Introduction: A Paradigm Shift in Vascular Medicine
Pulmonary arterial hypertension (PAH) represents one of the most formidable challenges in modern cardiovascular medicine. Once deemed a rapidly fatal disease, PAH is now increasingly viewed as a chronic but manageable condition, owing to advances in pharmacotherapy that target the molecular underpinnings of pulmonary vascular remodeling and vasoconstriction. Central to this therapeutic revolution is tadalafil, a long-acting phosphodiesterase type 5 (PDE5) inhibitor that exploits the nitric oxide–cyclic guanosine monophosphate (NO–cGMP) signaling pathway to restore pulmonary vascular tone and function.
The evolution of PAH management has been marked by the transition from nonspecific vasodilators to drugs with defined molecular targets. Where once therapies focused purely on symptom alleviation, the new era emphasizes pathway-specific intervention—treatments designed not just to relax pulmonary vessels temporarily but to remodel and sustain vascular health. Among the emerging pharmacological agents, tadalafil has distinguished itself through its efficacy, safety, and impact on quality of life. Its pharmacokinetic profile, coupled with sustained bioactivity, allows for once-daily dosing—an advantage of both clinical convenience and patient adherence.
In an expanding therapeutic landscape that includes prostacyclin analogs, endothelin receptor antagonists, and soluble guanylate cyclase stimulators, tadalafil occupies a distinct niche: a balance of potency, tolerability, and real-world practicality. Understanding its mechanism and clinical implications demands more than a pharmacologic summary—it requires an appreciation of how restoring endothelial communication translates into tangible improvements in exercise capacity, cardiopulmonary dynamics, and ultimately, patient survival.
The Pathophysiology of Pulmonary Arterial Hypertension
PAH is characterized by progressive narrowing of the small pulmonary arteries, resulting from endothelial dysfunction, smooth muscle proliferation, and extracellular matrix remodeling. The culmination of these processes is elevated pulmonary vascular resistance (PVR) and increased mean pulmonary arterial pressure (mPAP), which together burden the right ventricle and lead to heart failure.
At the molecular level, three principal signaling abnormalities drive this pathogenesis:
