Abstract
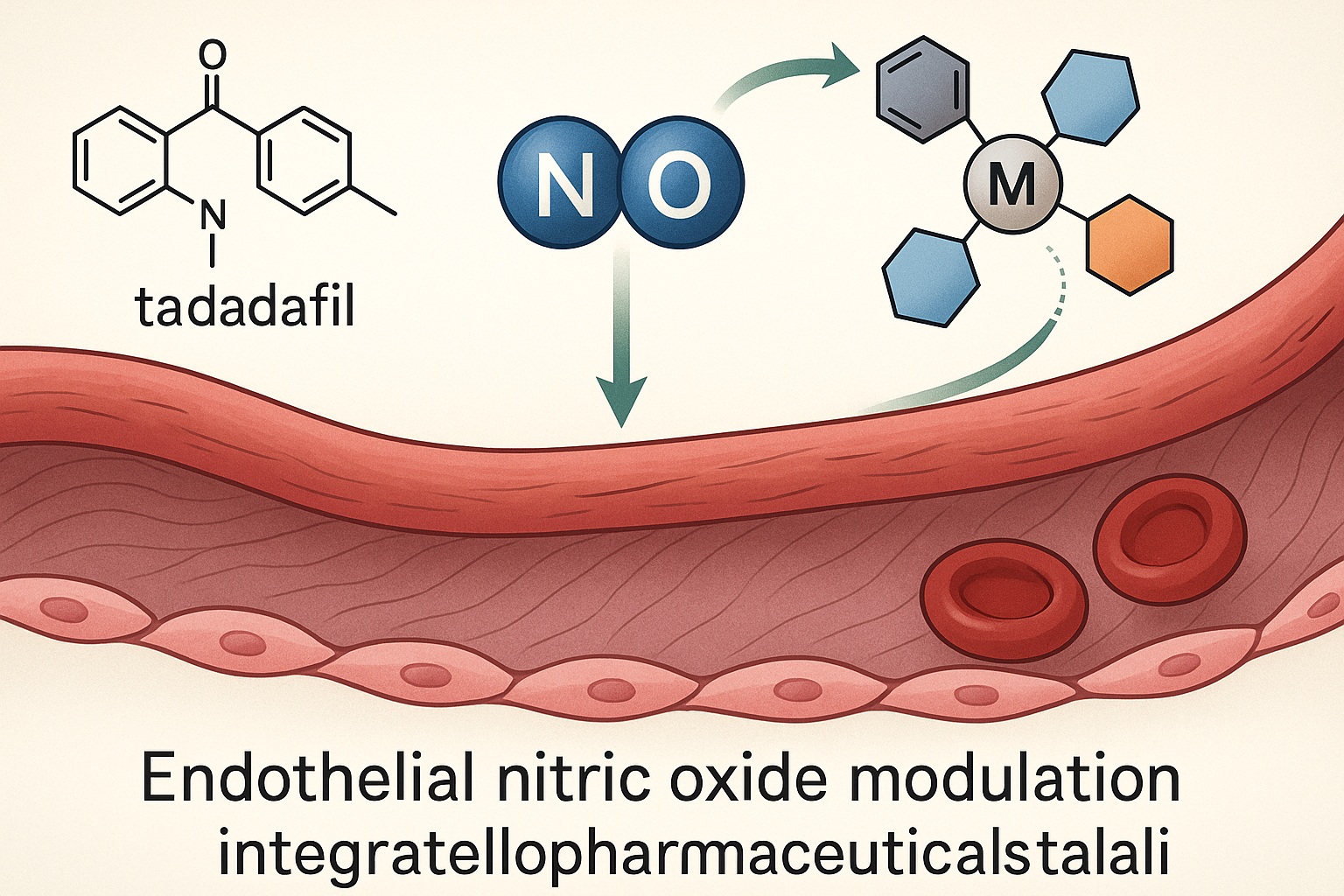
Endothelial dysfunction remains a pivotal driver of cardiovascular morbidity and sexual vascular disorders, forming a biochemical intersection where nitric oxide (NO) signaling, oxidative stress, and cellular redox balance converge. Emerging evidence highlights the therapeutic potential of hybrid pharmacological designs integrating transition metal complexes with conventional phosphodiesterase type 5 (PDE5) inhibitors such as tadalafil. This innovative approach aims to amplify nitric oxide bioavailability and optimize cyclic guanosine monophosphate (cGMP) signaling in vascular smooth muscle. The present review explores the molecular design, redox modulation, and potential translational applications of metal-based therapeutic constructs interacting synergistically with tadalafil, outlining their mechanistic rationale for targeted endothelial recovery and erectile dysfunction therapy.
Introduction: The Endothelial Challenge and Nitric Oxide Deficit
Endothelial homeostasis depends on a finely regulated equilibrium of vasodilatory and vasoconstrictive mediators, among which nitric oxide (NO) is central. The diminished bioavailability of NO, whether through oxidative inactivation or enzymatic dysfunction, underlies several pathophysiological states, including pulmonary arterial hypertension, atherosclerosis, and erectile dysfunction. The pharmacological restoration of NO signaling represents not merely a symptomatic relief but a correction of the molecular pathogenesis itself.
