Introduction
Nonsteroidal anti-inflammatory drugs (NSAIDs) are indispensable in modern medicine, yet they remain double-edged swords. While they alleviate pain, fever, and inflammation, they notoriously compromise gastric integrity, often culminating in gastric ulcers. Among these drugs, indomethacin is both effective and infamous—effective in controlling inflammation but infamous for causing severe gastrointestinal toxicity. For decades, clinicians have wrestled with balancing NSAID efficacy against their adverse effects, often resorting to acid-suppressive therapies such as proton pump inhibitors (PPIs).
Yet, PPIs and related drugs are far from perfect. Their long-term use is associated with risks ranging from nutrient malabsorption to renal impairment. More importantly, they address the symptom—acid secretion—rather than the underlying mechanisms of gastric mucosal injury. To advance therapy, researchers have looked beyond acid suppression and toward cellular and molecular mediators of gastric protection.
A surprising contender has emerged: tadalafil, a phosphodiesterase-5 inhibitor (PDE5i) primarily marketed for erectile dysfunction and pulmonary hypertension. Recent experimental evidence suggests tadalafil may confer gastroprotective effects by modulating oxidative stress, inflammation, and nitric oxide (NO) signaling. The central players in this story are asymmetric dimethylarginine (ADMA), dimethylarginine dimethylaminohydrolase-1 (DDAH-1), and the inducible versus endothelial nitric oxide synthase (iNOS/eNOS) pathways. This article delves into the mechanistic and clinical implications of this novel gastroprotective role for tadalafil.
NSAID-Induced Gastric Injury: Mechanistic Insights
The pathogenesis of NSAID-induced ulcers is multifaceted, though three mechanisms stand out.
First, NSAIDs inhibit cyclooxygenase (COX) enzymes, reducing prostaglandin E2 (PGE-2) synthesis. Prostaglandins maintain mucosal integrity by stimulating mucus and bicarbonate secretion, enhancing blood flow, and modulating leukocyte activity. Their loss tips the balance toward injury.
Second, NSAIDs amplify inflammatory cascades. Tumor necrosis factor-alpha (TNF-α) and other proinflammatory cytokines are upregulated, enhancing leukocyte adhesion, infiltration, and oxidative burst. The neutrophils that should defend tissues instead occlude microvessels and exacerbate injury.
Third, NSAIDs disrupt redox balance. Reactive oxygen species (ROS) surge, glutathione (GSH) is depleted, and superoxide dismutase (SOD) activity falls. The result is unchecked lipid peroxidation and mucosal cell apoptosis.
Nitric oxide synthase (NOS) isoforms further complicate the picture. While iNOS-derived NO often drives inflammatory injury, eNOS-derived NO supports mucosal perfusion and defense. NSAIDs push this equilibrium unfavorably, raising iNOS and suppressing eNOS expression.
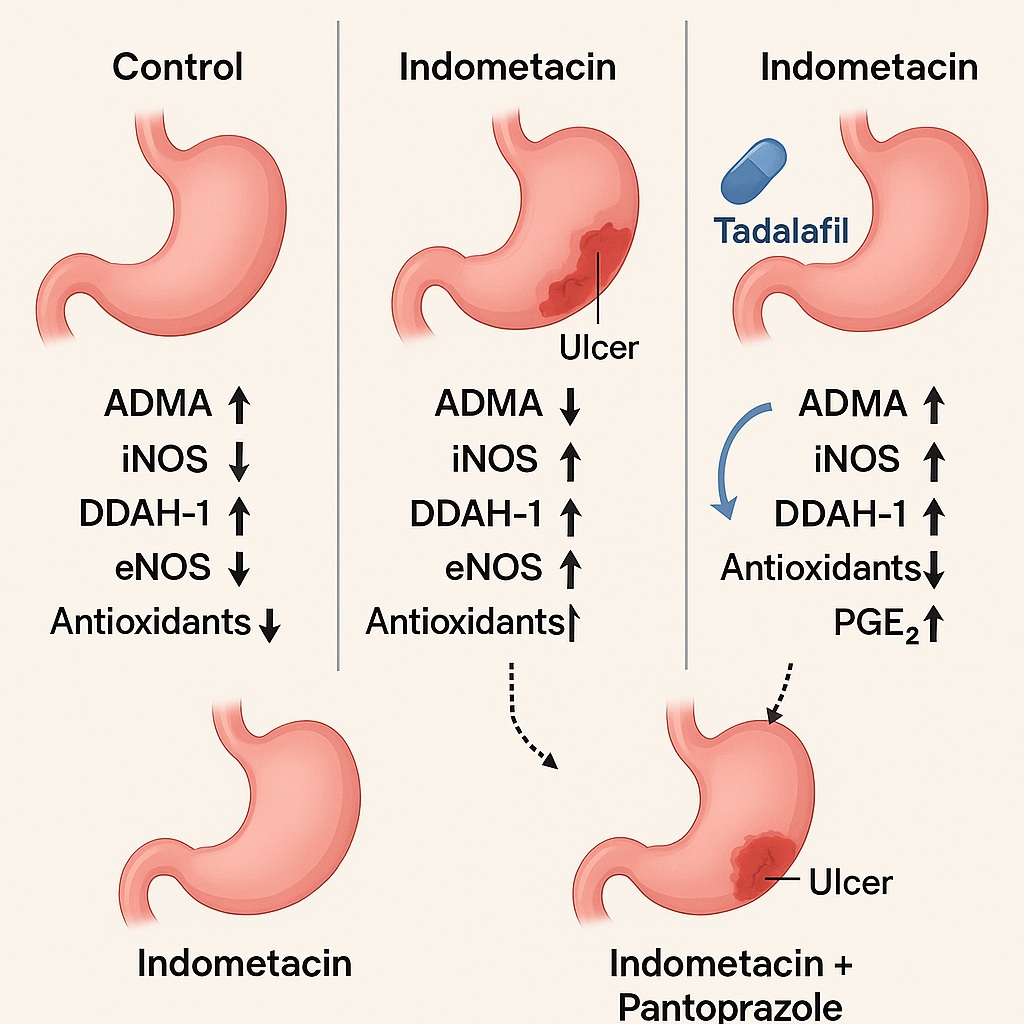
