Introduction
Nephrotic syndrome remains one of the most formidable challenges in nephrology. Characterized by severe proteinuria, hypoalbuminemia, edema, and hyperlipidemia, it is less a single disease than a final common pathway of glomerular injury. At its heart lies the podocyte, a highly specialized epithelial cell lining the glomerular basement membrane. Damage to podocytes is a decisive event in the initiation and progression of nephrotic syndrome, and their loss often seals the fate of patients toward chronic kidney disease (CKD) and ultimately end-stage kidney disease (ESKD).
For decades, therapy has centered around immunosuppressive regimens—glucocorticoids, calcineurin inhibitors, alkylating agents. While effective for many, these treatments are marred by toxicity, incomplete efficacy, and the looming specter of steroid resistance. A new therapeutic vision is urgently needed, one that protects podocytes directly rather than waging an immunological war.
Enter tadalafil, a phosphodiesterase-5 inhibitor (PDE5i) more famous in popular culture for treating erectile dysfunction. Yet science has a history of surprising repurposings. PDE5 inhibitors modulate cyclic guanosine monophosphate (cGMP) signaling, a pathway known not only for vascular smooth muscle relaxation but also for cellular survival and barrier integrity. Could tadalafil stabilize podocytes and restore glomerular filtration barrier function? Recent experimental work says: possibly yes.
Podocyte Injury: The True Culprit Behind Proteinuria
The pathophysiology of nephrotic syndrome is rooted in podocyte biology. These cells extend intricate foot processes that interdigitate, forming the slit diaphragm, a protein-rich filtration structure ensuring that plasma proteins remain in the circulation while waste products pass into urine. Key slit diaphragm proteins—nephrin and podocin—act as both structural and signaling components, preserving the selectivity of the glomerular barrier.
When podocytes are injured—by immune attack, toxins, drugs, or genetic mutations—they undergo foot process effacement, detachment, or apoptosis. The slit diaphragm collapses, nephrin and podocin expression fall, and the glomerular barrier becomes leaky. Proteinuria follows, initially subtle but ultimately massive, stripping plasma of albumin, disturbing oncotic balance, and fueling edema.
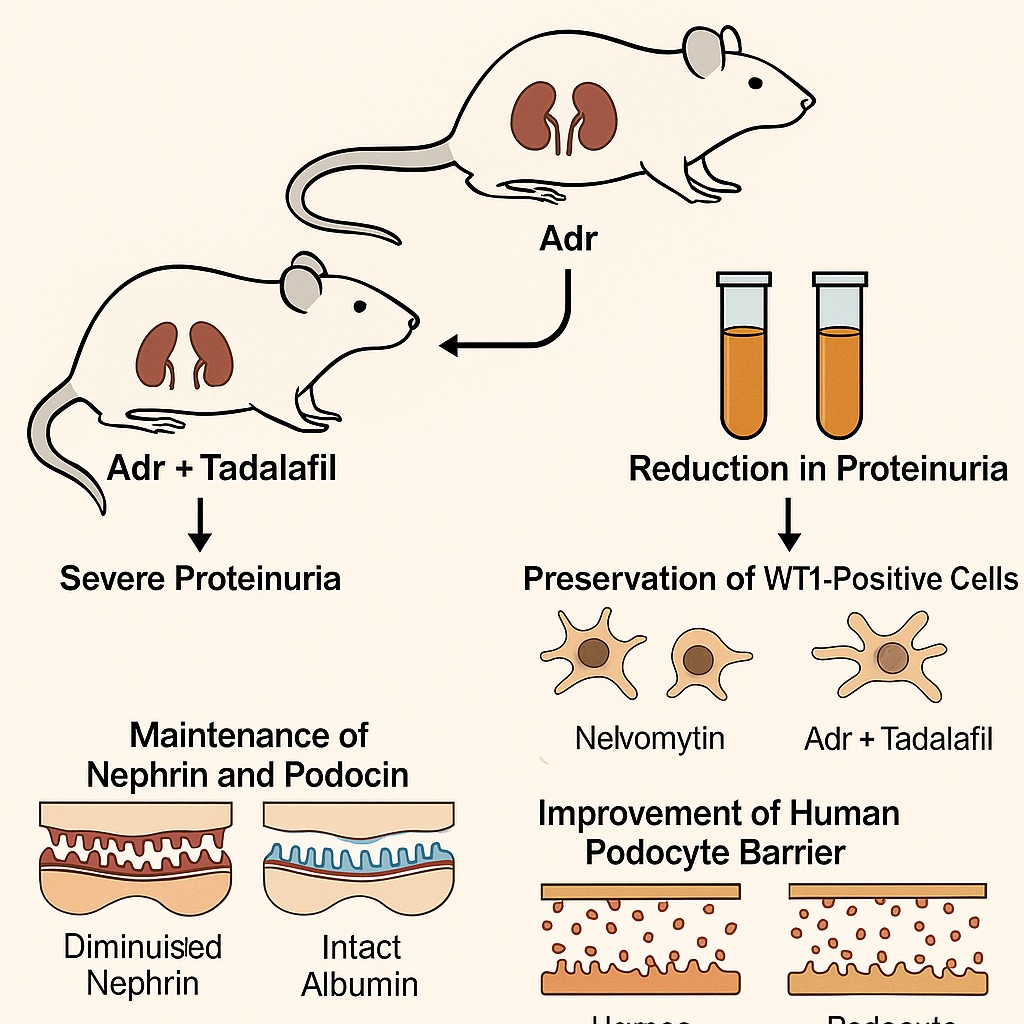
Adriamycin (doxorubicin), a chemotherapy agent, is notorious for inducing podocyte injury in experimental models. Rats exposed to adriamycin develop proteinuria, hypoalbuminemia, and histological hallmarks mimicking human nephrotic syndrome. This model has become the gold standard for preclinical evaluation of potential nephroprotective agents.
Tadalafil: From Vascular Smooth Muscle to Renal Podocytes
