Introduction: When Formulation Meets Function
Erectile dysfunction (ED) is not a rare inconvenience; it is a global medical condition with profound physiological and psychosocial consequences. Prevalence increases sharply with age, affecting up to 40% of men between 60 and 69 years and more than half of men over 70 . Beyond intimacy, ED correlates with cardiovascular risk, depression, and reduced quality of life. It demands not only effective pharmacology but also practical delivery.
Tadalafil, a selective phosphodiesterase type 5 (PDE5) inhibitor, has been a cornerstone of ED therapy since its introduction in 2003 . Its distinguishing feature is a prolonged half-life of approximately 17 hours, allowing sustained efficacy and greater spontaneity compared to shorter-acting agents.
Yet, even an excellent molecule can be limited by its formulation. Conventional film-coated tablets (FCTs) require water for swallowing. For younger men this is trivial. For elderly patients with dysphagia—or for anyone seeking discretion—it is not. The development of a tadalafil orodispersible film (ODF) reflects a broader pharmaceutical evolution: improving convenience without compromising pharmacokinetic integrity.
The study published in Drug Design, Development and Therapy rigorously compared the pharmacokinetics, safety, and tolerability of a newly developed tadalafil ODF with the established film-coated tablet formulation . The results provide compelling evidence of bioequivalence—and a glimpse into the future of patient-centered drug delivery.
Study Design: Bioequivalence Under Methodological Discipline
The investigators employed an open-label, randomized, two-period, two-sequence crossover design in healthy male volunteers aged 19–45 years . Each subject received both formulations—20 mg tadalafil ODF and 20 mg tadalafil FCT—with a 7-day washout between treatments.
This crossover model is the gold standard for bioequivalence testing. Each participant serves as his own control, minimizing inter-individual variability. Blood samples were collected from predose up to 72 hours post-administration, allowing full characterization of absorption and elimination phases.
