Introduction: A Familiar Drug in an Unexpected Role
Chronic kidney disease (CKD) remains one of the most pressing challenges in global health. Its progression is often silent, relentless, and tightly interwoven with other chronic conditions such as hypertension, diabetes, and atherosclerosis. Despite advances in pharmacotherapy, slowing the transition from early renal impairment to end-stage kidney disease (ESKD) continues to frustrate clinicians and researchers alike.
Traditionally, therapeutic strategies have focused on blood pressure control and modulation of the renin–angiotensin–aldosterone system. While effective to a degree, these approaches often fail to halt disease progression completely. The search for adjunctive or alternative therapies has therefore intensified.
Enter tadalafil—a phosphodiesterase type 5 (PDE5) inhibitor more commonly associated with erectile dysfunction. Recent experimental evidence suggests that this drug may possess renoprotective properties, extending its clinical relevance far beyond urology. The study analyzed here demonstrates that tadalafil can mitigate kidney injury in a salt-sensitive model of CKD, raising important questions about its broader therapeutic potential .
Pathophysiological Context: CKD as a Hemodynamic and Fibrotic Disease
CKD is not a single disease but a syndrome characterized by progressive loss of renal function. Its pathogenesis involves a complex interplay of hemodynamic stress, inflammation, and fibrosis. Among these, hypertension—particularly salt-sensitive hypertension—plays a central role.
In salt-sensitive individuals, excessive sodium intake leads to disproportionate increases in blood pressure, accelerating glomerular injury. Over time, this results in structural changes such as glomerulosclerosis, podocyte damage, and interstitial fibrosis. These alterations are not merely consequences of disease—they actively drive its progression.
The experimental model used in the study, Dahl salt-sensitive rats, mirrors this human condition with remarkable fidelity. As shown in Table 3 on page 4, animals exposed to a high-salt diet developed severe hypertension, with systolic blood pressure exceeding 200 mmHg . This hemodynamic burden sets the stage for renal injury.
However, CKD is not solely a hemodynamic disorder. Fibrotic remodeling, mediated by myofibroblast activation and extracellular matrix deposition, represents a critical and often irreversible component of disease progression. Understanding and targeting these processes is essential for effective intervention.
Tadalafil and the NO–cGMP Axis: Mechanistic Foundations
Tadalafil exerts its pharmacological effects through inhibition of PDE5, an enzyme responsible for the degradation of cyclic guanosine monophosphate (cGMP). By preventing cGMP breakdown, tadalafil enhances nitric oxide (NO) signaling, leading to smooth muscle relaxation and vasodilation.
In the renal context, this pathway has broader implications. cGMP is involved in regulating glomerular hemodynamics, endothelial function, and cellular signaling within the kidney. Disruption of the NO–cGMP axis has been linked to renal dysfunction, making it an attractive therapeutic target .
Importantly, PDE5 expression has been identified in key renal structures, including podocytes. These specialized cells form part of the glomerular filtration barrier and are essential for maintaining selective permeability. Their dysfunction is a hallmark of CKD.
By enhancing cGMP signaling, tadalafil may stabilize these structures, preserving filtration capacity and reducing protein leakage. This mechanism extends beyond simple vasodilation, suggesting a direct effect on renal cellular integrity.
