Introduction: When Metabolism Becomes an Immune Weapon
Cancer is often described as a genetic disease, but its most effective weapons are metabolic. Among them, lactic acid has long been viewed as a metabolic byproduct—an inconvenient consequence of aerobic glycolysis. That interpretation is no longer sufficient. Lactic acid is not merely waste; it is an active immunosuppressive signal embedded in the tumor microenvironment.
Invariant natural killer T (iNKT) cells sit at the crossroads of innate and adaptive immunity. They respond rapidly, produce large amounts of cytokines, and orchestrate downstream immune responses with remarkable efficiency. Yet in many solid tumors, these cells are functionally paralyzed. The question is no longer whether metabolism suppresses immunity, but how this suppression is executed at the molecular level.
Recent work reveals an unexpected player in this process: phosphodiesterase-5 (PDE5). Traditionally associated with smooth muscle tone and vascular biology, PDE5 now emerges as a metabolic switch capable of dampening immune function. The interaction between lactic acid, PDE5 expression, and cyclic GMP signaling provides a mechanistic explanation for iNKT cell dysfunction in acidic environments—and opens new therapeutic doors.
iNKT Cells: Rapid Responders with High Metabolic Sensitivity
iNKT cells are unconventional T lymphocytes that recognize lipid antigens presented by CD1d molecules. Upon activation, they release both Th1 and Th2 cytokines within hours, including interferon-γ (IFN-γ) and interleukin-4 (IL-4). This duality allows them to shape immune responses with speed and precision.
Unlike conventional T cells, iNKT cells rely heavily on rapid intracellular signaling cascades rather than prolonged transcriptional programs. This makes them exceptionally sensitive to changes in intracellular second messengers such as cyclic GMP (cGMP). Small perturbations in these pathways can produce disproportionately large functional effects.
In physiological conditions, iNKT cells downregulate inhibitory molecules after activation, allowing maximal cytokine output. However, the tumor microenvironment is anything but physiological. Hypoxia, nutrient depletion, and acidosis converge to reshape immune signaling at every level.
Lactic Acid: From Metabolic Footnote to Immune Suppressor
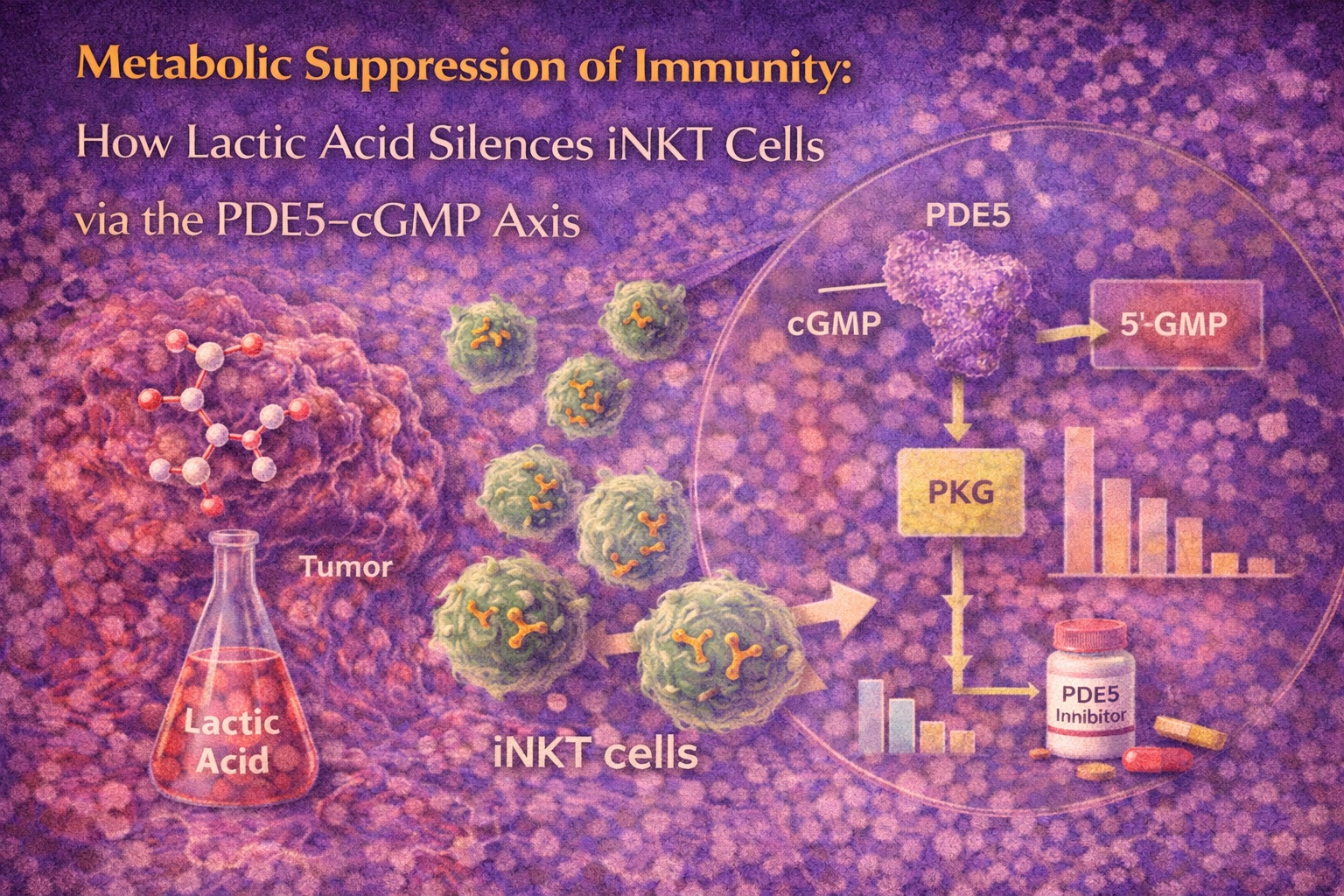
Tumor cells preferentially generate energy through aerobic glycolysis, producing large quantities of lactic acid even in the presence of oxygen. This leads to local acidification, with extracellular pH values often dropping below 6.8.
