Introduction: When Oxygen Falls, So Does Function
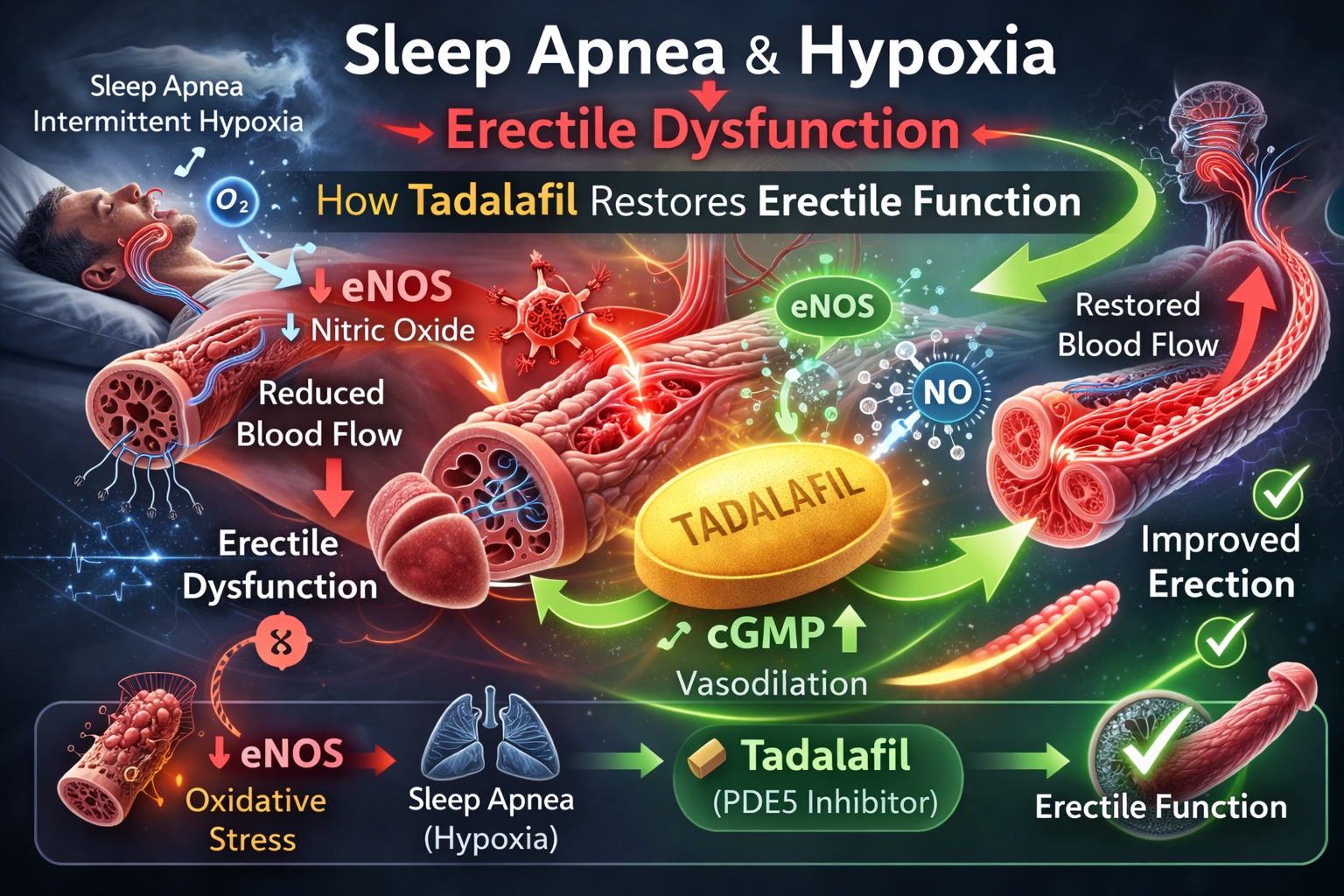
Erectile dysfunction (ED) is often framed as a vascular disorder of aging, diabetes, or atherosclerosis. Yet an underappreciated contributor lies in sleep physiology—specifically, obstructive sleep apnea (OSA). OSA affects 2–4% of adults and is characterized by repetitive upper airway obstruction during sleep, leading to chronic intermittent hypoxia (CIH) . Clinically, men with OSA exhibit a higher prevalence and severity of ED, and improvement with continuous positive airway pressure (CPAP) hints at a mechanistic link.
The experimental study published in American Journal of Respiratory and Critical Care Medicine provides compelling mechanistic evidence. Using a well-validated murine model of CIH designed to mimic moderate-to-severe sleep apnea, investigators demonstrated profound alterations in sexual behavior and erectile physiology. Importantly, these changes were partially reversible with tadalafil, a long-acting phosphodiesterase type 5 (PDE5) inhibitor .