Experimental Evidence: Functional Improvements in Kidney Health
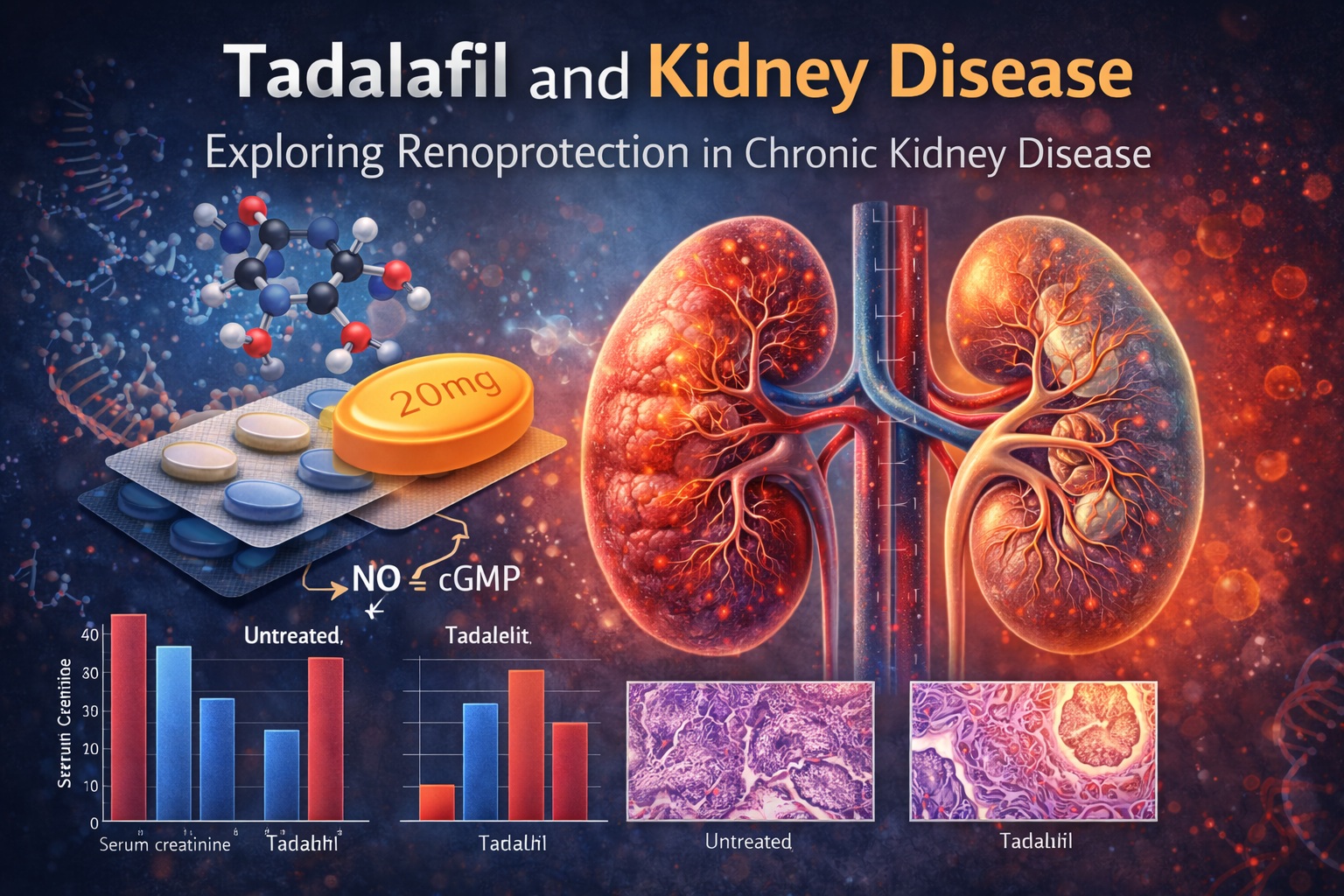
The study provides robust evidence of tadalafil’s beneficial effects on renal function. After eight weeks of high-salt exposure, untreated animals exhibited significant increases in serum creatinine and urinary protein levels—markers of impaired kidney function.
In contrast, rats treated with tadalafil—both low and high doses—showed markedly improved outcomes. As illustrated in the graph on page 5, serum creatinine levels in treated groups remained comparable to those in the normal-salt group, indicating preservation of renal function .
Similarly, urinary protein excretion, which increased dramatically in the high-salt group, was significantly reduced in tadalafil-treated animals. This reduction in proteinuria is particularly important, as it reflects improved integrity of the glomerular filtration barrier.
Notably, these benefits were observed even at low doses of tadalafil, suggesting that its renoprotective effects are not solely dependent on systemic hemodynamic changes.
Structural Preservation: Protecting the Kidney from Within
Beyond functional markers, the study offers compelling histological evidence of renal protection. Glomerulosclerosis—a key indicator of chronic damage—was significantly reduced in tadalafil-treated groups.
The microscopic images on page 5 reveal striking differences between groups. In untreated animals, glomeruli appeared distorted and sclerotic, whereas those in tadalafil-treated rats retained a more normal architecture .
