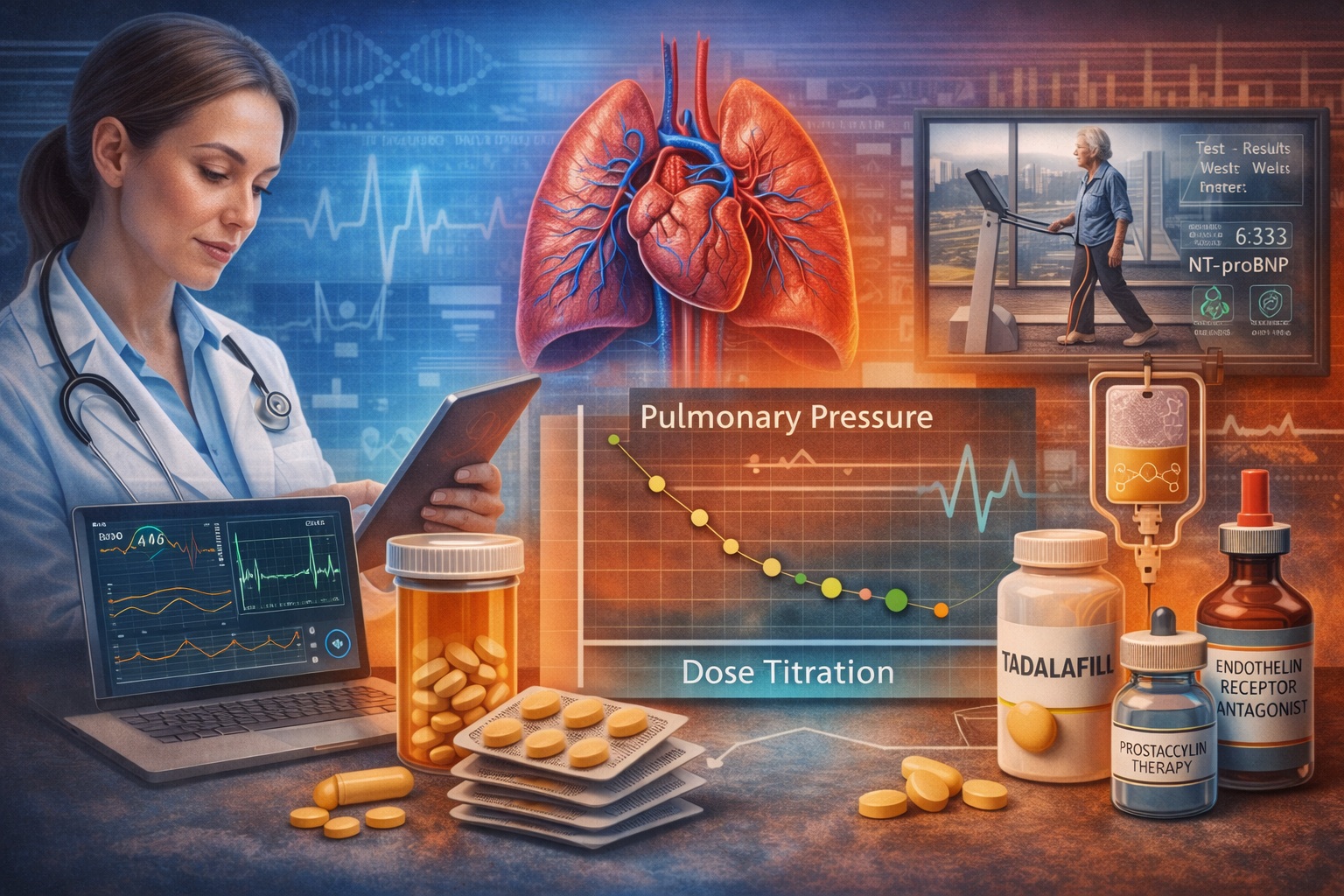
Pulmonary arterial hypertension (PAH) is a progressive and potentially fatal disease characterized by increased pulmonary vascular resistance and remodeling of the pulmonary arteries. Despite major advances in targeted therapies, the management of PAH remains complex and highly individualized. Modern treatment strategies focus not only on choosing the correct pharmacological class but also on how therapy is introduced, adjusted, and optimized over time. This process—known as drug titration—is often the difference between merely prescribing medication and achieving meaningful clinical improvement.