This article synthesizes the experimental findings and translates them into clinical insight. We will explore how intermittent hypoxia disrupts erectile biology, why nitric oxide synthase (NOS) pathways are central, and what the response to tadalafil teaches us about the neurovascular architecture of erection. The message is clear: oxygen matters—and so does cGMP.
The Neurovascular Architecture of Erection: A Delicate Balance
Penile erection is not a simple hydraulic event; it is a coordinated neurovascular phenomenon. Central neural circuits within the medial preoptic area and paraventricular nucleus integrate sensory, olfactory, and cognitive inputs. Peripheral autonomic pathways regulate smooth muscle tone. At the tissue level, nitric oxide (NO) is the principal mediator of relaxation.
Neuronal nitric oxide synthase (nNOS) initiates erection by triggering cavernosal smooth muscle relaxation. Endothelial nitric oxide synthase (eNOS) sustains erection by maintaining blood inflow. NO stimulates guanylate cyclase, increasing cyclic guanosine monophosphate (cGMP), which lowers intracellular calcium and promotes vasodilation. Phosphodiesterase type 5 (PDE5) degrades cGMP, terminating the response .
Tadalafil, like other PDE5 inhibitors, blocks cGMP degradation, thereby amplifying and prolonging the NO-mediated signal. However, PDE5 inhibition presupposes an intact upstream NO pathway. If hypoxia impairs NOS expression or activity, erectile capacity may deteriorate—even before vascular disease becomes apparent.
Modeling Sleep Apnea: Chronic Intermittent Hypoxia in Mice
The investigators exposed C57BL/6 male mice to alternating oxygen concentrations of 21% and 7.8% every 360 seconds for 12 hours per day, replicating the cyclical desaturations observed in human OSA . Oxyhemoglobin nadirs approximated 76–78%, simulating moderate-to-severe apnea.
A key methodological strength was the use of telemetry to measure corpus spongiosum pressure (CSP) in freely behaving mice. This allowed quantification of spontaneous erections, mating behavior, and noncontact “psychogenic” responses without restraint artifacts.
The experimental design evaluated sexual behavior after five weeks of CIH, assessed NOS isoform expression in penile tissue after eight weeks, measured sex hormones, and examined the effects of tadalafil administration. The model therefore addressed central behavior, peripheral tissue biology, and pharmacologic responsiveness.
Behavioral Suppression Under Hypoxia: Libido and Performance Decline
The consequences of CIH were rapid and dramatic. After just one week of intermittent hypoxia, spontaneous erections decreased by 55% (P<0.001) and remained suppressed throughout the exposure period . Even six weeks of normoxic recovery did not fully restore baseline frequency.
Copulatory behavior deteriorated profoundly. Latency to mounting increased more than 60-fold, latency to intromission increased over 40-fold, and the sexual activity index decreased sixfold . Ejaculation frequently failed to occur.
Noncontact tests—designed to assess libido without physical interaction—also demonstrated marked suppression of CSP peaks during female exposure. This suggests that CIH affects not only erectile hemodynamics but central sexual motivation.
The rapid onset of dysfunction implies neural mechanisms. Chronic intermittent hypoxia has been shown in other studies to induce neuronal apoptosis, alter hypothalamic signaling, and increase sympathetic outflow. In this context, ED is not merely vascular—it is neurovascular.
Nitric Oxide Synthase Disruption: The Molecular Signature
Western immunoblotting revealed a significant reduction in endothelial NOS (eNOS) expression in penile tissue after eight weeks of CIH (P<0.01), while neuronal NOS (nNOS) and inducible NOS (iNOS) levels remained unchanged .
This selective reduction is clinically meaningful. nNOS initiates erection; eNOS sustains it. Decreased eNOS compromises maintenance of tumescence and impairs maximal rigidity. In the study, tumescence pressure declined during mating tests under CIH and improved after tadalafil administration .
Importantly, total testosterone levels remained stable after 8 and 24 weeks of hypoxia exposure. Estradiol increased only after prolonged exposure, but this did not account for early behavioral suppression . Testicular histology was normal.
The findings therefore implicate endothelial dysfunction—not hormonal deficiency—as the primary mechanism.
Sympathetic Overactivity and Peripheral Tone
CIH is known to increase sympathetic activity. Elevated sympathetic tone promotes smooth muscle contraction in penile arteries and trabecular tissue, antagonizing erection. Increased oxidative stress further disrupts NO bioavailability and may promote eNOS uncoupling.
The study’s authors note that sympathetic overactivity likely contributed to reduced tumescence pressures. Tadalafil partially reversed this effect, increasing tumescence pressure significantly in CIH-exposed mice .