- Reduced nitric oxide (NO) bioavailability, leading to diminished cGMP-mediated vasodilation.
- Overexpression of endothelin-1, a potent vasoconstrictor and mitogen.
- Deficient prostacyclin signaling, resulting in loss of antiproliferative control.
These dysregulations reinforce each other, creating a self-perpetuating loop of vasoconstriction and vascular remodeling. The right ventricle, initially adaptive through hypertrophy, eventually decompensates under chronic pressure overload.
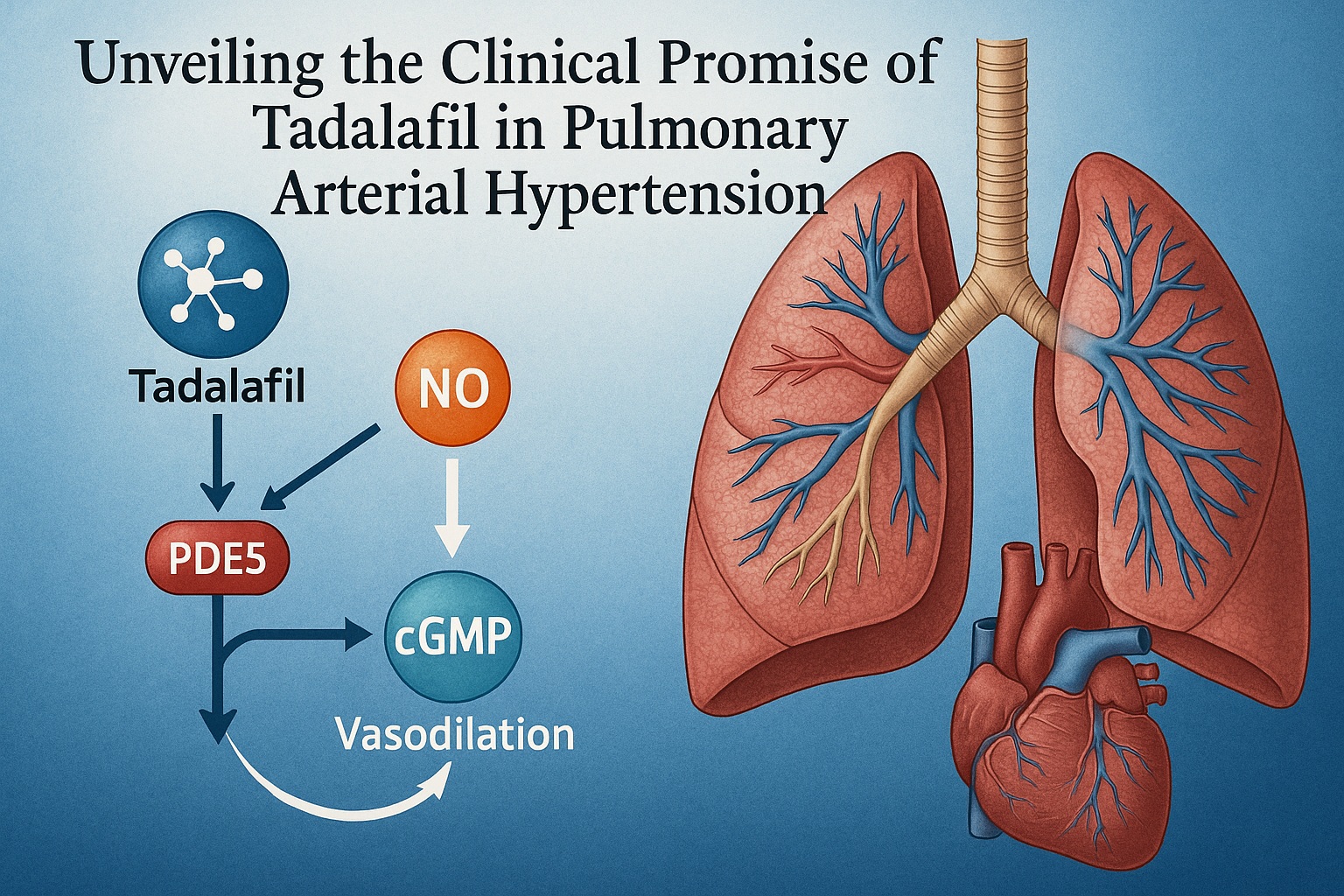
By targeting PDE5—the enzyme responsible for degrading cGMP—tadalafil effectively interrupts this pathological cycle, restoring the balance between vasodilatory and vasoconstrictive forces. This mechanistic specificity positions tadalafil as both a hemodynamic modulator and a structural stabilizer in the pulmonary vasculature.
Pharmacodynamics of Tadalafil: Precision at the Molecular Level
Tadalafil selectively inhibits phosphodiesterase type 5 (PDE5), the predominant PDE isoenzyme in pulmonary arterial smooth muscle cells. Under normal physiology, PDE5 limits cGMP accumulation, thereby constraining nitric oxide–mediated relaxation. Inhibition of PDE5 prolongs the bioavailability of cGMP, allowing enhanced smooth muscle relaxation, vasodilation, and antiproliferative effects.
Unlike other PDE5 inhibitors such as sildenafil, tadalafil possesses a longer half-life—approximately 17.5 hours—facilitating once-daily dosing with minimal fluctuation in plasma concentration. This pharmacological stability ensures continuous therapeutic activity, a key factor in maintaining consistent pulmonary hemodynamics and improving exercise tolerance.
Furthermore, tadalafil’s high selectivity for PDE5 over PDE6 (retinal enzyme) minimizes visual disturbances—a common side effect seen with other agents in the class. Its limited interaction with cytochrome P450 enzymes also contributes to a favorable safety and drug-interaction profile.
Mechanistically, the therapeutic benefits of tadalafil extend beyond vasodilation. Experimental evidence demonstrates inhibition of smooth muscle proliferation, reduction of oxidative stress, and attenuation of inflammatory cytokine activity. Collectively, these effects transform tadalafil from a mere symptomatic therapy into a disease-modifying agent within the continuum of PAH care.