While treatment algorithms for PAH have evolved substantially, real-world clinical experience continues to emphasize that successful therapy depends on careful dosing adjustments. Rapid escalation may provoke adverse effects, whereas insufficient titration may leave patients undertreated. Physicians must therefore balance pharmacological efficacy with tolerability, a task that requires both clinical judgment and an understanding of drug-specific pharmacodynamics.
This article explores the principles of titration in pulmonary arterial hypertension therapeutics, highlighting practical strategies used by experienced clinicians. Particular attention will be given to targeted agents such as endothelin receptor antagonists, prostacyclin pathway therapies, and phosphodiesterase type-5 inhibitors—including tadalafil, which has become an important component of modern PAH treatment. Ultimately, titration is less a rigid protocol and more a carefully guided process—one that blends physiology, pharmacology, and clinical insight.
Understanding Pulmonary Arterial Hypertension: Pathophysiology and Therapeutic Targets
Pulmonary arterial hypertension is defined by elevated pulmonary artery pressure resulting from progressive narrowing and remodeling of small pulmonary arteries. Over time, this increased vascular resistance places excessive strain on the right ventricle, eventually leading to right-sided heart failure if untreated. Although the disease may appear primarily vascular, it represents a complex interplay of endothelial dysfunction, smooth muscle proliferation, inflammation, and impaired vasodilation.
At the molecular level, three principal signaling pathways are involved in PAH pathogenesis: the endothelin pathway, the nitric oxide pathway, and the prostacyclin pathway. Each of these pathways contributes to vascular tone and remodeling, and modern PAH therapies aim to correct their imbalance.
Endothelin-1, a potent vasoconstrictor peptide, is overexpressed in PAH and contributes to sustained pulmonary vasoconstriction and vascular proliferation. Endothelin receptor antagonists (ERAs) counteract this process by blocking endothelin receptors in pulmonary vascular smooth muscle.
The nitric oxide pathway plays a crucial role in vascular relaxation through cyclic guanosine monophosphate (cGMP). In PAH, nitric oxide signaling is impaired, leading to reduced vasodilation. Phosphodiesterase type-5 inhibitors such as tadalafil enhance cGMP activity by preventing its breakdown, thereby promoting pulmonary vasodilation and improving hemodynamics.
The prostacyclin pathway represents another key regulator of pulmonary vascular tone. Prostacyclin analogs and receptor agonists mimic endogenous prostacyclin activity, producing vasodilation while also inhibiting platelet aggregation and vascular proliferation.
Modern PAH management frequently involves targeting multiple pathways simultaneously, which explains why combination therapy has become the standard of care in many patients. However, combining therapies introduces another challenge: determining the appropriate dose for each medication. This is where titration becomes critically important.
Why Titration Matters in PAH Treatment
The concept of titration may appear straightforward: gradually increase a medication’s dose until the desired therapeutic effect is achieved. In reality, titration in PAH therapy is far more nuanced. Each drug class has a distinct pharmacological profile, and patients with PAH often present with significant hemodynamic instability, making aggressive dosing strategies potentially hazardous.
One of the primary goals of titration is to maximize therapeutic benefit while minimizing adverse effects. Many PAH medications can produce systemic vasodilation, headache, hypotension, flushing, or gastrointestinal symptoms. If doses are increased too rapidly, patients may discontinue therapy due to intolerance before therapeutic levels are achieved.
Equally problematic is under-titration. Subtherapeutic dosing may leave pulmonary vascular resistance inadequately controlled, allowing disease progression despite apparent treatment. Clinicians must therefore navigate a delicate middle path—advancing therapy steadily but cautiously.
Another reason titration is crucial lies in the heterogeneity of PAH patients. Age, comorbidities, liver function, renal status, and concurrent medications all influence drug metabolism and tolerance. A dosing strategy that works perfectly for one patient may be unsuitable for another.
Furthermore, PAH therapies often exert their maximal benefits only after several weeks of continuous exposure. Rapid dose escalation may not accelerate clinical improvement but may significantly increase side effects. Experienced clinicians therefore recognize that patience, rather than haste, is often the most effective titration strategy.
