Benign prostatic hyperplasia (BPH) is among the most prevalent urological conditions in aging men, affecting up to 75% of individuals over 80 years old. For decades, its management has relied on familiar therapeutic pillars: α-adrenoceptor antagonists, 5-α reductase inhibitors, PDE5 inhibitors, and, when required, surgical intervention. While these treatments offer substantial benefit, they remain imperfect. Many patients experience incomplete symptom relief, disease progression, or intolerance to current medications. Meanwhile, a deeper understanding of BPH pathophysiology has revealed a biological complexity far exceeding simple prostatic overgrowth.
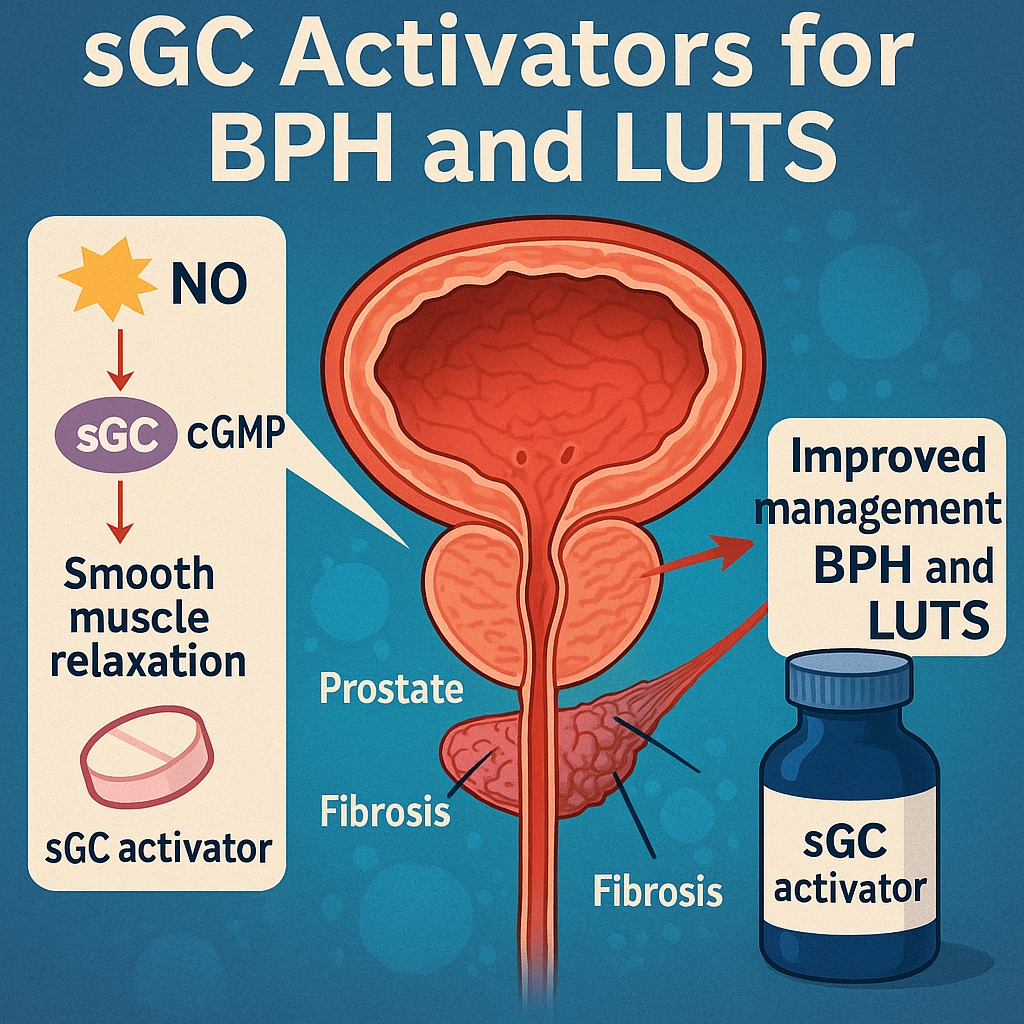
Against this backdrop, the workshop summarized in the referenced paper introduces a compelling new avenue in the management of BPH and associated lower urinary tract symptoms (LUTS): modulation of the nitric oxide (NO)–soluble guanylate cyclase (sGC)–cyclic GMP pathway. This signaling cascade, well known in vascular biology, is increasingly recognized as a central regulator of prostatic smooth muscle tone, neurotransmission, and fibrosis — all major contributors to BPH/LUTS. Importantly, innovative agents such as sGC activators are emerging with the potential to succeed where PDE5 inhibitors fall short, particularly under conditions of oxidative stress.