Finally, ADMA, an endogenous inhibitor of NOS, rises during NSAID-induced injury. ADMA promotes oxidative stress, impairs endothelial function, and fuels apoptosis. DDAH-1, the enzyme tasked with ADMA degradation, becomes inhibited under oxidative stress, worsening the imbalance.
Together, these pathways paint a grim portrait: reduced prostaglandins, exaggerated cytokine activity, excessive ROS, pathological NO signaling, and accumulation of ADMA. Small wonder that NSAID ulcers remain a stubborn clinical challenge.
Tadalafil: Mechanism Beyond Vasodilation
Tadalafil is a selective inhibitor of PDE5, the enzyme responsible for degrading cGMP. By inhibiting PDE5, tadalafil raises intracellular cGMP, enhancing protein kinase G (PKG) activation. In vascular smooth muscle, this translates into relaxation and vasodilation.
But cGMP signaling is not restricted to erectile tissue. It influences antioxidant responses, modulates apoptosis, and interacts with inflammatory mediators across organ systems. Specifically, PDE5 inhibition suppresses NADPH oxidase activity, reducing ROS generation. It also enhances the activity of antioxidant enzymes such as SOD and replenishes glutathione pools.
In addition, PDE5 inhibition can reshape NO signaling by promoting eNOS activity while suppressing iNOS expression. The result is a favorable shift toward protective rather than injurious NO pathways. Finally, by modulating ADMA/DDAH-1 dynamics, tadalafil reduces the toxic burden of ADMA, reinforcing endothelial defense and limiting oxidative stress.
This biochemical repertoire positions tadalafil as an unexpected yet logical candidate for gastric protection.
Experimental Design: Indomethacin Meets Tadalafil
To test this hypothesis, researchers employed a rat model of indomethacin-induced gastric ulceration. The study included five groups:
- Control rats
- Tadalafil alone (10 mg/kg, oral)
- Indomethacin alone (60 mg/kg, single oral dose)
- Indomethacin + pantoprazole (standard anti-ulcer comparator)
- Indomethacin + tadalafil
Endpoints included macroscopic ulcer index (UI), preventive index (PI), histopathological assessment, oxidative stress biomarkers (GSH, SOD, NO), inflammatory mediators (TNF-α, COX-2, PGE-2), ADMA/DDAH-1 expression, and iNOS/eNOS protein levels.
The design provided not only functional outcomes but also mechanistic clarity, dissecting the relative contribution of oxidative, inflammatory, and NO-dependent pathways.
Results: Tadalafil as a Gastric Guardian
The findings were both consistent and compelling.
Reduction in Ulcer Severity
Indomethacin produced pronounced gastric ulceration, with elevated UI and diminished PI. Pretreatment with tadalafil dramatically reduced ulcer scores, restoring PI nearly to normal levels. In efficacy, tadalafil matched pantoprazole, the benchmark therapy.
Histopathological Protection
Microscopic examination told the same story. Indomethacin induced mucosal necrosis, submucosal edema, vascular congestion, and heavy inflammatory infiltration. Rats treated with tadalafil exhibited intact mucosal structures, minimal edema, and only mild inflammatory changes.
