Introduction: When a Tablet Looks Right but Behaves Wrong
Counterfeit medicines represent one of the most underestimated threats to modern healthcare. Unlike dramatic medical emergencies, their damage unfolds quietly: through treatment failure, unpredictable adverse effects, erosion of patient trust, and long-term public health consequences. Among the many drug classes affected, phosphodiesterase-5 (PDE-5) inhibitors occupy a peculiar and revealing position.
In industrialized countries, counterfeit medicines are rarely life-saving drugs. Instead, they target so-called “lifestyle” indications—erectile dysfunction being the most prominent. This apparent triviality is deceptive. PDE-5 inhibitors are potent vasoactive agents, often used by middle-aged and elderly men with cardiovascular comorbidities. In this context, poor pharmaceutical quality is not merely a technical defect; it is a clinical risk.
Most evaluations of counterfeit medicines stop at identifying the active ingredient and measuring its quantity. While these steps are necessary, they are not sufficient. A tablet may contain the correct molecule in the correct amount and still fail therapeutically. The missing link is pharmaceutical performance, and dissolution behavior is one of its most critical determinants.
Dissolution as a Gatekeeper of Bioavailability
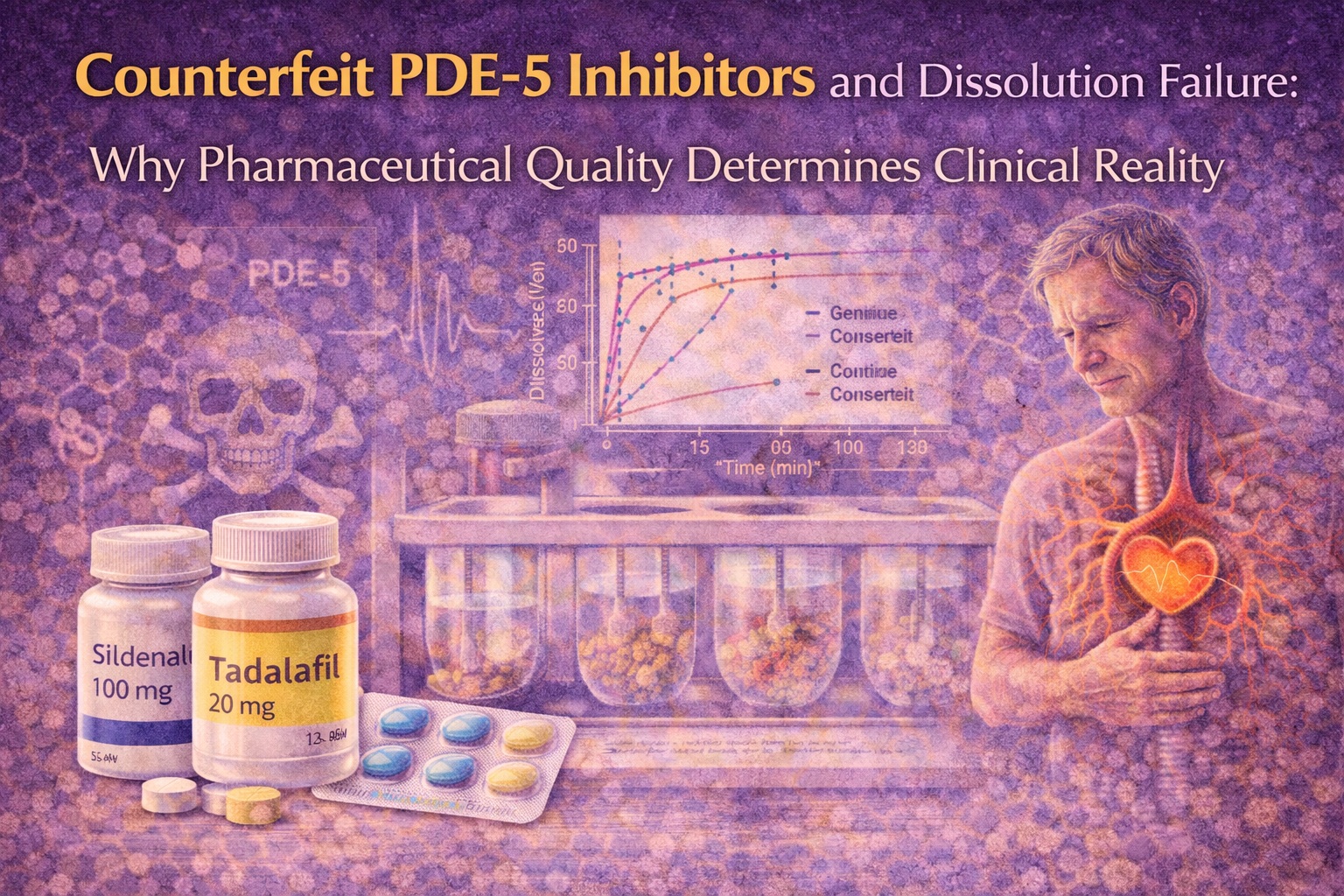
In vitro dissolution testing is not an academic exercise. It is a surrogate for in vivo drug release, absorption, and ultimately therapeutic effect. For immediate-release oral formulations, especially those containing poorly soluble compounds, dissolution is often the rate-limiting step for bioavailability.
