Pharmacokinetics is often perceived as an abstract discipline confined to curves and clearance values. In reality, it is the quiet architect of clinical safety. When a drug moves through the body, it negotiates with physiology — and physiology changes with age, chronic disease, and organ impairment. Understanding this dialogue is not optional; it is the foundation of rational dosing.
Tadalafil, a selective phosphodiesterase type 5 (PDE5) inhibitor, is widely used for erectile dysfunction and other vascular conditions. Its long half-life and hepatic metabolism via CYP3A4 make it pharmacologically elegant — and potentially sensitive to intrinsic patient factors. A series of six well-designed clinical pharmacology studies explored how gender, age, diabetes mellitus, renal impairment, end-stage renal failure (ESRF), and hepatic dysfunction influence tadalafil disposition and tolerability .
What emerges is a story both reassuring and instructive: most intrinsic factors exert modest effects, but renal dysfunction significantly alters systemic exposure. Let us examine the evidence carefully and translate it into clinical clarity.
Baseline Pharmacokinetic Profile of Tadalafil
Before considering special populations, we must understand tadalafil in healthy individuals. Following oral administration, tadalafil is rapidly absorbed, with a median time to maximum concentration (tmax) of approximately 2 hours. Its pharmacokinetics are linear across therapeutic doses. The mean elimination half-life is about 17.5 hours, a feature that distinguishes it from shorter-acting PDE5 inhibitors and explains its prolonged duration of effect.
Plasma protein binding is high (approximately 94%), primarily to albumin and α1-acid glycoprotein. Clearance is predominantly hepatic, mediated by CYP3A4, producing a catechol metabolite that is extensively glucuronidated. Roughly one-third of the administered dose is excreted in urine, almost entirely as metabolites rather than parent compound .
This profile immediately suggests where vulnerability may lie: hepatic metabolism, renal elimination of metabolites, and alterations in clearance or distribution that accompany aging or systemic disease.
Gender: Similar Exposure, Minor Differences, No Clinical Consequence
Pharmacokinetic comparisons between healthy men and women revealed remarkably similar exposure after a 10 mg dose. At steady state during once-daily dosing, systemic exposure in females was approximately 13% higher than in males, but this difference narrowed when normalized for body weight .
The half-life and accumulation patterns were comparable. Steady state was achieved by day five of daily dosing. The modest female-to-male AUC ratio did not indicate clinically meaningful divergence.
From a mechanistic standpoint, this is unsurprising. Tadalafil is metabolized by CYP3A4, an enzyme not dramatically influenced by sex under normal physiological conditions. Minor differences in body composition and plasma protein levels may account for slight exposure variations.