This article synthesizes the scientific rationale, experimental evidence, and therapeutic implications of sGC activators—most notably cinaciguat—and explores how they may reshape future medical treatment of BPH and bladder outlet obstruction (BOO). The discussion integrates mechanistic insights, animal model data, and broader physiologic context, forming a coherent perspective on the potential for a new antifibrotic, urofunctional therapeutic class.
Understanding BPH Beyond Hyperplasia: The Triad of Smooth Muscle Tone, Fibrosis, and Bladder Remodeling
The workshop emphasizes that BPH is not simply a disease of prostate enlargement. While hyperplasia and increased glandular volume contribute to urethral narrowing, this represents only one aspect of a broader pathophysiologic constellation. Increasing evidence supports a three-factor model of BPH-related obstruction: hyperplastic growth, increased adrenergic smooth muscle tone, and progressive fibrosis of both prostate and bladder tissues. Together, these factors impair urethral compliance, elevate outlet resistance, and disrupt bladder mechanics.
The data presented reinforce the significance of smooth muscle contraction in determining intraurethral pressure. Approximately 40% of total urethral pressure can be attributed to α-adrenergic–mediated prostatic stromal tone, with the remainder derived from static compression due to glandular enlargement. This dual mechanism explains why α-blockers often provide rapid symptom relief, while 5-α reductase inhibitors offer slower structural benefit. Yet neither effectively addresses fibrotic remodeling, an increasingly recognized driver of long-term LUT dysfunction.
Indeed, fibrosis alters not only the prostate but also the bladder itself. According to the physiologic model highlighted in the workshop, BOO initiates a sequence of bladder changes: early hypertrophy, compensatory hypercontractility, eventual detrusor overactivity, and later decompensation. The histologic images on pages 3–4 show increased collagen deposition and decreased tissue compliance in aged mouse bladder walls, demonstrating how fibrosis reshapes bladder biomechanics. This fibrosis is not merely a byproduct of obstruction; it reflects deeper alterations in signaling milieu, particularly involving TGF-β and impaired NO–sGC–cGMP activity.
Thus, treating BPH requires more than suppressing growth or relaxing smooth muscle. It requires a therapeutic approach capable of addressing fibrosis — a goal that current drug classes do not adequately achieve.
Why the NO–sGC–cGMP Pathway Matters in BPH and LUTS
Nitric oxide is an essential neuromodulator and smooth muscle relaxant within the lower urinary tract. It exerts its biologic effects primarily through activation of soluble guanylate cyclase, which catalyzes cGMP production. cGMP, in turn, reduces calcium sensitivity of the contractile apparatus, decreases phosphorylation of myosin light chains, and facilitates smooth muscle relaxation.
In prostate tissue, cGMP decreases stromal tone. In bladder tissue, it modulates detrusor contractility and influences neurotransmitter release from both sympathetic and parasympathetic nerves. This dual effect positions the NO–sGC–cGMP axis as a central regulator of lower urinary tract physiology. Pedagogically speaking, it is the conductor of the pelvic orchestra — and when oxidative stress disrupts its function, the performance deteriorates markedly.