Sildenafil and tadalafil exemplify this principle. Both molecules exhibit limited aqueous solubility and depend heavily on formulation design, excipient selection, and manufacturing parameters to ensure reliable release. Genuine products are engineered to meet strict dissolution criteria, ensuring predictable pharmacokinetics across patients.
Counterfeit medicines, by contrast, are produced without adherence to good manufacturing practices. Even when the correct active pharmaceutical ingredient is present, the absence of formulation science transforms the tablet into a pharmacological gamble. Dissolution testing exposes this gap with uncomfortable clarity.
The Global Context of Counterfeit PDE-5 Inhibitors
Counterfeit PDE-5 inhibitors dominate the illicit pharmaceutical market in high-income countries. Their appeal is obvious: high demand, discreet purchasing channels, and the perception of low medical risk. Online sales amplify this problem, bypassing regulatory oversight entirely.
From a public health perspective, the risk is often misunderstood. While erectile dysfunction itself is not life-threatening, PDE-5 inhibitors interact with cardiovascular physiology, nitrates, antihypertensives, and underlying vascular disease. Unpredictable drug release translates directly into unpredictable hemodynamic effects.
The study under analysis focuses on counterfeit products circulating in a regulated European market, underscoring an uncomfortable truth: pharmaceutical counterfeiting is not confined to weak regulatory environments. It exploits consumer behavior rather than regulatory failure.
Why Content Uniformity Alone Is Not Enough
A striking finding in counterfeit medicine analysis is that many samples contain approximately the declared dose of the active ingredient. Superficially, this suggests acceptable quality. Clinically, it is misleading.
Content uniformity assesses quantity, not performance. It does not capture how quickly or completely the drug becomes available for absorption. Two tablets with identical drug content can produce radically different plasma concentration–time profiles if their dissolution characteristics diverge.
In the context of PDE-5 inhibitors, this distinction is critical. Rapid dissolution may lead to unexpectedly high peak concentrations, increasing the risk of adverse effects. Slow or incomplete dissolution may result in treatment failure, prompting patients to increase dosage or combine products, compounding risk.
Dissolution Variability as a Marker of Manufacturing Chaos
One of the most revealing aspects of dissolution testing in counterfeit PDE-5 inhibitors is variability—both between different products and between units of the same product. High variability at early time points reflects inconsistent disintegration and poor control of tablet properties.
This variability is not random. It is the predictable consequence of uncontrolled particle size, non-standard excipients, inappropriate compression forces, and lack of formulation optimization. In genuine products, early dissolution behavior is tightly regulated because it shapes the entire release profile.
In counterfeit products, early dissolution often resembles a coin toss. Some units release the drug rapidly, others barely at all. For the patient, this translates into erratic clinical response that undermines both efficacy and safety.
Comparing Counterfeit and Genuine Products: A Statistical Reality Check
Objective comparison of dissolution profiles requires more than visual inspection. Statistical tools such as similarity factors and time-point-by-time-point comparisons provide quantitative insight into equivalence—or the lack thereof.
When counterfeit PDE-5 inhibitors are compared with genuine reference products, true equivalence is rare. In most cases, dissolution profiles deviate substantially, particularly during the early phases of release. Even when later time points approach similar cumulative dissolution, early divergence remains clinically relevant.
This finding has practical implications. Early drug release determines onset of action and peak exposure, both of which are critical for PDE-5 inhibitors. Late convergence does not compensate for early failure.
Formulation Matters More Than the Molecule
The assumption that “sildenafil is sildenafil” collapses under dissolution testing. Pharmaceutical formulation transforms a chemical entity into a medicine. Without proper formulation, the molecule loses its therapeutic reliability.
Counterfeit products often employ inappropriate excipients or incorrect proportions of otherwise acceptable ones. Lubricants, binders, disintegrants, and surfactants are not interchangeable components; they are integral to performance. Small deviations can produce large effects.