Clinical Evaluation: Methodology and Patient Population
The referenced study, a double-blind, randomized, placebo-controlled clinical trial, investigated the efficacy and safety of tadalafil in patients with pulmonary arterial hypertension. Participants included both idiopathic and connective tissue disease–associated PAH cases, predominantly classified as World Health Organization (WHO) functional class II–III—indicative of moderate-to-severe disease with significant limitation of physical activity.
Patients were randomized to receive placebo or tadalafil (2.5 mg to 40 mg daily) for 16 weeks. The primary efficacy endpoint was change in 6-minute walk distance (6MWD), a validated measure of exercise capacity and functional improvement. Secondary endpoints included quality of life assessments (SF-36 and EQ-5D), hemodynamic parameters (mPAP, cardiac index, PVR), and time to clinical worsening.
Importantly, the trial design incorporated a dose-ranging structure to establish the optimal therapeutic threshold while monitoring for tolerability. This approach not only ensured clinical robustness but also allowed for precise mapping of dose-response relationships—a critical consideration in chronic pharmacotherapy.
Results: From Pulmonary Pressure to Patient Performance
At the end of 16 weeks, the findings were unequivocal: tadalafil significantly improved exercise capacity, with the 40 mg daily dose producing the most pronounced gains. Patients in this group exhibited a mean increase of approximately 33 meters in 6MWD compared with placebo—a clinically meaningful improvement corroborated by enhanced WHO functional class in a substantial proportion of participants.
Beyond exercise capacity, hemodynamic markers demonstrated corresponding benefits. Mean pulmonary arterial pressure decreased, while cardiac index and stroke volume improved, reflecting both reduced right ventricular afterload and enhanced cardiac performance. Importantly, these changes occurred without compromising systemic blood pressure, highlighting tadalafil’s vascular selectivity.
Quality of life indices further reinforced the physiological outcomes. Across multiple domains—physical functioning, vitality, and social engagement—tadalafil-treated patients reported notable improvements in HRQoL scores. This dual enhancement of function and perception underscores the holistic therapeutic effect of PDE5 inhibition: restoring not just circulation, but also confidence and participation in daily life.
Adverse events were generally mild and transient, consisting mainly of headache, myalgia, and facial flushing. These side effects seldom led to treatment discontinuation, affirming tadalafil’s excellent tolerability in long-term use.
The 40 mg Revelation: Defining the Optimal Dose
The trial’s dose-ranging design revealed a clear dose-response relationship—a hallmark of true pharmacologic efficacy. While lower doses (10–20 mg) produced moderate benefits, only the 40 mg daily regimen consistently achieved statistical significance across all major endpoints.
This finding has clinical relevance beyond statistical modeling. The sustained plasma levels achieved with 40 mg tadalafil not only optimize pulmonary vasodilation but also provide steady cGMP modulation throughout the 24-hour dosing cycle. By minimizing fluctuations in vascular tone, this regimen ensures continuous right ventricular unloading, which is critical for disease stabilization.
From a practical standpoint, once-daily 40 mg dosing enhances patient adherence, a factor that often dictates long-term success in chronic PAH management. This convenience, paired with durable efficacy, situates tadalafil as an ideal first-line oral therapy—especially in patients unable or unwilling to tolerate parenteral prostacyclin formulations.
Translating Molecular Modulation into Clinical Reality
Tadalafil’s clinical success represents the culmination of molecular insight translated into patient benefit. The drug’s ability to restore endothelial communication through the NO–cGMP axis forms the mechanistic bridge between microscopic and macroscopic healing. The downstream consequences—improved perfusion, oxygenation, and cardiac performance—are not merely theoretical; they manifest in tangible human outcomes such as increased walking distance, improved mood, and reduced fatigue.
The implications extend to long-term disease trajectory. By alleviating right ventricular strain and preventing further remodeling, tadalafil may delay or even prevent the transition to decompensated right heart failure—a turning point often associated with irreversible decline. Thus, beyond immediate functional gains, tadalafil embodies the principle of preemptive repair: targeting molecular dysfunction before it cascades into organ failure.
