Phosphodiesterase-5 (PDE5) inhibitors occupy an unusual space in pharmacology. Created for cardiovascular indications, immortalized by their role in treating erectile dysfunction, and now increasingly repositioned in metabolic medicine, these agents are again under scrutiny—this time for their ability to influence haemoglobin A1c (HbA1c). The idea that a drug best known for restoring sexual function may also improve glycaemic control has long hovered at the edge of scientific curiosity, supported by mechanistic speculation yet lacking cohesive clinical evidence.
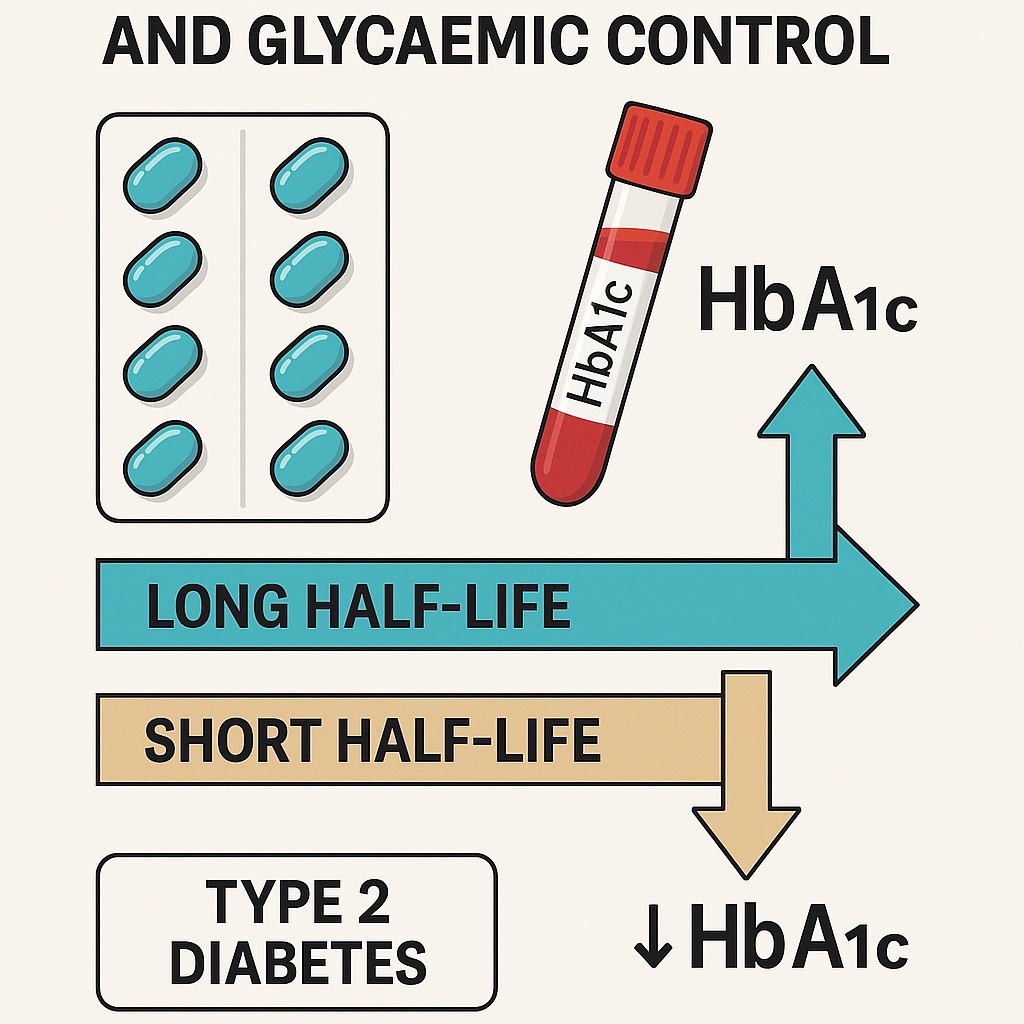
This recently published systematic review and meta-analysis finally brings clarity. By analyzing randomized controlled trials (RCTs) involving 1083 participants across 13 studies, the authors compare the glycaemic effects of long half-life PDE5 inhibitors (notably tadalafil and PF-00489791, with half-lives of 15–17.5 hours) versus short half-life PDE5 inhibitors (sildenafil and avanafil, ~4 hours). Their conclusion is deceptively simple: the half-life of a PDE5 inhibitor appears to determine whether it meaningfully reduces HbA1c.
While short half-life drugs show no appreciable effect, long half-life inhibitors demonstrate statistically significant and potentially clinically relevant HbA1c reductions—comparable to oral antihyperglycaemic medications at similar baseline glycaemic levels.
In the following article, we will explore the mechanisms, statistical findings, clinical implications, and research opportunities uncovered by this ambitious meta-analysis, rendering them into an accessible, coherent, and compelling narrative for medical professionals.
Understanding the Metabolic Promise of PDE5 Inhibitors
The hypothesis that PDE5 inhibitors might influence glucose metabolism is not new. Mechanistic studies show that these drugs enhance cGMP signaling, which can stimulate glucose uptake independently of insulin through PI3K/AKT pathways and improve endothelial function—both impaired in type 2 diabetes.