Tadalafil is best known as a long-acting PDE5 inhibitor. By preventing degradation of cGMP, it amplifies nitric oxide (NO) signaling, leading to vasodilation and improved perfusion. But cGMP signaling does not belong solely to smooth muscle cells. It also regulates cytoskeletal dynamics, apoptosis, and intercellular junctions in diverse cell types.
Preclinical studies hinted that PDE5 inhibitors could improve renal outcomes by attenuating glomerulosclerosis, preserving podocyte integrity, and improving renal blood flow. Tadalafil in particular, with its longer half-life and higher PDE5 selectivity compared to sildenafil or vardenafil, emerged as a strong candidate.
The study under discussion sought to clarify whether tadalafil acts directly on podocytes, preserving their survival and function under toxic assault. To this end, researchers used both an adriamycin-induced nephrotic syndrome rat model and human primary podocyte cultures. The results provide compelling, though preliminary, evidence of tadalafil’s podocyte-protective effects.
Experimental Model: From Rats to Human Cells
The experimental design included both in vivo and in vitro approaches.
Animal Model
Male Wistar rats were divided into four groups:
- Control + Vehicle
- Control + Tadalafil (10 mg/kg orally, 2 weeks)
- Adriamycin + Vehicle
- Adriamycin + Tadalafil
Renal function was assessed through proteinuria, serum albumin, creatinine, and blood urea nitrogen. Histological evaluation included Wilms’ tumor 1 (WT1) staining to quantify podocyte nuclei, and immunofluorescence to detect nephrin and podocin expression.
Human Podocyte Model
Primary human renal epithelial podocytes were cultured and exposed to adriamycin, with or without tadalafil. Endpoints included:
- Cell viability (CCK-8 assay)
- Albumin permeability (Transwell assay)
- PDE5 expression (PCR and Western blot)
- Pathway analysis with a PKG inhibitor (KT5823)
This dual-pronged design allowed the authors to test not only whether tadalafil attenuated proteinuria in vivo, but also whether it directly stabilized human podocytes in vitro.
Results: Tadalafil as a Guardian of the Filtration Barrier
The findings were striking.
- Reduction in Proteinuria
Adriamycin induced severe proteinuria in rats. Tadalafil treatment cut urinary protein excretion significantly, halving the protein-to-creatinine ratio compared to ADR + vehicle animals. - Preservation of Podocyte Number
WT1 staining revealed marked podocyte loss in ADR-treated rats. Tadalafil rescued podocyte counts, bringing them closer to control levels. - Maintenance of Nephrin and Podocin
ADR diminished nephrin and podocin expression along the glomerular basement membrane. Tadalafil preserved their expression, suggesting intact slit diaphragm architecture. - Improved Human Podocyte Viability
In cell culture, ADR reduced podocyte survival. Tadalafil improved viability, but this effect vanished when PKG was inhibited. This confirmed a cGMP–PKG–dependent mechanism. - Protection of Barrier Function
ADR increased albumin permeability across podocyte monolayers. Tadalafil attenuated this leakage, preserving barrier selectivity.