The analytical methodology deserves mention. Plasma tadalafil concentrations were quantified using validated liquid chromatography–tandem mass spectrometry, with calibration linear from 2 to 1,000 μg/L and precision coefficients of variation below 7% . Such methodological rigor ensures that observed pharmacokinetic equivalence reflects true biological similarity rather than analytic artifact.
Forty subjects were enrolled; 36 completed the study. Demographic characteristics were balanced between sequences, and no carryover effect was detected .
Pharmacokinetic Outcomes: Absorption Without Compromise
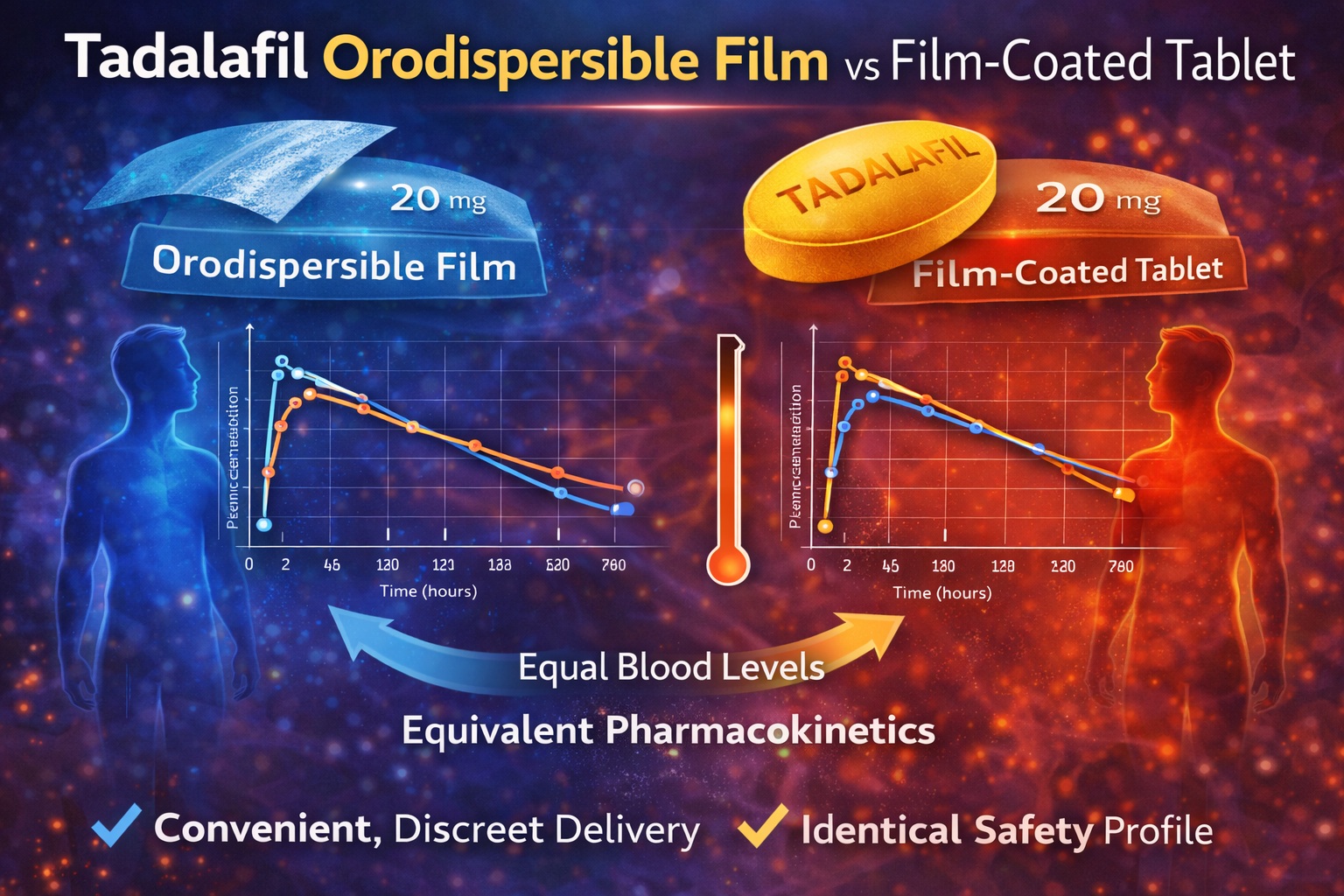
The primary pharmacokinetic endpoints were maximum plasma concentration (Cmax) and area under the plasma concentration–time curve to last measurable concentration (AUClast).
The geometric mean ratio (ODF/FCT) for:
- Cmax was 0.927 (90% CI 0.882–0.974)
- AUClast was 0.972 (90% CI 0.918–1.029)
Both confidence intervals fell entirely within the conventional bioequivalence range of 0.80–1.25.
Additional parameters confirmed similarity:
- Median Tmax: 2.5 hours for both formulations
- Half-life (t1/2): ~17.5 hours
- Apparent clearance (CL/F): comparable between groups
The mean concentration–time curves for ODF and FCT were superimposable from 0 to 72 hours . Individual subject comparisons revealed no systematic deviation in exposure.
In pharmacokinetic terms, the orodispersible film behaves as the tablet behaves. It neither accelerates absorption dramatically nor compromises systemic exposure. It simply delivers tadalafil effectively—without water.
Understanding the ODF Platform: Mechanism and Rationale
An orodispersible film is a thin polymeric strip that dissolves rapidly when placed on the tongue. Unlike sublingual formulations designed for transmucosal absorption, most ODF systems dissolve in saliva and are swallowed, with gastrointestinal absorption following conventional pathways .
The technological advantages include:
- Rapid disintegration (typically <1 minute)
- No need for water
- Reduced risk of choking
- Precise unit dosing
- Improved portability and discretion
Taste masking and polymer selection are critical. Tadalafil’s physicochemical properties differ from sildenafil and vardenafil, yet modern film technology accommodates these challenges.