Several biochemical insights help explain why this class of medications may lower HbA1c. Improved nitric oxide signaling reduces oxidative stress and chronic inflammation, two hallmarks of insulin resistance. Enhanced endothelial function, in turn, promotes more efficient glucose transport into peripheral tissues, particularly skeletal muscle. Early translational investigations have revealed that intermediates in these signaling pathways—from soluble guanylate cyclase stimulators to PI3K modulators—have measurable effects on glycaemic parameters, adding mechanistic credibility to the PDE5 story.
Despite these findings, earlier clinical meta-analyses remained inconclusive. A 2016 meta-analysis, limited to sildenafil studies alone, found no significant HbA1c change. The crucial missing pieces, it turns out, were long half-life PDE5 inhibitors—an omission this new meta-analysis addresses with precision.
The Importance of Drug Half-Life: Why Duration Matters
The cornerstone of this study is its distinction between long and short half-life PDE5 inhibitors. This seemingly simple pharmacokinetic characteristic carries major physiological implications.
Short half-life drugs such as sildenafil are metabolized quickly—so quickly that their active window may be largely absent during sleep, a time when glycaemic fluctuations are meaningful. In contrast, long half-life agents provide steady plasma exposure and more consistent modulation of glucose-related pathways over 24 hours.
The study’s authors argue that this continuity is vital for measurable HbA1c reductions. This explanation aligns well with Michaelis–Menten modeling of HbA1c kinetics: because HbA1c reflects glycaemia over 2–3 months, medications that exert only intermittent effects are unlikely to influence observed HbA1c levels in short- or medium-term trials.
Thus, half-life appears to be not a trivial pharmacological detail, but the central determinant of metabolic impact.
How the Meta-Analysis Was Conducted
Adhering to PRISMA and Cochrane standards, the authors performed a global literature search across six major databases and registries. Of 1096 records assessed, 13 RCTs met the inclusion criteria—each enrolling adults with elevated HbA1c (>6%) and involving at least four weeks of PDE5 inhibitor treatment versus placebo or control.
Studies included a variety of geographic regions, dosing regimens, and participant characteristics. Importantly, only chronic, not on-demand, use of PDE5 inhibitors was evaluated. The database search revealed an important gap: many PDE5 trials in diabetic populations failed to measure HbA1c at all, based on the assumption that PDE5 inhibitors have no metabolic effect. This analysis challenges that assumption.
The authors extracted HbA1c changes from text, tables, and figures and performed inverse-variance random-effects meta-analysis. Given the broad diversity of study designs, heterogeneity was expected and rigorously assessed. Sensitivity analyses addressed trial duration, baseline HbA1c, methodological differences, and potential outliers.
What the Evidence Shows: A Tale of Two Pharmacokinetic Profiles
Long Half-Life Inhibitors: A Meaningful and Statistically Significant Reduction
Across seven RCTs involving 742 participants, long half-life inhibitors demonstrated a mean HbA1c reduction of −0.40% (95% CI −0.66 to −0.14; p = 0.002).
This decrease closely mirrors the −0.35% average reduction seen with standard oral antihyperglycaemic medications at similar baseline HbA1c levels (~7–7.9%).
These findings imply that long half-life PDE5 inhibitors, although not candidates for monotherapy, may produce clinically relevant improvements in glycaemic control—particularly when added to existing diabetes regimens.
The most remarkable effect appeared in trials including participants with high baseline HbA1c (~10%). The only available RCT in this category, Hegazy 2024, demonstrated a dramatic HbA1c reduction of −2.81%, while a non-RCT case series showed a reduction of −2.26%.
Both results fall squarely within the range expected for intensification with frontline diabetic therapies, making long half-life PDE5 inhibitors particularly promising for patients with poor baseline glycaemic control.
Short Half-Life Inhibitors: No Improvement and Possible Deterioration
Six trials evaluating short half-life PDE5 inhibitors (311 participants) found no significant HbA1c change, with a mean difference of +0.18% (95% CI −0.09 to 0.45).
In subgroup analyses of longer trials involving participants with type 2 diabetes and baseline HbA1c ≥6.5%, sildenafil and avanafil actually showed a statistically significant increase in HbA1c (+0.36%; p = 0.03).
This opposing trend accentuates the importance of sustained pharmacologic exposure: while tadalafil stabilizes glucose-related pathways around the clock, sildenafil’s brief activity window may be insufficient to sustain metabolic benefits.
Why Baseline HbA1c Matters More Than We Thought
One of the most illuminating insights from this study is the sensitivity of glycaemic reduction to baseline HbA1c levels. Across all drug classes—not just PDE5 inhibitors—there is a direct correlation between baseline glycaemia and observed HbA1c reduction. This relationship is clearly depicted in Figure 2e, showing that the higher the baseline HbA1c, the greater the potential drug effect.