Acidic environments impair immune cells through multiple mechanisms: reduced receptor signaling, altered ion flux, and suppression of transcriptional activity. For iNKT cells, lactic acid exposure results in a marked reduction in IFN-γ and IL-4 production—two cytokines essential for anti-tumor immunity.
What makes lactic acid particularly insidious is its selectivity. It does not simply kill immune cells or prevent their activation. Instead, it subtly reprograms intracellular signaling pathways, rendering activated cells functionally inert while leaving them phenotypically intact.
PDE5: An Unexpected Immune Checkpoint
Phosphodiesterase-5 is best known for its role in hydrolyzing cGMP in vascular smooth muscle cells. Inhibition of PDE5 increases cGMP levels, leading to vasodilation—a principle exploited in the treatment of erectile dysfunction and pulmonary hypertension.
The presence of PDE5 in immune cells was initially considered incidental. However, accumulating evidence suggests that PDE5 plays an active regulatory role in immune signaling. In iNKT cells, PDE5 acts as a brake on cytokine production by limiting cGMP availability.
Under normal activation conditions, iNKT cells reduce PDE5 expression, effectively lifting this brake. This allows cGMP to accumulate, activate protein kinase G (PKG), and facilitate robust cytokine release. Lactic acid disrupts this balance by reversing PDE5 downregulation.
The cGMP–PKG Pathway: A Silent Amplifier of Immune Function
cGMP is a second messenger with diverse cellular effects. In immune cells, it acts primarily through PKG, a serine/threonine kinase that modulates transcription factors, cytoskeletal dynamics, and calcium signaling.
Activation of the cGMP–PKG axis enhances iNKT cell cytokine production without directly altering antigen receptor signaling. This distinction is crucial. It means that metabolic signals can modulate immune output independently of antigen recognition.
By increasing PDE5 expression, lactic acid effectively drains the cGMP pool. The result is PKG inactivity, blunted transcriptional responses, and reduced cytokine secretion—despite otherwise intact activation machinery.
Experimental Evidence: Connecting Metabolism to Signaling
Experimental data demonstrate that both sildenafil and tadalafil—classic PDE5 inhibitors—significantly enhance IFN-γ and IL-4 production in iNKT cells, both in vitro and in vivo . This effect is reproduced by cGMP analogues and abolished by PKG inhibition, confirming pathway specificity.