The problem arises when sGC becomes oxidized, rendering it unresponsive to endogenous nitric oxide. As explained on page 1 and illustrated in Fig. 1A, oxidative stress—common in aging tissues—can downregulate CYB5R3, the reductase responsible for maintaining sGC in its ferrous, NO-sensitive state. Once oxidized, sGC becomes inactive, cGMP levels fall, and smooth muscle tone increases. Additionally, reduced cGMP signaling promotes TGF-β-mediated fibroblast-to-myofibroblast transition, a key driver of prostatic and bladder fibrosis (Fig. 1B).
This mechanistic insight explains several clinical phenomena:
• why PDE5 inhibitors help some patients but not others;
• why older men with oxidative stress show limited response to tadalafil;
• why fibrosis progresses even in men receiving standard BPH therapy.
In short, when sGC itself is dysfunctional, stimulating PDE5 inhibition is akin to pressing the gas pedal with no engine running.
sGC Activators: A Mechanistically Targeted Solution for Oxidized Enzyme States
Unlike PDE5 inhibitors, sGC activators such as cinaciguat bind directly to the heme site of oxidized or heme-free sGC, restoring its ability to generate cGMP independently of NO. This means that even in tissues with oxidative damage — common in aged prostates and bladders — sGC activators remain effective. By bypassing the need for nitric oxide, these agents restore downstream signaling that modulates muscle tone, neurotransmission, and fibrotic remodeling.
The workshop presented compelling preclinical data supporting cinaciguat’s multi-level benefits. In aged mouse models exhibiting BPH-like obstruction, cinaciguat reversed both functional and histologic abnormalities. Telemetric cystometry recordings on page 3 (Fig. 2A–C) demonstrate markedly improved bladder pressure profiles and increased voided volumes after two weeks of treatment. In these same animals, cinaciguat did not substantially lower systemic blood pressure (Fig. 2D), reinforcing its favorable cardiovascular safety profile.
Histologic sections (Fig. 3) on page 3 reveal dramatic reversal of urethral constriction in aged mice. Collagen content decreased, luminal diameter increased, and prostate stromal hyperplasia diminished with treatment. These histologic improvements mirror human BPH pathology and suggest that sGC activation may offer structural disease modification rather than symptomatic relief alone.
In bladder tissue, cinaciguat reduced fibrosis and restored compliance, as shown on page 4 (Fig. 4A–D). Passive tension curves shifted toward youthful elasticity, demonstrating the agent’s antifibrotic effects at a functional level. Reversing bladder wall stiffness may have profound long-term benefit, as reduced compliance contributes significantly to LUTS severity and detrusor instability.
Taken together, these data position sGC activators as promising candidates for disease-modifying therapy in BPH/LUTS — a category sorely lacking in current pharmacology.
PNPase Inhibition and Oxidative Stress: A Complementary Therapeutic Avenue
Interestingly, the workshop also explored the role of purine nucleoside phosphorylase (PNPase) in generating oxidative metabolites that exacerbate LUT inflammation, nerve dysfunction, and tissue damage. Aging rodent bladders display increased intervoid intervals, reduced contractile efficiency, and altered collagen architecture. According to page 4 (Fig. 5–6), treatment with the PNPase inhibitor 8-aminoguanine (8-AG) restored bladder function and normalized collagen fiber structure.
The link between PNPase activity and oxidative stress intersects elegantly with sGC biology: by reducing oxidative stress, PNPase inhibition may preserve sGC function in its reduced, NO-responsive state. Thus, combining sGC activators or stimulators with agents that decrease oxidative injury could theoretically yield synergistic benefits.
