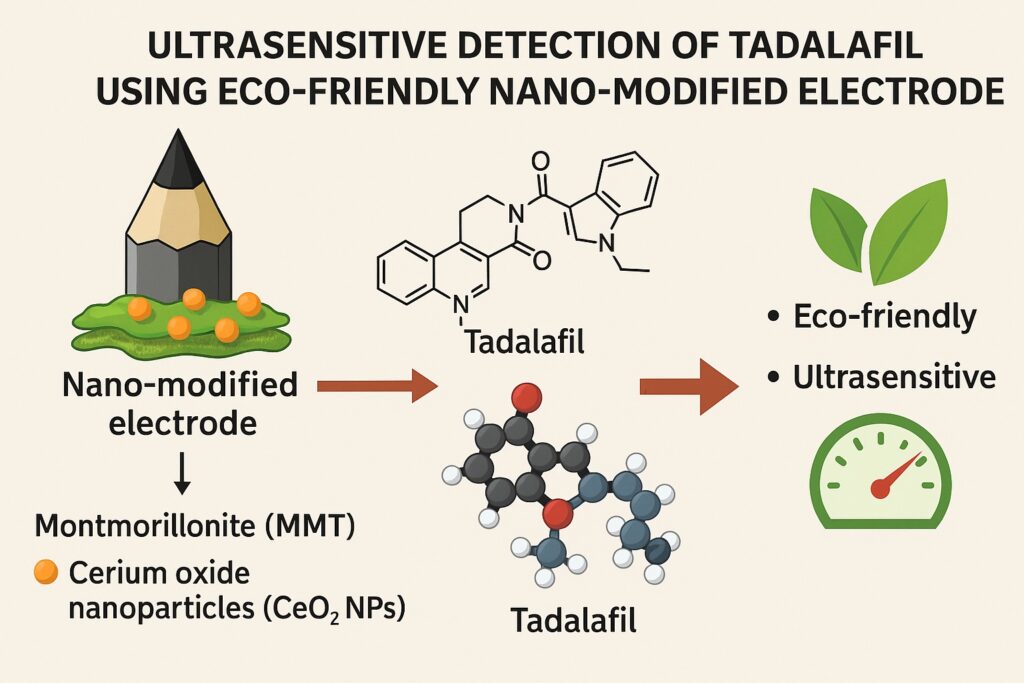
Traditional analytical techniques such as HPLC, LC-MS/MS, and spectrophotometry have set the gold standard. They are highly accurate, but they come at a cost: sophisticated infrastructure, high maintenance, expensive reagents, and long turnaround times. In contrast, electrochemical sensors have emerged as nimble alternatives, offering speed, sensitivity, and cost-effectiveness. The recent development of a montmorillonite clay (MMT)–cerium oxide nanoparticle (CeO₂ NPs)–modified composite pencil graphite paste electrode (MMT-CeO₂ NPs/CPG-PE) represents a remarkable stride in this field. By blending sustainability with nanoscale innovation, this sensor achieves ultrasensitive tadalafil detection even in complex biological fluids.
This article provides a detailed exploration of this eco-friendly electrochemical platform. We will examine the scientific rationale, fabrication method, detection mechanism, analytical performance, and broader implications for pharmaceutical quality control and clinical diagnostics. Along the way, we will confront the limitations of existing methods, highlight the sustainability dimension, and consider how such tools may reshape the future of drug monitoring.
Why Reinvent the Analytical Wheel?
It is tempting to ask: why develop a new method when existing ones already serve the purpose? The answer lies in context. HPLC and LC-MS/MS are indeed powerful, but they are poorly suited for rapid screening, decentralized testing, or low-resource settings. Imagine a customs officer at a border checkpoint tasked with evaluating a suspicious batch of tablets. Sending samples to a central lab for HPLC analysis may take days, during which counterfeit drugs could already be distributed. An on-site, portable, low-cost sensor capable of detecting tadalafil at nanogram levels could change that scenario entirely.
Furthermore, counterfeit drugs are often produced under conditions that escape regulatory oversight. In regions where laboratory capacity is limited, the very technologies that could prevent harm are inaccessible. Democratizing pharmaceutical analysis by developing simple, reusable, and inexpensive sensors is therefore a moral imperative, not just a scientific challenge.
Electrochemical detection is particularly attractive because it requires minimal instrumentation, delivers results in real time, and achieves remarkable sensitivity when electrodes are properly modified. The trick is to design electrode surfaces that enhance electron transfer and reduce interference, while being robust and easy to fabricate. This is where nanotechnology and natural materials enter the picture.