In short, titration is not merely about increasing a number on a prescription pad. It is about guiding the patient safely toward the optimal therapeutic window.
Principles of Safe and Effective Drug Titration
Successful titration begins with a thorough baseline evaluation. Before initiating therapy, clinicians assess disease severity using parameters such as functional class, exercise capacity, echocardiographic findings, and hemodynamic measurements obtained through right heart catheterization. These baseline data provide a reference point against which treatment response can later be evaluated.
Once therapy begins, titration should proceed systematically. Dose adjustments are typically performed at predefined intervals, allowing sufficient time for the patient’s body to adapt to each incremental change. The timing varies depending on the medication class and route of administration.
During this process, clinicians monitor both clinical improvement and treatment tolerance. Improvements may include increased exercise capacity, reduced dyspnea, improved biomarkers, and better hemodynamic measurements. At the same time, side effects must be carefully tracked.
Key factors considered during titration include:
- Symptom response and functional class
- Blood pressure and heart rate stability
- Laboratory monitoring, including liver function tests when relevant
- Patient-reported tolerability and quality of life
These elements guide clinicians in deciding whether to continue escalation, maintain the current dose, or occasionally reduce therapy.
An often overlooked aspect of titration is patient education. Individuals with PAH must understand that medication adjustments are a deliberate process rather than a sign of treatment failure. When patients are informed about potential side effects and the rationale behind gradual dosing increases, adherence improves significantly.
Titration Strategies for Major Classes of PAH Therapies
Different classes of PAH medications require different titration approaches. Understanding these distinctions is essential for optimizing treatment outcomes.
Endothelin receptor antagonists are typically introduced at standard starting doses with relatively limited titration requirements. However, clinicians must monitor liver function and hemoglobin levels during therapy, as these agents can occasionally affect hepatic metabolism and erythropoiesis.
Phosphodiesterase type-5 inhibitors represent another cornerstone of PAH therapy. Among these agents, tadalafil has gained particular attention due to its once-daily dosing and long half-life. In PAH treatment, tadalafil enhances pulmonary vasodilation by preserving cyclic GMP signaling within vascular smooth muscle cells.
Titration of tadalafil is generally straightforward compared with other PAH therapies. Many patients begin at a moderate dose that can be increased based on clinical response and tolerability. Because tadalafil’s pharmacokinetics provide sustained plasma levels, dose adjustments typically occur over several weeks rather than days.
Prostacyclin pathway therapies present a more complex titration challenge. Intravenous or subcutaneous prostacyclin analogs require extremely careful dose escalation because they are potent vasodilators with narrow therapeutic windows. Patients may experience headaches, jaw pain, flushing, or gastrointestinal discomfort during titration.
Despite these challenges, prostacyclin titration remains one of the most effective ways to improve pulmonary hemodynamics in severe PAH. Clinicians often describe the process as an exercise in persistence: small incremental increases, careful monitoring, and gradual achievement of the therapeutic target.
Combination Therapy and Sequential Escalation
The modern management of pulmonary arterial hypertension increasingly relies on combination therapy, targeting multiple signaling pathways simultaneously. While this approach improves clinical outcomes, it also complicates the titration process.
When several medications are introduced, clinicians must decide whether to begin them simultaneously or sequentially. Simultaneous initiation can produce rapid hemodynamic improvement but increases the risk of overlapping side effects. Sequential initiation allows clearer attribution of adverse reactions but may delay maximal therapeutic benefit.
In practice, treatment strategies often combine both approaches. Patients with advanced disease may begin with dual therapy targeting the endothelin and nitric oxide pathways. Over time, additional agents such as prostacyclin analogs may be introduced if treatment goals are not achieved.
Sequential titration requires careful planning. Each medication should reach a stable dose before another therapy is introduced, allowing clinicians to evaluate its individual effect. For example, a patient receiving an endothelin receptor antagonist may subsequently begin tadalafil, followed later by a prostacyclin pathway agent if necessary.
Combination therapy introduces an important clinical reality: drug interactions and cumulative vasodilatory effects. Blood pressure monitoring becomes essential, as excessive systemic vasodilation may produce dizziness or hypotension. Experienced clinicians therefore escalate therapy gradually, ensuring that the patient’s cardiovascular system adapts safely.