Clinically, no dose adjustment based solely on gender is justified. The data support equal therapeutic expectations in men and women — an important point given tadalafil’s investigation in pulmonary arterial hypertension and systemic vascular disorders affecting both sexes.
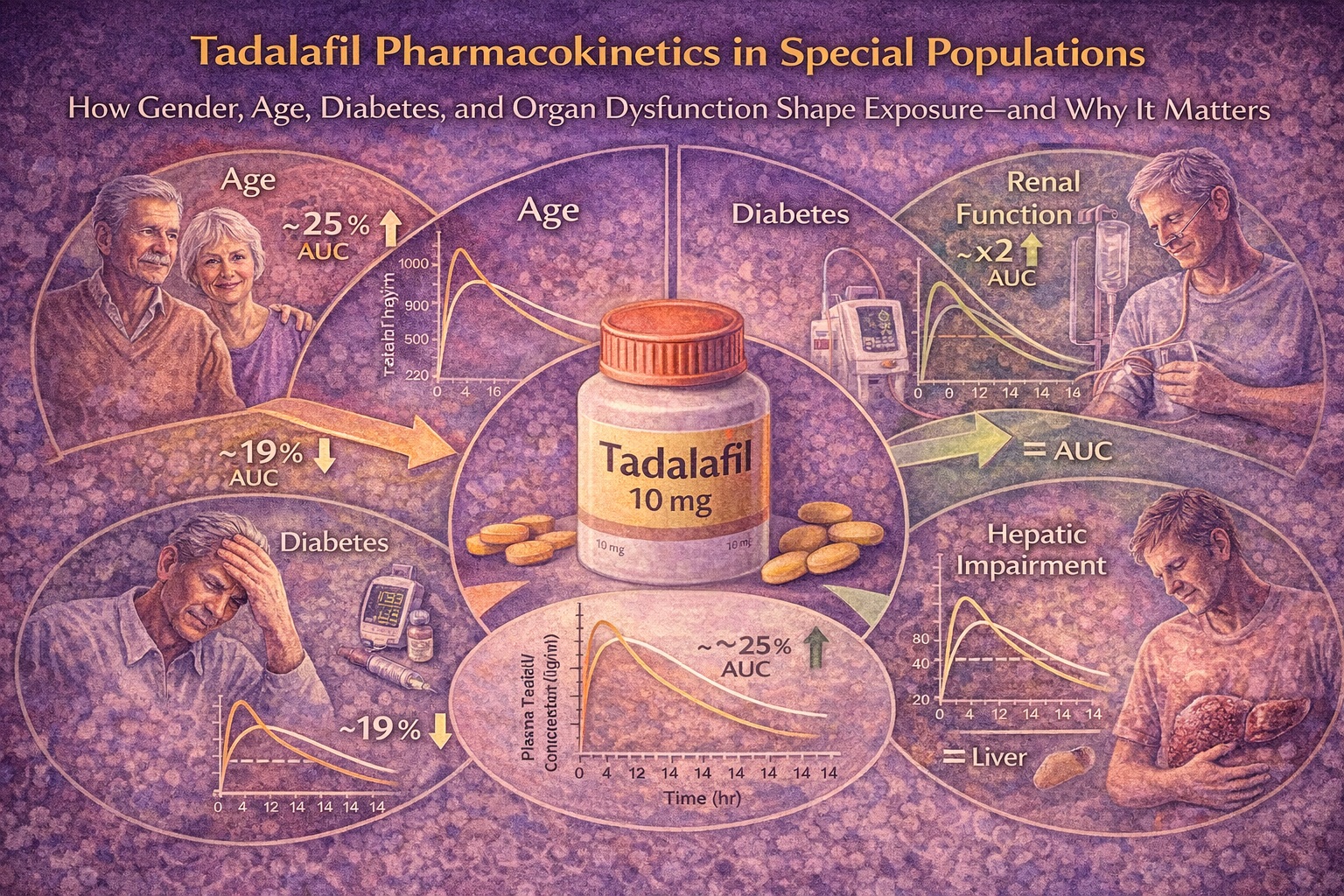
Age: A Mild Increase in Exposure Without Clinical Alarm
Aging affects hepatic blood flow, renal function, and body composition. It was therefore reasonable to question whether tadalafil exposure would rise meaningfully in elderly patients.
In the study comparing elderly men (65–80 years) with younger controls, the area under the concentration–time curve (AUC) was approximately 25% higher in older subjects . The elimination half-life was prolonged by roughly 5 hours. However, maximum concentration (Cmax) remained similar.
These findings are pharmacologically coherent. Age reduces hepatic blood flow more consistently than it alters intrinsic CYP3A4 activity. For a low-clearance drug such as tadalafil, modest decreases in clearance translate into moderate AUC increases.
Yet a 25% exposure increase, in the absence of safety concerns, does not mandate automatic dose reduction. Clinical trial data did not demonstrate increased adverse event rates solely attributable to age. Therefore, advanced age alone does not require dosage adjustment.
The practical message: respect age, but do not overreact to it.
Diabetes Mellitus: A Slight Reduction in Exposure
In patients with Type 1 or Type 2 diabetes mellitus receiving insulin therapy, systemic exposure to tadalafil was modestly lower — approximately 19% less than in healthy controls . The half-life was slightly shorter, and concentration–time curves showed only subtle differences.
This small reduction in AUC is clinically negligible. Importantly, the variability across individuals overlapped substantially between diabetic and non-diabetic cohorts. Diabetes, in itself, did not impair absorption or clearance in a clinically meaningful manner.
Given the high prevalence of erectile dysfunction in diabetic patients, this finding is particularly reassuring. No dose modification is necessary solely on the basis of diabetes mellitus.
One could argue that the more significant determinant of efficacy in diabetes is endothelial dysfunction rather than pharmacokinetics. Exposure is preserved; responsiveness may vary.
Renal Impairment: The Decisive Factor
Renal dysfunction emerged as the most influential intrinsic factor affecting tadalafil pharmacokinetics.
In subjects with mild or moderate renal insufficiency, systemic exposure approximately doubled compared with healthy controls . The elimination half-life was substantially prolonged (up to 26 hours in mild impairment). Cmax increased by approximately 20%.
Interestingly, tadalafil itself undergoes negligible renal clearance. The mechanism behind increased exposure likely involves reduced hepatic intrinsic clearance or altered first-pass metabolism associated with uremic physiology. Renal disease is known to affect non-renal drug metabolism — a reminder that organs do not function in isolation.