Together, these results strongly suggest that tadalafil exerts direct cytoprotective effects on podocytes, preventing functional collapse of the glomerular filtration barrier.
Mechanistic Insights: cGMP–PKG Pathway at Work
The involvement of the cGMP–PKG axis was elegantly demonstrated. PDE5 inhibition boosts cGMP, which activates protein kinase G (PKG). PKG in turn modulates cytoskeletal proteins, enhances cell survival signaling, and stabilizes intercellular junctions.
When PKG was inhibited with KT5823, tadalafil’s protective effect vanished. This indicates that the drug does not act through secondary hemodynamic changes but directly through intracellular signaling in podocytes.
Interestingly, other studies suggest PDE5 inhibitors may also act through PKG-independent cGMP pathways, including modulation of calcium handling and other kinases. Whether these contribute to podocyte protection remains a question for future research.
Clinical Relevance: Why Should Nephrologists Care?
Proteinuria is more than a symptom—it is a driver of CKD progression. Every gram of urinary protein lost accelerates renal decline. Therapies that reduce proteinuria, even without altering systemic hemodynamics, are considered renoprotective.
Current mainstays—RAAS blockade and SGLT2 inhibitors—demonstrate this principle. Could PDE5 inhibitors join this list? The evidence from this study is promising: tadalafil reduced proteinuria in rats and preserved podocyte function in human cells.
Furthermore, PDE5 inhibitors are already widely used in clinical practice, with established safety profiles in cardiovascular disease, pulmonary hypertension, and erectile dysfunction. Repurposing them for kidney protection would be a rational, cost-effective strategy.
Limitations and Open Questions
As compelling as these findings are, several caveats must be acknowledged:
- Species differences: Rat pharmacokinetics differ from human; extrapolation must be cautious.
- Single-agent focus: The study did not compare tadalafil to other PDE5 inhibitors like sildenafil or vardenafil. Class effects remain uncertain.
- Timing of treatment: Tadalafil was given simultaneously with injury induction. Clinical nephrotic syndrome patients present after injury has occurred; efficacy in that setting remains untested.
- Small sample size: Animal numbers were limited, and long-term outcomes (fibrosis, progression to CKD) were not studied.
Future studies must address these limitations with longer treatment, post-injury intervention, and comparative PDE5i trials.
The Bigger Picture: Podocyte-Centric Therapies
This research reflects a paradigm shift: protecting the podocyte itself rather than merely managing downstream consequences of its injury. Similar strategies are under exploration with TRPC6 inhibitors, endothelin antagonists, and cytoskeletal stabilizers. PDE5 inhibition may prove to be a practical, immediately available addition to this armamentarium.
Moreover, PDE5 inhibitors may offer dual benefits—improving both renal outcomes and vascular health. Given the tight interplay between kidney disease and cardiovascular morbidity, such dual-action drugs are particularly attractive.
Conclusion
The study provides robust experimental evidence that tadalafil protects podocytes and maintains the glomerular filtration barrier in adriamycin-induced nephrotic syndrome. By reducing proteinuria, preserving nephrin and podocin, and improving podocyte viability through the cGMP–PKG pathway, tadalafil demonstrates renoprotective potential beyond its established urological applications.
While translation to clinical practice will require careful validation, the concept is tantalizing: a familiar, well-tolerated drug could be repurposed to tackle one of nephrology’s most stubborn challenges. For now, tadalafil remains better known in the context of date nights than dialysis prevention—but perhaps not for long.
FAQ
1. How does tadalafil protect the kidneys?
Tadalafil inhibits PDE5, increasing cGMP and activating PKG signaling. This pathway enhances podocyte survival, maintains slit diaphragm proteins (nephrin and podocin), and reduces albumin leakage, thereby preserving kidney filtration function.
2. Does this mean tadalafil can be prescribed to patients with nephrotic syndrome today?
Not yet. Current data come from animal and cell studies. Clinical trials are needed to confirm efficacy and safety in patients with nephrotic syndrome or CKD.
3. Is this effect unique to tadalafil, or do other PDE5 inhibitors work similarly?
Other PDE5 inhibitors (sildenafil, vardenafil) may also protect podocytes, but tadalafil’s long half-life and high PDE5 selectivity may offer unique advantages. Comparative studies are still needed.