The objective of combination titration is not simply to add more drugs but to create a balanced pharmacological strategy that targets multiple disease mechanisms simultaneously.
Clinical Monitoring During the Titration Process
Titration is inseparable from monitoring. Without regular clinical evaluation, dose adjustments become guesswork rather than evidence-based decision making.
Functional assessment plays a central role in monitoring PAH patients. Tools such as the six-minute walk test provide valuable information about exercise capacity and disease progression. Improvements in walking distance often correlate with better pulmonary hemodynamics and quality of life.
Biomarkers also contribute to titration decisions. Levels of natriuretic peptides, particularly NT-proBNP, reflect right ventricular strain and can indicate whether therapy is effectively reducing cardiac workload. A declining biomarker level during titration generally suggests improved cardiovascular status.
Imaging studies provide another important layer of information. Echocardiography allows clinicians to evaluate right ventricular function, pulmonary pressures, and structural adaptations to therapy. Although echocardiography does not replace invasive hemodynamic measurements, it offers a non-invasive way to track disease trajectory.
Finally, clinicians must never underestimate the importance of patient-reported symptoms. Dyspnea, fatigue, dizziness, and exercise tolerance often provide the earliest clues about whether titration is proceeding successfully. In the management of PAH, attentive listening remains one of the most valuable diagnostic tools.
Practical Challenges and Real-World Considerations
While clinical guidelines provide valuable frameworks, real-world titration rarely follows a perfectly predictable path. Patients may experience unexpected side effects, concurrent illnesses, or logistical barriers that complicate therapy adjustments.
For example, prostacyclin therapies requiring infusion pumps demand extensive patient education and support. Even minor technical issues can influence dosing accuracy and therapeutic stability. In these situations, multidisciplinary care teams—including nurses and pharmacists—play a crucial role.
Adherence represents another challenge. PAH therapies often involve complex dosing schedules, and side effects during titration can discourage patients from continuing treatment. Clear communication about expected symptoms and the long-term benefits of therapy can help mitigate this issue.
Financial considerations also influence titration strategies. Some PAH medications are extremely costly, and insurance authorization may determine which drugs can be initiated or escalated. Clinicians must sometimes balance ideal pharmacological strategies with practical healthcare system constraints.
Despite these obstacles, the fundamental principle remains unchanged: titration should always aim to move patients toward optimal disease control while preserving quality of life.
The Future of Personalized PAH Titration
The future of pulmonary arterial hypertension treatment is moving toward increasingly personalized approaches. Advances in molecular diagnostics, genetic profiling, and digital monitoring may soon allow clinicians to tailor titration strategies to each patient’s unique physiological characteristics.
Emerging technologies may also provide new ways to monitor treatment response in real time. Wearable devices capable of tracking cardiopulmonary parameters could offer continuous insight into how patients respond to therapy adjustments. Such tools may help clinicians detect early signs of deterioration or therapeutic success.
Pharmacogenomics may also influence titration strategies. Individual genetic variations can affect drug metabolism, potentially explaining why some patients tolerate rapid dose escalation while others require more gradual adjustments. Understanding these differences could allow clinicians to predict optimal dosing strategies from the outset.
As research advances, medications like tadalafil will likely remain integral components of PAH therapy, but their use may become more precisely calibrated to each patient’s biological profile. Ultimately, the goal is not merely to treat PAH but to manage it with the precision and adaptability that modern medicine increasingly demands.
FAQ
What is drug titration in pulmonary arterial hypertension?
Drug titration refers to the gradual adjustment of medication doses to achieve the best therapeutic effect while minimizing side effects. In PAH, this process is essential because many medications influence vascular tone and must be increased carefully.
Why is tadalafil used in PAH treatment?
Tadalafil is a phosphodiesterase type-5 inhibitor that enhances nitric oxide signaling in the pulmonary vasculature. By increasing cyclic GMP levels, it promotes vasodilation and helps reduce pulmonary artery pressure.
How long does titration usually take in PAH therapy?
The duration varies depending on the medication class and patient response. Some therapies reach target doses within weeks, while others—especially prostacyclin agents—may require months of careful dose escalation to achieve optimal therapeutic levels.