The Role of Nanotechnology in Electrochemical Sensing
Nanomaterials have transformed sensor design by offering extraordinary surface-to-volume ratios, high catalytic activity, and tunable physicochemical properties. Cerium oxide nanoparticles (CeO₂ NPs), in particular, bring unique advantages. Their ability to switch between Ce³⁺ and Ce⁴⁺ oxidation states allows them to participate in redox cycling and catalyze oxidation reactions. Additionally, CeO₂ is biocompatible, inexpensive, and relatively easy to synthesize.
Montmorillonite (MMT) clay, though less glamorous than nanoparticles, plays an equally important role. A naturally abundant aluminosilicate, MMT is valued for its layered structure, high cation-exchange capacity, and adsorptive properties. It prevents nanoparticle aggregation, increases porosity, and offers a large active surface for analyte interaction. When combined with CeO₂ NPs, MMT forms a hybrid material that maximizes conductivity, stability, and sensitivity.
This marriage of nanoscience and natural clay highlights a broader trend in modern research: solving high-tech problems with eco-friendly, sustainable materials. It is not enough for a sensor to be sensitive—it must also be scalable, reproducible, and kind to the environment.
Fabricating the Composite Electrode
The beauty of the MMT-CeO₂ NPs/CPG-PE lies in its simplicity. Instead of relying on expensive electrodes coated with exotic chemicals, the researchers used a pencil graphite electrode—yes, essentially the core of a common pencil. By mixing graphite powder with paraffin oil, they created a paste electrode, which is inexpensive, disposable, and easy to renew by simple polishing.
To this base, they added the MMT-CeO₂ composite. The preparation involved dispersing CeO₂ nanoparticles into montmorillonite clay, drying the mixture, and grinding it into a fine powder. This powder was incorporated into the graphite paste, creating a modified surface with vastly enhanced electrochemical properties.
Characterization studies confirmed the success of this modification. Scanning electron microscopy revealed a porous, roughened surface with well-distributed nanoparticles. Electrochemical impedance spectroscopy demonstrated reduced resistance and improved conductivity. Cyclic voltammetry showed higher current responses compared with unmodified electrodes. In other words, the nanocomposite was not just decorative—it made the electrode far more effective at transferring electrons and detecting tadalafil.
The Electrochemical Mechanism of Tadalafil Detection
At the molecular level, tadalafil undergoes an irreversible two-electron, two-proton oxidation process, primarily at its indole nitrogen site. On a bare electrode, this reaction is sluggish and produces weak signals. On the MMT-CeO₂ modified surface, however, the reaction is accelerated.
CeO₂ nanoparticles play the role of catalytic mediators. Their ability to cycle between Ce³⁺ and Ce⁴⁺ creates a redox-active surface that promotes electron transfer. Meanwhile, MMT provides abundant adsorption sites, stabilizes the nanoparticles, and facilitates analyte access. The synergy ensures that tadalafil molecules are efficiently oxidized, producing strong, reliable signals.
The optimal conditions were found at pH 8.0, where tadalafil exists in its anionic form. At this pH, the molecule is more prone to oxidation, and the electrode achieves maximum sensitivity. Interestingly, the electrode displayed two distinct linear response ranges—0.005–0.1 μM and 0.1–9.9 μM—with a limit of detection (LOD) as low as 1.97 × 10⁻¹⁰ M. To translate: this sensor can detect tadalafil at concentrations approaching one ten-billionth of a mole per liter. Such sensitivity outperforms many traditional techniques by orders of magnitude.
Analytical Performance: Numbers That Matter
For any analytical technique, numbers speak louder than theory. The modified electrode achieved a sensitivity of 3916 μA μM⁻¹ cm⁻², ranking it among the most sensitive tadalafil sensors reported to date. Stability tests showed that the electrode retained 98% of its activity after 30 days, demonstrating robustness and reusability.
Selectivity was another strength. The sensor showed minimal interference from common biological compounds such as ascorbic acid, uric acid, and dopamine, as well as from metal ions. This is crucial, since real-world samples like serum and urine are chemically crowded environments where false signals can easily arise.
Accuracy was confirmed through recovery experiments. Pharmaceutical tablets labeled to contain 5 mg or 20 mg of tadalafil were tested, and the measured values aligned closely with the expected content. Spiked human serum and urine samples yielded recovery rates of 97–104%, indicating the method’s reliability in both clinical and pharmaceutical contexts.