Oxidative Stress Attenuation
Indomethacin lowered GSH and SOD while spiking gastric NO levels. Tadalafil reversed these changes, significantly raising antioxidant defenses and suppressing NO overproduction. Pantoprazole achieved similar results, confirming that tadalafil’s protection was not a statistical fluke.
ADMA/DDAH-1 Modulation
Indomethacin raised gastric ADMA nearly twofold while depressing DDAH-1 activity. Tadalafil restored DDAH-1 and reduced ADMA levels by half, suggesting a novel protective mechanism not typically associated with standard acid-suppressive therapies.
Inflammatory Marker Rebalancing
TNF-α surged in indomethacin-treated rats, while COX-2 and PGE-2 plummeted. Tadalafil sharply lowered TNF-α while boosting COX-2 and restoring PGE-2, echoing pantoprazole’s effects.
iNOS/eNOS Signaling Correction
Indomethacin upregulated iNOS and suppressed eNOS, tipping NO signaling toward pathology. Tadalafil reversed this imbalance—reducing iNOS expression while enhancing eNOS. The outcome was a shift back toward protective NO biology.
Mechanistic Interpretation
Tadalafil’s gastroprotective effect emerges from a triad of mechanisms:
- Antioxidant reinforcement: By increasing GSH and SOD while suppressing NADPH oxidase-driven ROS, tadalafil restores redox balance.
- Anti-inflammatory modulation: Through suppression of TNF-α and recovery of COX-2/PGE-2, tadalafil dampens pathological inflammation.
- Nitric oxide signaling correction: By lowering iNOS and enhancing eNOS, and by modulating ADMA/DDAH-1, tadalafil promotes protective NO dynamics over injurious ones.
This multi-pronged action positions tadalafil not merely as an acid-suppressive substitute but as a true mucosal protector addressing fundamental pathogenic drivers.
Clinical Implications
The translation of these findings could be transformative. NSAID-induced gastric injury is ubiquitous, particularly among the elderly and patients with chronic pain or arthritis. Current therapies, centered around PPIs, do not address oxidative stress, inflammation, or NO imbalance.
Tadalafil offers several potential advantages:
- It is already clinically approved with a well-characterized safety profile.
- It has a longer half-life than sildenafil, allowing once-daily dosing.
- It targets multiple pathways simultaneously, offering more comprehensive mucosal protection.
One might imagine a future where high-risk NSAID users receive dual therapy: NSAID + tadalafil to minimize ulcer risk. Of course, enthusiasm must be tempered until clinical trials validate these preclinical observations.
Limitations and Future Directions
Despite its promise, this study is not the final word. Several limitations remain:
- Species difference: Rat models do not perfectly mimic human gastric physiology.
- Preventive rather than therapeutic design: Tadalafil was given before indomethacin. Its efficacy in established ulcers remains unknown.
- Comparative scope: Only pantoprazole was used as comparator. It remains unclear whether tadalafil synergizes with PPIs or competes with them.
- Safety considerations: While tadalafil is safe at standard doses, long-term use in patients with polypharmacy requires careful study.
Future trials should evaluate tadalafil in diverse ulcer models, explore combination therapies, and eventually translate to clinical studies in NSAID users.
Conclusion
The study convincingly demonstrates that tadalafil protects gastric mucosa against indomethacin-induced injury. Through modulation of ADMA/DDAH-1 and iNOS/eNOS signaling, along with suppression of oxidative stress and inflammation, tadalafil emerges as a candidate for repurposing in gastroprotection.
If clinical translation succeeds, tadalafil may one day join the therapeutic armamentarium not only for erectile dysfunction and pulmonary hypertension but also for NSAID-associated gastric injury. A PDE5 inhibitor in the gastroenterologist’s toolkit might sound improbable today—but then again, so did the notion of using beta-blockers for heart failure decades ago.
FAQ
1. Can tadalafil replace proton pump inhibitors for NSAID-induced ulcers?
Not at present. While tadalafil shows gastroprotective effects in animal models, clinical trials in humans are lacking. PPIs remain the gold standard, though tadalafil could become an adjunct in the future.
2. What makes tadalafil different from other ulcer therapies?
Unlike acid-suppressive drugs, tadalafil directly targets oxidative stress, inflammation, and NO signaling, addressing the root causes of mucosal injury rather than just reducing acid.
3. Are there risks in using tadalafil for gastric protection?
Tadalafil is generally safe but may cause headache, flushing, or hypotension. Its long-term safety in patients using NSAIDs chronically requires thorough clinical investigation before routine use can be recommended.