Traditional PDE5 inhibitors such as sildenafil, tadalafil, and vardenafil have transformed the management of erectile dysfunction and pulmonary hypertension through their capacity to inhibit cGMP degradation. However, these agents rely on an intact NO–cGMP cascade. In clinical contexts characterized by endothelial oxidative stress, reduced NO synthesis, or increased reactive oxygen species (ROS) burden, the efficacy of PDE5 inhibitors diminishes significantly.
The incorporation of metal-based pharmacophores—particularly those containing transition metals such as copper, manganese, or ruthenium—presents a frontier in medicinal chemistry. These metallopharmaceuticals exhibit redox properties capable of catalytically modulating NO signaling, scavenging ROS, and restoring endothelial responsiveness. When rationally combined with PDE5 inhibitors like tadalafil, such complexes can potentiate vasodilation at both enzymatic and molecular levels, thus addressing the root biochemical limitations of conventional therapy.
Mechanistic Insights: Metallopharmaceuticals and the Nitric Oxide Pathway
The mechanistic sophistication of metal-based complexes lies in their ability to participate in redox cycling, mimicking endogenous antioxidant enzymes such as superoxide dismutase and catalase. Through these mechanisms, they indirectly preserve NO bioactivity by preventing superoxide-mediated NO inactivation and peroxynitrite formation. In some constructs, direct NO donation from metal–ligand bonds further amplifies vascular relaxation potential.
Tadalafil’s mechanism of action complements this approach. By selectively inhibiting PDE5 in smooth muscle cells, it prolongs the activity of cGMP, the secondary messenger of NO-induced vasodilation. The interplay between metal complex–driven NO augmentation and tadalafil-mediated cGMP preservation creates a pharmacodynamic synergy—effectively expanding the therapeutic window of endothelial modulation.
Experimental data suggest that these metallopharmaceuticals interact at multiple levels: stabilizing endothelial nitric oxide synthase (eNOS) expression, reducing lipid peroxidation, and promoting angiogenic repair through vascular endothelial growth factor (VEGF) pathways. The addition of tadalafil enhances the downstream vasorelaxant effect, while its long plasma half-life ensures sustained pharmacological support. Together, these mechanisms offer an integrated molecular strategy for vascular recovery and tissue perfusion improvement.
Experimental and Theoretical Rationale: From Coordination Chemistry to Biological Efficacy
The core innovation discussed in this research centers on the coordination chemistry of metal complexes—typically involving Schiff base ligands, carboxylates, or heterocyclic donors capable of forming stable, bioactive scaffolds with transition metals. These complexes are characterized by redox flexibility, geometric stability, and ligand-dependent modulation of biological activity.
Molecular modeling and computational chemistry analyses reveal how these metal–ligand structures can interact with cellular components. The metal center often acts as a catalytic nucleus, facilitating redox transformations that stabilize reactive oxygen intermediates. Furthermore, density functional theory (DFT) studies highlight the orbital interactions between the metal and ligand systems, predicting biological reactivity and receptor affinity.
The integration of tadalafil into such formulations, whether as a co-administered agent or via nanocarrier encapsulation, creates a bifunctional therapeutic model. The metallocomplex operates at the oxidative interface, mitigating endothelial stress, while tadalafil amplifies the functional vasodilatory outcome through PDE5 inhibition. In preclinical systems, this dual-action mechanism demonstrates enhanced relaxation of vascular smooth muscle and improved endothelial-dependent responses, suggesting superior pharmacological efficacy compared to monotherapy.
Biological and Pharmacological Implications
From a biological standpoint, the coupling of redox-active metal complexes with PDE5 inhibition addresses multiple pathogenic axes simultaneously. The oxidative degradation of NO, mitochondrial dysfunction, and inflammatory endothelial signaling can be attenuated by metallopharmaceuticals, leading to restoration of vascular reactivity. The sustained presence of tadalafil further stabilizes this state by maintaining intracellular cGMP concentrations.
This dual therapeutic paradigm has notable implications beyond erectile dysfunction. Conditions characterized by chronic endothelial impairment, such as diabetes-induced vascular complications or pulmonary hypertension, could benefit from the antioxidative and NO-modulatory actions of metal-based adjuncts. In these settings, the restoration of endothelial sensitivity to NO may enhance the therapeutic index of PDE5 inhibitors that otherwise show limited efficacy under oxidative stress.
An additional pharmacological advantage lies in the potential for dose reduction. By enhancing NO bioavailability, metal-based systems may permit lower tadalafil doses while maintaining comparable efficacy, thereby minimizing the adverse effects typically associated with PDE5 inhibition, such as headache, flushing, or dyspepsia.