When compared with HPLC, LC-MS/MS, and previously reported electrochemical methods, this sensor excelled in sensitivity, simplicity, cost, and speed. It may not replace mass spectrometry for high-precision pharmacokinetics, but for quality control and routine screening, it is a game-changer.
Applications in Real Samples
The true test of any sensor lies in real-world application. In pharmaceutical quality control, the electrode successfully detected tadalafil in commercial formulations, confirming dosage accuracy and exposing the potential to catch counterfeit or substandard products.
In biological contexts, the sensor performed admirably. Human serum and urine samples spiked with tadalafil were analyzed directly, with minimal pre-treatment. The electrode delivered precise results, suggesting its potential use in clinical monitoring, therapeutic drug management, and even anti-doping testing.
This versatility is particularly valuable. A single sensor platform that works across tablets, serum, and urine simplifies workflows and expands utility. It positions the MMT-CeO₂ electrode not only as a laboratory tool but also as a field-ready device.
Sustainability and Cost-Effectiveness
The emphasis on eco-friendliness is not mere lip service. Montmorillonite clay is abundant, inexpensive, and environmentally benign. Cerium oxide nanoparticles, while synthetic, are relatively low-cost and non-toxic compared with many other nanomaterials. The use of pencil graphite as the electrode base further lowers costs and simplifies fabrication.
Unlike disposable test strips or one-use sensors, the electrode can be regenerated by simple polishing, extending its lifespan and reducing waste. In a world increasingly concerned with sustainability, this aspect cannot be overlooked. It allows laboratories to maintain high analytical performance without contributing to excessive environmental burden.
Broader Implications for Pharmaceutical Analysis
The success of this sensor reflects broader trends in analytical science. First, it demonstrates that eco-friendly materials can achieve performance on par with—or even superior to—traditional high-cost methods. Second, it highlights the power of nanotechnology to revolutionize everyday tools. And third, it suggests a future where pharmaceutical quality control is not confined to central laboratories but distributed across supply chains, clinics, and even pharmacies.
Imagine a pharmacist verifying the authenticity of tadalafil tablets before dispensing them, or a clinician monitoring patient compliance in real time. Such scenarios are no longer speculative—they are within reach if sensors like the MMT-CeO₂ electrode are scaled and commercialized.
Limitations and Future Directions
No technology is perfect. While the MMT-CeO₂ electrode shows remarkable sensitivity and stability, it remains confined to proof-of-concept research. Large-scale manufacturing, regulatory validation, and long-term field testing are needed before clinical deployment. Furthermore, the reproducibility of nanoparticle synthesis and electrode modification must be carefully standardized.
Another limitation is scope. While effective for tadalafil, the electrode may not be directly transferable to other drugs without optimization. Expanding the platform to cover a broader range of pharmaceuticals will be a key step in maximizing impact.
Finally, integration into portable, user-friendly devices remains a challenge. A sensor is only as good as its interface; unless coupled with simple readers and digital platforms, it risks remaining trapped in the laboratory.
Conclusion
The development of the MMT-CeO₂ NPs/CPG-PE sensor marks a significant advance in tadalafil detection. By blending the catalytic properties of cerium oxide nanoparticles with the structural advantages of montmorillonite clay, researchers have created a low-cost, eco-friendly, and ultrasensitive electrode. Its performance in pharmaceutical formulations and biological fluids demonstrates both robustness and versatility.
Beyond tadalafil, the principles demonstrated here—nanocomposite modification, eco-friendly materials, and electrochemical detection—could inspire a new generation of analytical tools. These sensors promise not only to improve drug quality assurance but also to democratize access to high-performance pharmaceutical analysis. In a global landscape where counterfeit drugs remain a persistent threat, innovations like this may prove as life-saving as the medications they monitor.
FAQ
1. Why is ultrasensitive detection of tadalafil necessary?
Because counterfeit and substandard formulations are common, and precise dosing is critical for efficacy and safety. Ultra-trace detection ensures authenticity, supports clinical monitoring, and aids pharmacokinetic research.
2. How does this sensor compare with traditional methods like HPLC or LC-MS/MS?
It offers comparable or greater sensitivity, lower cost, faster results, and simpler operation. While it cannot entirely replace mass spectrometry in advanced research, it is ideal for quality control and field testing.
3. Could this technology be applied to drugs other than tadalafil?
Yes. The platform is adaptable. With appropriate modification, similar nanocomposite electrodes could be developed for detecting a wide range of pharmaceuticals and even environmental contaminants.