The glucuronide metabolite showed even greater accumulation, as expected for a renally eliminated compound. AUC for the metabolite increased up to 3.6-fold in moderate impairment .
Tolerability data provided practical confirmation: a 10 mg dose was poorly tolerated in subjects with moderate renal dysfunction in early studies, largely due to musculoskeletal adverse events. Subsequent lower-dose administration improved tolerability.
Regulatory guidance appropriately reflects these findings:
- Mild renal impairment: no adjustment required.
- Moderate impairment: dose limitation recommended.
- Severe impairment or dialysis: further restriction advised.
Renal function, unlike age or diabetes, materially alters tadalafil exposure. Here, pharmacokinetics truly matter.
End-Stage Renal Failure and Hemodialysis
In patients undergoing hemodialysis, tadalafil exposure increased approximately twofold relative to healthy individuals . The metabolite AUC increased more markedly (over threefold).
Hemodialysis contributed negligibly to tadalafil elimination. This is entirely predictable given the drug’s high protein binding and large distribution volume.
Importantly, single doses up to 20 mg were tolerated in dialysis patients, though careful clinical evaluation remains prudent. The absence of dialysis-mediated clearance underscores the need for preemptive dose adjustment rather than reliance on extracorporeal removal.
Hepatic Impairment: A Surprisingly Modest Effect
Given that tadalafil is metabolized primarily via CYP3A4, hepatic impairment might be expected to increase systemic exposure. Surprisingly, mild and moderate hepatic dysfunction did not produce clinically significant increases in AUC .
Cmax values tended to be slightly lower, and half-life was modestly prolonged with increasing severity. Apparent distribution volume increased with worsening hepatic disease, possibly reflecting altered tissue binding rather than changes in intrinsic clearance.
The absence of substantial AUC elevation suggests preserved metabolic capacity in mild-to-moderate cirrhosis or compensatory mechanisms mitigating exposure increases.
However, because severe hepatic impairment was represented by only one subject, robust conclusions cannot be drawn in that population. Prudence remains warranted.
Safety Profile Across Special Populations
Across all studies, tadalafil demonstrated favorable tolerability. No serious adverse events or discontinuations due to adverse effects were reported .
The most commonly reported adverse events were:
- Headache
- Back pain
- Myalgia
These are well-established class effects of PDE5 inhibition and were generally mild to moderate in severity. The incidence in moderate renal impairment appeared higher in small cohorts but did not reflect a consistent pattern in dialysis patients.
No clinically significant changes in laboratory parameters, ECG findings, or vital signs were observed.
The safety message is consistent: exposure elevation in renal impairment warrants caution, but the overall risk profile remains manageable with appropriate dosing.
Clinical Implications: Translating Pharmacokinetics into Practice
When integrating these findings into everyday care, clarity is essential.
Tadalafil pharmacokinetics are resilient to most intrinsic patient factors. Gender, advanced age, and diabetes mellitus do not necessitate routine dose adjustment. Mild-to-moderate hepatic impairment does not substantially alter exposure.
Renal impairment, however, meaningfully increases systemic levels. Here, dose modification is evidence-based and necessary.
The broader lesson is conceptual: intrinsic factors do not uniformly alter pharmacokinetics. Mechanistic understanding must guide decisions, not assumptions.
Conclusion
The comprehensive evaluation of tadalafil pharmacokinetics across diverse populations demonstrates a reassuring stability of exposure in most physiological variations. Age and diabetes exert minimal impact. Gender differences are negligible. Mild and moderate hepatic impairment do not significantly elevate systemic exposure.
Renal dysfunction stands apart as the principal determinant of altered pharmacokinetics, doubling systemic exposure and prolonging elimination. Dose adjustment in this setting is rational and supported by both pharmacokinetic and tolerability data.
In pharmacotherapy, precision lies not in complexity but in disciplined interpretation of evidence. Tadalafil’s pharmacokinetic profile, when understood correctly, enables confident and individualized prescribing.
FAQ
1. Does elderly age require lowering the tadalafil dose?
No. Although elderly patients show about a 25% increase in exposure, this difference is not clinically significant and does not require routine dose adjustment .
2. Why does renal impairment increase tadalafil levels if the drug is not primarily renally cleared?
Renal dysfunction can reduce hepatic metabolic capacity and alter intrinsic clearance, leading to higher systemic exposure. Additionally, metabolites accumulate due to reduced renal elimination .
3. Should tadalafil be avoided in patients with liver disease?
Mild and moderate hepatic impairment do not significantly increase systemic exposure. Standard doses (up to 10 mg studied) are generally acceptable, but caution is advised in severe hepatic impairment due to limited data .