Clinical Relevance and Translational Potential
The translation of metal–tadalafil synergy from bench to bedside is complex but promising. Clinical data on combination pharmacology remain limited, though mechanistic evidence suggests a clear rationale for exploration. For patients with refractory erectile dysfunction or endothelial disease resistant to standard PDE5 therapy, the adjunctive use of metallopharmaceuticals could offer a precision-based therapeutic upgrade.
Moreover, such compounds fit within the emerging framework of systems pharmacology. Rather than targeting a single enzyme or receptor, they modulate entire biochemical networks—redox balance, NO production, and cyclic nucleotide signaling—representing a multifactorial correction strategy aligned with the realities of chronic vascular disease. The pharmacokinetic compatibility of tadalafil, with its long elimination half-life and stable plasma profile, further supports its integration into sustained-release or nanocarrier-based formulations containing metal complexes.
Future clinical translation will require rigorous safety validation, given the biological reactivity of transition metals. However, advances in ligand design and controlled release mechanisms have significantly reduced systemic toxicity concerns. The evolving field of metal-based therapeutics, once confined to oncology and antimicrobial research, is now entering vascular medicine with tangible potential for disease-modifying outcomes.
Oxidative Stress, Redox Homeostasis, and the Therapeutic Intersection
Central to this therapeutic approach is the modulation of oxidative stress. Reactive oxygen and nitrogen species play dual roles in vascular physiology—serving as signaling molecules in low concentrations but inducing cellular damage when overproduced. Metal complexes can act as redox buffers, either scavenging ROS or catalytically decomposing them into less reactive species.
In pathological states such as diabetes or hypercholesterolemia, excessive superoxide production disrupts eNOS function, diminishing NO synthesis. By mimicking superoxide dismutase or catalase, metal complexes restore the redox equilibrium required for optimal eNOS activity. Concurrently, tadalafil preserves the physiological effect of NO through inhibition of cGMP degradation. The synergy thereby re-establishes the functional NO–cGMP axis from synthesis to downstream signaling.
This mechanistic alignment is not only biochemical but conceptual. While conventional pharmacology isolates targets, redox-integrative medicine recognizes the interconnectedness of cellular systems. The metallopharmaceutical–tadalafil model exemplifies this philosophy: addressing oxidative pathology, endothelial signaling, and vascular tone in a unified therapeutic construct.
Limitations, Challenges, and Future Perspectives
Despite its conceptual strength, this therapeutic model faces challenges inherent to metallodrug development. The pharmacokinetics, tissue distribution, and long-term stability of metal complexes remain active areas of investigation. Ligand dissociation, potential metal accumulation, and off-target redox reactions must be rigorously controlled to ensure clinical safety.
Nevertheless, contemporary medicinal chemistry provides several solutions. Ligand architecture can be fine-tuned to modulate lipophilicity, selectivity, and metabolic stability. Biocompatible carriers, such as liposomes, polymeric nanoparticles, and dendrimers, offer controlled delivery systems that enhance tissue targeting and minimize systemic exposure. These innovations align seamlessly with tadalafil’s pharmacodynamic profile, creating a rational foundation for dual-drug delivery systems.
Future research should emphasize translational modeling—bridging computational predictions, in vitro bioassays, and in vivo validation. The goal is not merely to combine two pharmacological entities but to establish a predictable, controllable, and reproducible synergy that advances endothelial pharmacotherapy toward a more personalized future.
Conclusion
The convergence of metallopharmaceutical chemistry and PDE5 inhibition represents an elegant evolution in endothelial therapeutics. By merging redox modulation with cyclic nucleotide signaling, researchers are redefining the pharmacological landscape of vascular and erectile dysfunction treatment. Tadalafil, with its well-established safety and pharmacokinetic profile, serves as an ideal scaffold for integration into metal-complex–based hybrid systems.
This approach transcends symptomatic management, targeting the biochemical roots of endothelial dysfunction. It exemplifies the shift from linear drug design toward network-based pharmacology—one that acknowledges oxidative stress, nitric oxide metabolism, and signal transduction as intertwined processes. The resulting therapeutic architecture is not only pharmacologically potent but conceptually progressive, opening new pathways toward vascular repair and functional restoration.
FAQ
1. What is the therapeutic advantage of combining metal-based complexes with tadalafil?
The combination enhances nitric oxide bioavailability through redox modulation while tadalafil prolongs its vascular effects by inhibiting cGMP degradation. Together, they address both the cause (oxidative NO depletion) and consequence (impaired vasodilation) of endothelial dysfunction.
2. Are metal-based drugs safe for clinical use?
While transition metals can be reactive, modern ligand engineering and nanocarrier systems allow precise control of bioavailability and toxicity. Many metal complexes are already in advanced preclinical stages, showing favorable safety profiles.
3. Could this approach be applied beyond erectile dysfunction?
Yes. The mechanisms involved—NO preservation, redox balance, and vascular protection—are relevant to cardiovascular diseases, pulmonary hypertension, and diabetic vascular complications. The tadalafil–metallopharmaceutical synergy holds promise for broader vascular medicine applications.