Because many included trials recruited participants with relatively well-controlled diabetes (baseline ~7.7%), the observed effects likely underestimate the true potential of long half-life PDE5 inhibitors in populations with more severe hyperglycaemia.
This also explains the substantial heterogeneity seen in pooled results. When high-HbA1c outliers were removed in sensitivity analyses, heterogeneity dropped dramatically while effect sizes remained significant.
Secondary Metabolic Endpoints: Signals Worth Further Exploration
The meta-analysis evaluated several secondary metabolic parameters, including:
- HOMA-IR
- BMI
- fasting glucose
- postprandial glucose
- fasting insulin
- lipid profile components
None of these achieved statistical significance, with the exception of postprandial glucose in a single long half-life PDE5 inhibitor trial (Hegazy 2024).
However, every mean difference for long half-life drugs moved in a metabolically favorable direction, a pattern that warrants further investigation. In contrast, mean differences for short half-life agents appeared directionally random.
The limitation here is clear: the analyses were underpowered by small sample sizes and incomplete reporting. Future trials measuring insulin sensitivity, inflammatory markers, and endothelial function could greatly enrich our understanding.
Clinical Implications: Where Do PDE5 Inhibitors Fit in Diabetes Care?
This study offers several compelling insights that may influence clinical practice and future drug repurposing strategies.
1. A Case for Combination Therapy
Long half-life PDE5 inhibitors operate through a mechanism largely distinct from metformin, SGLT2 inhibitors, GLP-1 receptor agonists, or sulfonylureas. This non-redundant mechanism makes them attractive adjuncts in combination therapy—especially for patients with high baseline HbA1c who require multi-pathway intervention.
2. A Dual-Benefit Therapy for a Common Comorbidity
Approximately 66% of patients with type 2 diabetes experience erectile dysfunction, making PDE5 inhibitors already widely prescribed in this population.
If long half-life agents also improve glycaemic control, clinicians could address two conditions with one medication, enhancing adherence and reducing polypharmacy.
3. Optimizing Pharmacokinetics for Metabolic Outcomes
Given the large disparity between long and short half-life agents, the field would benefit from trials exploring:
- extended-release tadalafil formulations
- optimized dosing frequencies
- novel PDE5 inhibitors with prolonged half-lives
Improving exposure consistency may amplify metabolic benefits.
4. Cost and Accessibility Are Increasingly Favorable
With the expansion of low-cost generics and services such as CostPlus and GoodRx, affordability is no longer the barrier it once was.
This creates an opportunity to evaluate tadalafil as a practical adjunctive therapy in real-world diabetes management.
Research Gaps and Future Directions
Despite promising results, several limitations underscore the need for further study.
First, many included trials were too short (<8 weeks) to detect full HbA1c changes, potentially underestimating drug effects. Trials exceeding 3 months are necessary for reliable assessment.
Second, populations with poor glycaemic control are underrepresented. The striking reductions observed in high-HbA1c groups demand additional dedicated trials.
Third, secondary metabolic endpoints were inconsistently reported, limiting insights into mechanism.
Finally, the meta-analysis challenges the assumption—common in clinical trial design—that PDE5 inhibitor use is metabolically irrelevant. The authors found that 85% of potentially eligible trials failed to report HbA1c at all. This oversight could obscure important findings in other therapeutic areas.
Conclusion: Rethinking PDE5 Inhibitors Through a Metabolic Lens
This comprehensive meta-analysis sheds new light on the metabolic potential of PDE5 inhibitors, revealing a pharmacokinetic split between drugs capable of lowering HbA1c and those that do not. Long half-life PDE5 inhibitors such as tadalafil consistently reduce HbA1c by clinically relevant margins, aligning them with established oral antihyperglycaemic agents—particularly in patients with elevated baseline glycaemia.
Short half-life inhibitors do not confer these benefits and in some cases worsen glycaemic control, strongly suggesting that sustained exposure is essential for metabolic efficacy.
While more robust and longer-term clinical trials are needed, the evidence is already compelling enough to consider long half-life PDE5 inhibitors as candidates for combination therapy in type 2 diabetes—especially among patients with both hyperglycaemia and erectile dysfunction.
Perhaps most importantly, this study reminds us that sometimes, the pharmacological story is hiding in plain sight: not in the molecule itself, but in how long it stays in the body.
FAQ
1. Can tadalafil or other long half-life PDE5 inhibitors be used as standalone treatments for diabetes?
No. Although long half-life PDE5 inhibitors reduce HbA1c, the effect size is modest compared to first-line therapies. Their best role is likely adjunctive, supporting other medications through a distinct mechanism.
2. Why do short half-life PDE5 inhibitors show no HbA1c improvement?
Short half-life agents such as sildenafil provide only intermittent pathway stimulation. Their metabolic activity fades quickly—especially overnight—making them insufficient to produce sustained glycaemic effects measurable via HbA1c.
3. Should clinicians start measuring HbA1c in trials involving PDE5 inhibitors?
Absolutely. This meta-analysis shows that PDE5 inhibitors can influence glycaemic control, contradicting the long-held assumption that their metabolic effects are negligible. HbA1c should be routinely measured in relevant RCTs.