The key question was whether dissolution in the oral cavity would alter the rate or extent of absorption. The data demonstrate that it does not.
Food Effects, Smoking, and Age: External Validity
Previous studies have shown that tadalafil absorption is unaffected by food intake . This characteristic reduces variability and enhances practical flexibility.
Population pharmacokinetic analyses have also indicated that neither age nor smoking significantly alters systemic tadalafil exposure . Dose adjustment is generally unnecessary on these bases.
Although this bioequivalence study enrolled healthy young men, these broader data support extrapolation to older ED populations. Nevertheless, confirmation in elderly cohorts would strengthen translational confidence.
Safety and Tolerability: Comparable and Predictable
No serious adverse events occurred during the study .
Reported adverse drug reactions were mild and consistent with known tadalafil effects:
- Headache
- Hot flush
- Back pain
- Chest discomfort
- Palpitations
The incidence of adverse events did not differ significantly between ODF and FCT groups. No clinically meaningful changes in laboratory values, vital signs, or ECG findings were observed.
From a safety perspective, the film behaves as the tablet behaves.
Clinical Implications: Beyond Bioequivalence
Bioequivalence establishes interchangeability. Clinical value extends further.
An orodispersible tadalafil film offers practical advantages:
- Improved compliance in patients with swallowing difficulties
- Greater discretion in social settings
- Reduced dependence on water access
- Enhanced portability
These features may appear minor, yet medication adherence is profoundly influenced by convenience. In ED treatment—where timing, spontaneity, and privacy matter—formulation can influence satisfaction as much as pharmacodynamics.
It is worth noting that tadalafil’s long half-life already differentiates it from shorter-acting PDE5 inhibitors. Combining prolonged efficacy with simplified administration strengthens its clinical appeal.
Methodological Strengths and Limitations
The crossover design, sufficient washout (more than five half-lives), and comprehensive sampling window (72 hours) enhance internal validity .
Limitations include:
- Enrollment limited to healthy young males
- Single-dose evaluation only
- Modest sample size
However, these parameters are standard for regulatory bioequivalence studies.
The Broader Context: Evolution of PDE5 Inhibitor Delivery
Orodispersible formulations have already been developed for sildenafil and vardenafil . Tadalafil’s distinct pharmacokinetic profile—particularly its extended half-life—makes preservation of exposure particularly important.
Unlike drugs requiring rapid onset via mucosal absorption, tadalafil benefits from systemic consistency rather than speed alone. The ODF formulation respects that principle.
Pharmaceutical innovation increasingly focuses on patient-centered design. Convenience, discretion, and adherence are no longer secondary considerations—they are central therapeutic determinants.
Conclusion: Equivalent Exposure, Enhanced Convenience
The study conclusively demonstrates that a 20 mg tadalafil orodispersible film is bioequivalent to the conventional 20 mg film-coated tablet .
Cmax, AUClast, AUCinf, half-life, and clearance parameters were statistically comparable. Safety and tolerability profiles were indistinguishable. The 90% confidence intervals for key exposure metrics fell squarely within accepted bioequivalence margins.
In practical terms, tadalafil ODF delivers the same molecule, at the same exposure, with the same safety profile—while offering greater convenience.
Pharmacology remains unchanged. Delivery evolves. And sometimes, that evolution is precisely what patients need.
FAQ
1. Is tadalafil orodispersible film as effective as the tablet?
Yes. The study demonstrated bioequivalence between the two formulations. Systemic exposure (Cmax and AUC) fell within accepted regulatory equivalence ranges.
2. Does the orodispersible film act faster?
The median time to peak concentration (Tmax) was identical (2.5 hours) for both formulations. The ODF does not significantly accelerate systemic absorption.
3. Who benefits most from the ODF formulation?
Patients who have difficulty swallowing tablets, prefer discretion, or want dosing flexibility without water may benefit most. The pharmacokinetic and safety profiles remain comparable to the tablet form.