Electron microscopy further highlights these effects. As shown in Figure 3 on page 6, tadalafil preserved the integrity of endothelial cells and podocyte foot processes—structures essential for filtration . In untreated animals, these structures were disrupted, with evidence of detachment and effacement.
This structural preservation is not merely cosmetic. It underpins the functional improvements observed, reinforcing the concept that tadalafil acts at both cellular and systemic levels.
Fibrosis and Molecular Signaling: The Role of PAI1
Renal fibrosis represents the final common pathway of CKD progression. It is driven by activation of myofibroblasts and deposition of extracellular matrix proteins, leading to irreversible scarring.
In this study, fibrosis was assessed using α-smooth muscle actin (αSMA) staining, a marker of myofibroblast activity. As shown in the histological panels on page 7, αSMA expression was markedly increased in the high-salt group but significantly reduced in tadalafil-treated animals .
At the molecular level, these changes were associated with alterations in PAI1 (plasminogen activator inhibitor-1) expression. PAI1 is a key regulator of fibrosis, promoting extracellular matrix accumulation and inhibiting its degradation.
Tadalafil treatment led to a reduction in PAI1 mRNA levels, suggesting that its antifibrotic effects may be mediated through modulation of this pathway. While the exact mechanisms remain to be fully elucidated, the findings provide a plausible link between PDE5 inhibition and fibrosis attenuation.
Blood Pressure: Cause, Effect, or Secondary Player?
One of the most intriguing aspects of the study is the relationship between tadalafil’s renoprotective effects and blood pressure reduction. While high-dose tadalafil significantly lowered blood pressure, low-dose treatment did not produce a comparable effect.
Despite this, both doses conferred similar improvements in renal function and structure. This observation suggests that the benefits of tadalafil may be, at least in part, independent of its antihypertensive properties.
This distinction is clinically significant. It implies that tadalafil may offer renoprotection even in patients whose blood pressure is already controlled by other means. In other words, it may complement existing therapies rather than replace them.
However, the study acknowledges limitations in blood pressure measurement, noting that the tail-cuff method may lack sensitivity. Further research using more precise techniques is needed to clarify this relationship.
Clinical Implications: From Bench to Bedside
The translation of these findings into clinical practice requires careful consideration. While animal models provide valuable insights, human physiology introduces additional complexities.
Nevertheless, the implications are promising. Tadalafil is already widely used, with a well-characterized safety profile. Its potential repurposing for CKD could accelerate clinical adoption, bypassing many of the barriers associated with novel drug development.
For clinicians, the key takeaway is the importance of looking beyond traditional indications. Drugs developed for one purpose may have unexpected benefits in entirely different domains—a reminder that pharmacology is as much about discovery as it is about application.
At the same time, caution is warranted. PDE5 inhibitors are not without risks, including hypotension and rare visual complications. Patient selection and monitoring will be critical in any future clinical application.
Conclusion: A New Chapter in Renal Pharmacotherapy
The study provides compelling evidence that tadalafil, a familiar drug, may hold untapped potential in the management of chronic kidney disease. By preserving renal structure, reducing proteinuria, and attenuating fibrosis, it addresses multiple facets of disease progression.
Perhaps most importantly, it challenges the conventional view that renoprotection is synonymous with blood pressure control. Tadalafil appears to operate through additional mechanisms, offering a more nuanced approach to therapy.
While further research is needed to confirm these findings in humans, the implications are clear. The future of CKD treatment may not lie solely in new drugs, but in new ways of using the ones we already have.
FAQ: Key Questions About Tadalafil and Kidney Disease
1. Can tadalafil improve kidney function in humans?
Current evidence comes primarily from animal studies. Human trials are needed to confirm its effectiveness.
2. Does tadalafil protect the kidneys only by lowering blood pressure?
No. The study suggests renoprotective effects even at doses that do not significantly reduce blood pressure.
3. What is the role of PAI1 in kidney disease?
PAI1 promotes fibrosis by inhibiting matrix degradation. Reducing its expression may help prevent kidney scarring.
4. Is tadalafil safe for patients with CKD?
It may be safe in selected patients, but clinical evaluation is essential due to potential side effects.
5. Could tadalafil replace standard CKD treatments?
Unlikely. It is more likely to serve as an adjunct therapy rather than a replacement.