Compression force is another underestimated factor. Excessive compression may produce tablets that resist disintegration, while insufficient compression leads to friable units with erratic dissolution. Genuine manufacturers optimize these parameters through extensive development. Counterfeit producers do not.
Clinical Consequences: From Inefficacy to Toxicity
The clinical implications of poor dissolution extend beyond inconvenience. Incomplete or delayed dissolution can lead to therapeutic failure, prompting patients to self-adjust dosing or seek alternative illicit products. This behavior increases exposure to unknown risks.
Conversely, rapid and uncontrolled dissolution may result in supratherapeutic peaks, increasing the likelihood of adverse effects such as hypotension, headache, visual disturbances, or dangerous interactions with nitrates. In vulnerable populations, these effects are not benign.
Importantly, patients are unlikely to attribute these outcomes to pharmaceutical quality. Instead, they may blame the drug class, their physician, or themselves—further eroding trust in legitimate healthcare.
Dissolution as a Public Health Indicator
In vitro dissolution testing serves as a proxy for bioequivalence, and by extension, for therapeutic reliability. When counterfeit products consistently fail dissolution equivalence, they signal a broader public health threat.
If similar manufacturing practices are applied to life-saving medicines—antibiotics, antivirals, cardiovascular drugs—the consequences escalate dramatically. Treatment failure, resistance development, and avoidable mortality become real possibilities.
PDE-5 inhibitors thus function as a canary in the pharmaceutical coal mine. Their counterfeits reveal what happens when drug quality is treated as optional rather than essential.
Regulatory and Enforcement Implications
The findings of dissolution studies challenge enforcement strategies that focus solely on chemical identification. A counterfeit tablet containing the correct molecule can still be a dangerous product.
Regulatory authorities must therefore integrate pharmaceutical performance testing into counterfeit surveillance. Dissolution testing is relatively accessible, reproducible, and informative. Its broader use could refine risk assessment and enforcement prioritization.
Public communication also matters. Educating consumers about the risks of counterfeit medicines should move beyond scare tactics and focus on concrete, understandable concepts—such as unpredictable drug release and inconsistent effects.
Why Patients Still Buy Counterfeit PDE-5 Inhibitors
Despite known risks, demand for counterfeit PDE-5 inhibitors persists. Cost, convenience, anonymity, and stigma all play a role. Many patients perceive erectile dysfunction treatment as optional or cosmetic, reducing perceived risk.
Ironically, this perception increases danger. Patients self-medicate without medical supervision, often combining counterfeit drugs with prescribed cardiovascular medications. Dissolution variability amplifies these risks unpredictably.
Addressing counterfeit medicines therefore requires not only regulation but also cultural change—normalizing discussion of sexual health within legitimate healthcare systems.
Lessons for Pharmaceutical Science and Clinical Practice
The comparative dissolution behavior of counterfeit PDE-5 inhibitors reinforces a fundamental lesson: drug quality is inseparable from clinical outcome. Active ingredient identity is only the starting point.
For clinicians, this underscores the importance of asking patients about non-prescribed medication use, particularly for erectile dysfunction. For pharmacists, it highlights the role of patient education. For regulators, it strengthens the case for performance-based surveillance.
For pharmaceutical science, it is a reminder that formulation is not an afterthought—it is the medicine.
Conclusion: Dissolution Profiles Do Not Lie
Counterfeit PDE-5 inhibitors expose the fragile illusion that a tablet’s appearance or label guarantees its behavior. Dissolution testing strips away that illusion, revealing profound and clinically relevant differences between genuine and counterfeit products.
The evidence is clear: most counterfeit PDE-5 inhibitors do not behave like their legitimate counterparts, even when they contain the correct active ingredient. Their unpredictable dissolution profiles translate into unpredictable clinical effects, posing real risks to patients.
In an era of expanding online pharmaceutical markets, dissolution testing stands as a powerful reminder that quality is measurable, and that ignoring it carries consequences. A tablet that dissolves incorrectly is not merely substandard—it is unfit to be called a medicine.
FAQ
Why is dissolution testing important for detecting counterfeit medicines?
Because it assesses how a drug is released, not just what it contains. Poor dissolution can cause treatment failure or toxicity even when the active ingredient is present.
Can a counterfeit drug with the correct dose still be dangerous?
Yes. Incorrect dissolution leads to unpredictable absorption and clinical effects, especially for vasoactive drugs like PDE-5 inhibitors.
Are counterfeit PDE-5 inhibitors only a problem in developing countries?
No. They are particularly prevalent in industrialized countries through online and informal markets, targeting lifestyle indications.