Although clinical correlation remains hypothetical, the convergence of these pathways reflects a broader principle: LUT dysfunction in aging is fundamentally a disease of oxidative imbalance, fibrotic remodeling, and neuromuscular dysregulation. Each mechanistic insight opens the door for targeted therapy that moves beyond simple relaxation of prostate smooth muscle.
cGMP-Dependent Modulation of Neurotransmission: Refining Bladder Function Through Signaling Precision
A particularly intriguing element of the workshop involved the differential effect of cGMP elevation on neurotransmitter release. As shown in Fig. 7 on page 5, agents that raise cGMP — including cinaciguat and sildenafil — significantly reduce ATP release from bladder parasympathetic fibers during low-frequency stimulation while leaving acetylcholine (ACh) release relatively unchanged.
This distinction matters clinically. ATP acts as a powerful excitatory cotransmitter in detrusor overactivity, contributing to urgency and frequency. ACh, however, remains the primary physiologic driver of contraction during normal voiding. Therefore, selectively inhibiting ATP release may reduce urgency without impairing coordinated voiding. This pharmacodynamic finesse distinguishes sGC activators from antimuscarinics, which globally suppress cholinergic signaling and often worsen emptying.
The ability to modulate pathological, but not physiological, neurotransmission makes sGC activators a uniquely appealing therapeutic class. They do not merely quiet an overactive bladder — they refine its communication, preserving functional integrity while reducing dysfunctional excitability.
Potential Clinical Applications and Future Directions
Although human data are still emerging, the mechanistic and animal evidence suggests several potential applications for sGC activators in BPH/LUTS management:
- treatment of patients refractory to PDE5 inhibitors, particularly older men with high oxidative burden;
- reduction of prostate and bladder fibrosis, offering disease modification rather than symptomatic palliation;
- improvement of bladder compliance and reduction of detrusor overactivity without impeding normal voiding;
- combination therapy alongside PDE5 inhibitors, α-blockers, or PNPase inhibitors for synergistic benefits.
The antifibrotic effect alone sets this class apart. No currently approved BPH medication demonstrates consistent reversal of stromal or bladder fibrosis. Even 5-α reductase inhibitors primarily influence epithelial growth rather than stromal remodeling.
The challenges ahead include translating animal findings to human studies, determining optimal dosing strategies, and identifying biomarkers of oxidative sGC inactivation. Nonetheless, the foundational science is compelling, and the therapeutic rationale is strong.
Conclusion
The exploration of soluble guanylate cyclase activators marks an exciting and scientifically grounded advancement in the management of benign prostatic hyperplasia and LUTS. As shown in the workshop summarized in the paper , sGC activators such as cinaciguat hold potential to correct underlying molecular dysfunction, restore cGMP signaling, reduce fibrosis, improve urethral patency, and normalize bladder mechanics. Their ability to function under oxidative stress distinguishes them from PDE5 inhibitors and positions them as promising candidates for patients who fail existing therapies.
Equally important is the recognition that BPH and LUTS are not merely obstructive phenomena but complex disorders of neuromuscular regulation, extracellular matrix remodeling, and aging-related metabolic imbalance. Targeting these pathways with mechanistically sophisticated agents offers a path toward genuine disease modification.
The future of BPH treatment may well include sGC activators not as replacements for current options but as critical components in a multimodal, personalized therapeutic strategy. As human trials progress, these agents may redefine how clinicians approach the aging lower urinary tract — with deeper insight, greater precision, and renewed optimism.
FAQ
1. Why might sGC activators work when PDE5 inhibitors fail?
Because PDE5 inhibitors require functioning NO-responsive sGC to increase cGMP. In aging or oxidative stress, sGC becomes oxidized and unresponsive. sGC activators bypass this limitation by directly activating oxidized or heme-free sGC.
2. Can sGC activators reverse prostate or bladder fibrosis?
Preclinical studies show significant antifibrotic effects, including reduced collagen deposition and improved tissue compliance. While human data are pending, animal models strongly suggest disease-modifying potential.
3. Are sGC activators likely to replace existing BPH medications?
Not in the near term. They are more likely to complement existing therapies, particularly in patients with refractory symptoms, high oxidative stress, or significant bladder dysfunction.