Crucially, lactic acid exposure increases Pde5a mRNA levels in activated iNKT cells. This effect is mirrored by simple extracellular acidification, indicating that pH—not lactate itself—is the dominant signal.
When PDE5 is pharmacologically inhibited in lactic acid-treated cells, cytokine production is partially restored. This rescue is incomplete but biologically meaningful, suggesting that PDE5 is a central—though not exclusive—mediator of metabolic immunosuppression.
Acidosis as a Regulatory Signal, Not a Toxic Insult
One of the most important conceptual shifts arising from this work is the reframing of acidosis. Acidic pH does not merely damage immune cells; it actively instructs them to reduce effector function.
By inducing PDE5 expression, acidosis introduces a reversible, signal-dependent checkpoint into immune responses. This allows tumors to suppress immunity without provoking inflammation or cell death—a strategy that is both elegant and effective.
From an evolutionary perspective, this makes sense. Metabolic cues have long been used to regulate immune activity in nutrient-limited environments. Cancer simply exploits this ancient regulatory system.
Clinical Implications: Repurposing PDE5 Inhibitors
PDE5 inhibitors are among the most widely prescribed drugs worldwide. Their safety profile is well characterized, their pharmacokinetics are predictable, and their cost is relatively low. This makes them attractive candidates for immunomodulatory repurposing.
The data suggest that PDE5 inhibition could enhance iNKT cell-mediated anti-tumor immunity, particularly in metabolically hostile environments. This does not imply that PDE5 inhibitors are standalone immunotherapies. Rather, they may function as metabolic adjuvants, restoring immune competence where it has been silenced.
Importantly, the effect is partial. This is not a flaw but a reflection of biological complexity. Tumor-induced immunosuppression is multifactorial, and no single intervention is likely to reverse it completely.
Combination Strategies: Where the Real Potential Lies
The most promising applications of PDE5 inhibition likely involve combination therapies. iNKT cell-based immunotherapies, adoptive cell transfer, and cancer vaccines all depend on robust cytokine production.
By relieving metabolic suppression, PDE5 inhibitors could amplify the effectiveness of these approaches without directly activating immune cells. This reduces the risk of systemic immune toxicity while enhancing local anti-tumor responses.
There is also potential synergy with checkpoint inhibitors. While PD-1 and CTLA-4 blockade release inhibitory receptor signaling, PDE5 inhibition restores intracellular signal amplification. Together, they may address complementary layers of immune suppression.
Limitations and Open Questions
Despite its elegance, the PDE5–cGMP model does not explain all aspects of lactic acid-induced immunosuppression. Other pathways, including mTOR inhibition and altered calcium signaling, also contribute.
Furthermore, the degree to which these findings translate to human tumors remains to be fully established. iNKT cell frequency and function vary widely between individuals, and tumor microenvironments are highly heterogeneous.
Finally, systemic PDE5 inhibition may have off-target effects on other immune subsets. While some of these effects may be beneficial, careful evaluation is essential before clinical translation.
Conclusion
Lactic acid is no longer just a metabolic byproduct—it is an active immunoregulatory signal that suppresses iNKT cell function by inducing PDE5 expression and collapsing cGMP-dependent signaling.
By identifying PDE5 as a metabolic immune checkpoint, this work bridges cancer metabolism and immunology with remarkable clarity. It also offers a practical therapeutic insight: metabolic suppression of immunity is reversible, at least in part, using well-established pharmacological tools.
In the evolving landscape of cancer immunotherapy, understanding—and manipulating—the metabolic language of immune cells may prove just as important as targeting their receptors.
FAQ
Why are iNKT cells particularly sensitive to metabolic changes?
Because their rapid cytokine release depends heavily on second-messenger signaling rather than prolonged transcriptional activation.
Does lactic acid suppress iNKT cells by killing them?
No. It primarily reduces their functional output by altering intracellular signaling pathways.
Can PDE5 inhibitors fully restore immune function in tumors?
No, but they can partially reverse metabolic suppression and may significantly enhance combination immunotherapies.