Thus, ED under hypoxic stress likely arises from a triad: central neural impairment, endothelial dysfunction, and sympathetic dominance.
Tadalafil Intervention: Restoring the cGMP Axis
Administration of tadalafil after five weeks of CIH produced significant improvement in erectile function and sexual behavior . Spontaneous erections increased by nearly 39% in hypoxic mice, effectively normalizing activity. Latencies to mounting and intromission improved, and tumescence pressure rose.
Interestingly, tadalafil also enhanced behavioral parameters, not solely erectile hemodynamics. This raises the possibility that PDE5 inhibition may exert central effects, perhaps via improved cerebral perfusion or modulation of neural cGMP pathways.
However, tadalafil did not fully reverse structural or molecular deficits. It augmented the residual NO-cGMP pathway but did not directly restore eNOS expression. This distinction is critical: PDE5 inhibitors amplify existing signals; they do not create them de novo.
The response suggests that despite reduced eNOS, sufficient NO remained to permit pharmacologic potentiation. In severe endothelial compromise, PDE5 inhibitors may be less effective—a phenomenon occasionally observed in advanced OSA patients.
Translational Implications: Sleep Apnea and Clinical ED
The murine findings mirror clinical observations. Men with OSA exhibit higher rates of ED, and CPAP therapy improves sexual function in a subset of patients. Yet CPAP does not uniformly restore erectile capacity, and PDE5 inhibitors remain more effective in many cases.
The study supports a mechanistic link: CIH reduces eNOS expression and impairs neurovascular coordination. Tadalafil improves function but does not address underlying hypoxia. Therefore, optimal management of ED in OSA should include treatment of apnea alongside pharmacologic support.
Clinicians should recognize OSA as a modifiable risk factor for ED. Screening for sleep-disordered breathing in men presenting with unexplained ED may reveal an actionable diagnosis.
Oxidative Stress and Endothelial Vulnerability
CIH generates reactive oxygen species and oxidative stress. eNOS uncoupling and peroxynitrite formation may further diminish NO bioavailability. While iNOS levels did not increase in this model, oxidative stress likely contributed to endothelial dysfunction.
Penile tissue possesses substantial antioxidant capacity, but repetitive ischemia-reperfusion cycles during hypoxic episodes may overwhelm protective mechanisms. Over time, cumulative endothelial injury could impair vascular compliance.
The study therefore reinforces a broader principle: erectile tissue is exquisitely sensitive to oxidative imbalance. Interventions targeting oxidative stress may complement PDE5 inhibition in hypoxia-related ED.
Limitations and Experimental Context
As with all animal studies, caution is warranted in extrapolating directly to humans. The murine model isolates CIH without fully replicating sleep fragmentation, obesity, or metabolic comorbidities often present in OSA patients.
The duration of exposure was limited to eight weeks for NOS analysis and five weeks for behavioral assessment. Longer exposures may produce additional structural changes.
Nevertheless, the controlled experimental design provides strong mechanistic insight. The consistent reduction in eNOS and the reproducible behavioral deficits lend credibility to the conclusions.
Conclusion: Oxygen, Endothelium, and the Therapeutic Role of Tadalafil
The study conclusively demonstrates that chronic intermittent hypoxia—an essential feature of sleep apnea—induces erectile dysfunction in mice through reductions in endothelial nitric oxide synthase expression and neurobehavioral suppression .
Tadalafil substantially ameliorates these impairments by enhancing cGMP signaling, restoring tumescence pressure, and improving sexual performance. However, it does not correct the underlying hypoxic insult.
The clinical message is twofold:
First, ED in OSA is biologically plausible and mechanistically rooted in endothelial dysfunction.
Second, tadalafil remains an effective therapeutic adjunct, particularly when combined with correction of nocturnal hypoxia.
Erection is not merely a vascular reflex; it is a barometer of systemic oxygenation and endothelial health. When oxygen falls, function falters. When cGMP rises, performance recovers—at least partially.
FAQ
1. Does sleep apnea directly cause erectile dysfunction?
This experimental study demonstrates that chronic intermittent hypoxia—mimicking sleep apnea—reduces endothelial nitric oxide synthase expression and suppresses sexual behavior, providing strong mechanistic evidence that sleep apnea can directly impair erectile function.
2. Can tadalafil fully reverse hypoxia-related erectile dysfunction?
Tadalafil significantly improves erectile function by enhancing cGMP signaling. However, it does not correct the underlying hypoxia or fully restore endothelial NOS expression. Combined treatment of sleep apnea and ED is likely most effective.
3. Are testosterone levels affected by intermittent hypoxia?
In this study, testosterone levels remained unchanged after 8 and 24 weeks of hypoxia exposure, suggesting that ED under these conditions is primarily endothelial and neural rather than hormonal.