Quality of Life: The Human Dimension of Hemodynamic Success
In chronic diseases like PAH, the true measure of success lies not solely in numerical improvements but in qualitative transformation. Patients enrolled in tadalafil trials consistently described enhanced physical endurance, improved mood, and renewed social interaction. These seemingly subjective outcomes, captured through SF-36 and EQ-5D instruments, complement the objective data, illustrating the therapy’s full spectrum of impact.
Interestingly, domains such as social functioning and emotional well-being showed the largest proportional gains—a reminder that the relief of physical symptoms reverberates through psychological and relational dimensions. The integration of HRQoL metrics into clinical endpoints thus marks an important paradigm shift: from treating the disease to healing the patient.
Safety and Tolerability: Managing the Long Game
Long-term safety remains paramount in chronic PAH management, and tadalafil has consistently demonstrated an excellent tolerability profile. The most common adverse events—headache, flushing, nasal congestion, and dyspepsia—were mild and self-limiting. Serious adverse events related to systemic hypotension or visual disturbances were rare.
From a pharmacovigilance perspective, tadalafil’s lack of significant interactions with nitrates and alpha-blockers in this population broadens its clinical applicability. Furthermore, hepatic and renal function remained stable across treatment groups, underscoring its suitability for sustained administration.
Integrating Tadalafil into Contemporary PAH Therapy
In the modern therapeutic algorithm for PAH, PDE5 inhibitors like tadalafil occupy a foundational role, particularly for WHO functional class II–III patients. Their oral administration and favorable safety make them first-line options in monotherapy or combination regimens.
In advanced disease or in cases of suboptimal response, tadalafil may be co-administered with endothelin receptor antagonists (e.g., ambrisentan, bosentan) or prostacyclin analogs to achieve additive effects through complementary mechanisms. Such combination therapy, when guided by individualized hemodynamic monitoring, offers a tailored approach to disease control.
The strategic use of tadalafil not only delays the need for invasive therapies but also contributes to cost-effective long-term disease management, making it an appealing option in both high-resource and resource-limited settings.
The Broader Context: Beyond Pulmonary Vasculature
Emerging evidence suggests that tadalafil’s benefits extend beyond pulmonary circulation. Studies have demonstrated improved endothelial function in systemic vessels, anti-inflammatory modulation, and even beneficial effects on right ventricular remodeling independent of afterload reduction. This systemic pleiotropy hints at tadalafil’s potential utility in related conditions, including chronic thromboembolic pulmonary hypertension and right heart dysfunction secondary to left-sided heart disease.
Moreover, ongoing investigations are exploring combination strategies that pair tadalafil with soluble guanylate cyclase stimulators or novel anti-inflammatory agents, aiming to potentiate NO–cGMP signaling from multiple biochemical angles.
Conclusion: Restoring Breath and Balance
Tadalafil’s journey from a molecular concept to a clinical cornerstone encapsulates the progress of cardiovascular pharmacology in the 21st century. By targeting the PDE5–cGMP pathway with precision, it has redefined the management of pulmonary arterial hypertension, offering not only survival but a renewed quality of life. Its combination of efficacy, convenience, and safety makes it a model of modern therapeutic design—a pharmacologic intervention that is as humane as it is effective.
In the evolving narrative of PAH, tadalafil stands as both a scientific achievement and a symbol of medicine’s deeper mission: to translate biochemical insight into restored human vitality. For patients who once viewed breathlessness as their horizon, it represents, quite literally, a breath of new life.
FAQ
1. What is tadalafil’s main role in pulmonary arterial hypertension?
Tadalafil selectively inhibits phosphodiesterase type 5, leading to increased cGMP levels, vasodilation, and improved pulmonary hemodynamics. This results in reduced pulmonary arterial pressure, improved exercise capacity, and enhanced quality of life.
2. How is tadalafil administered for PAH?
The optimal dose established by clinical evidence is 40 mg once daily, which provides sustained therapeutic plasma levels and maximal benefit in exercise tolerance and quality-of-life outcomes.
3. Can tadalafil be safely used long-term?
Yes. Clinical trials and post-marketing data confirm its excellent tolerability for chronic administration, with minimal adverse effects and no major safety concerns when used according to clinical guidelines.